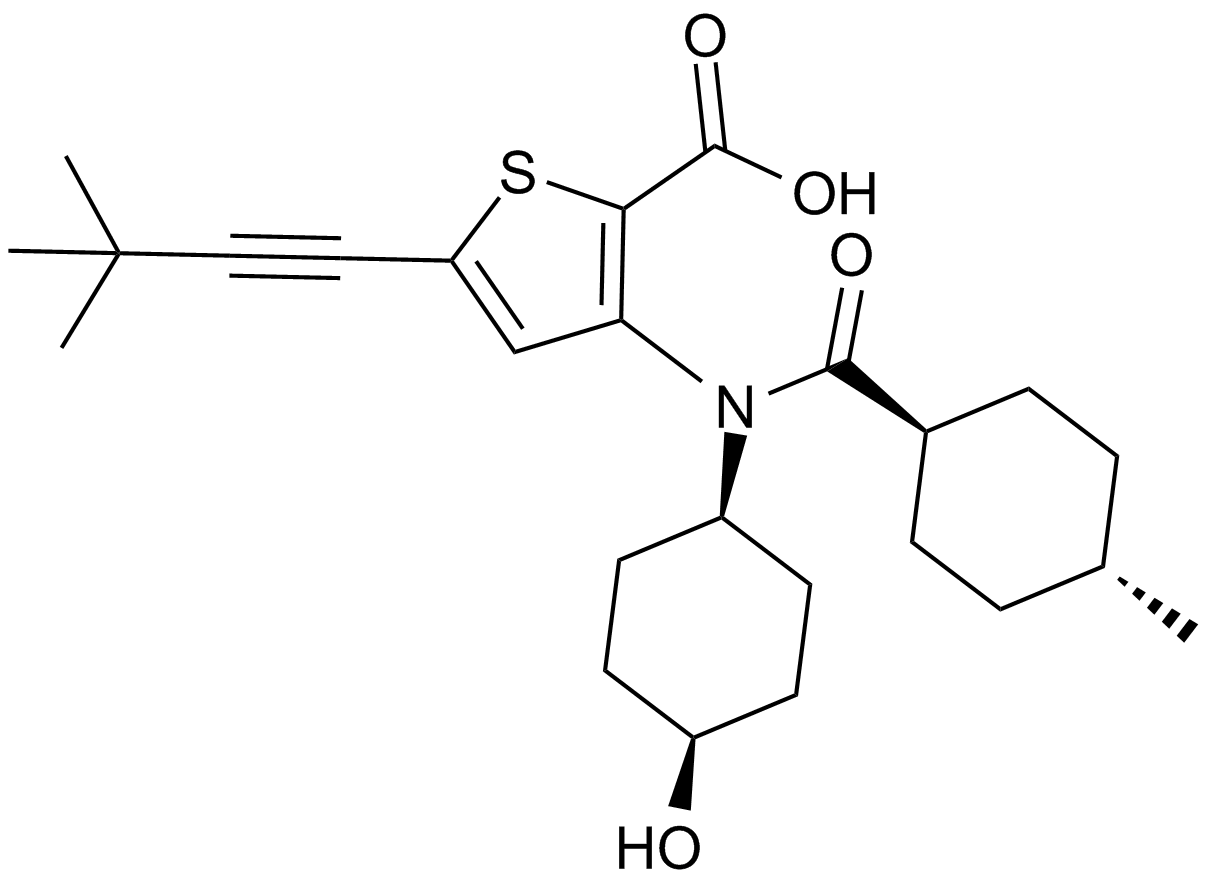
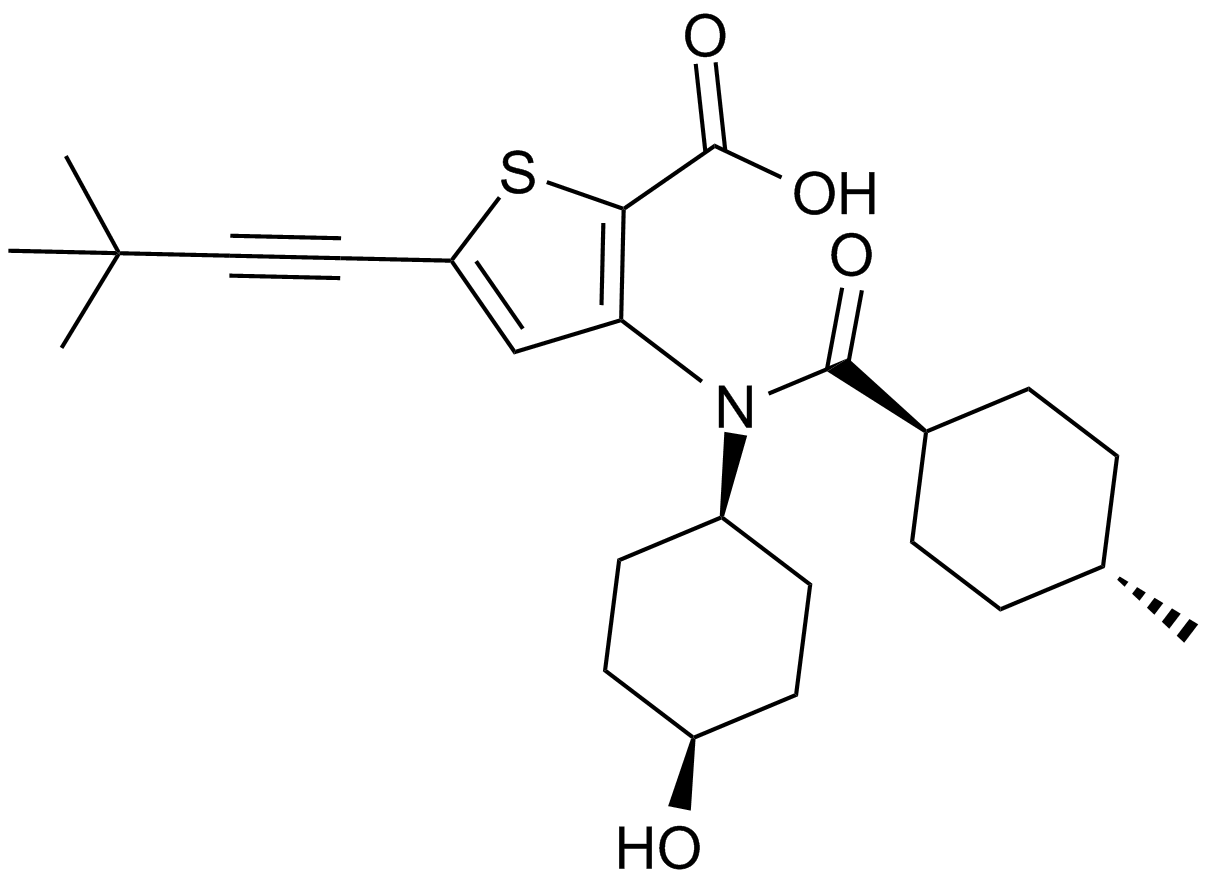
VX-222 (VCH-222, Lomibuvir)
VX-222, a derivative of thiophene-2-carboxylic acid, is a potent non-nucleoside inhibitor (NNI) of the hepatitis C virus (HCV) RNA polymerase, an enzyme regulating RNA synthesis by a de novo-initiated mechanism or by extension from a primed template, that binds to the thumb II allosteric pocket of HCV RNA-dependent RNA polymerase with hydrophobical interactions between the 4-methycyclohexanoyl group of VX-222 and L412, M423 and I482 of thumb II domain. VX-222 exhibits preferential inhibition against primer-dependent RNA synthesis rather than de novo-initiated RNA synthesis with 50% inhibition concentration IC50 values ranging from 0.011 to >5 μM in five different templates of RNA synthesis.
Reference
Yi G, Deval J, Fan B, Cai H, Soulard C, Ranjith-Kumar CT, Smith DB, Blatt L, Beigelman L, Kao CC. Biochemical study of the comparative inhibition of hepatitis C virus RNA polymerase by VX-222 and filibuvir. Antimicrob Agents Chemother. 2012 Feb;56(2):830-7. doi: 10.1128/AAC.05438-11. Epub 2011 Dec 5.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 445.6 |
| Cas No. | 1026785-59-0 |
| Formula | C25H35NO4S |
| Synonyms | Lomibuvir, VCH222,VCH-222, VX222 |
| Solubility | ≥44.6 mg/mL in DMSO; insoluble in H2O; ≥97.2 mg/mL in EtOH |
| Chemical Name | 5-(3,3-dimethylbut-1-ynyl)-3-[(4-hydroxycyclohexyl)-(4-methylcyclohexanecarbonyl)amino]thiophene-2-carboxylic acid |
| Canonical SMILES | CC(CC1)CCC1C(N(C(CC1)CCC1O)c1c(C(O)=O)[s]c(C#CC(C)(C)C)c1)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Kinase experiment [1]: | |
|
Inhibitory activities |
Huh7.5 cells harboring replicons were trypsinized and plated into 48-well plates at 40,000 cells/well. The next day the medium was changed and VX-222 was added to the cells at seven different concentrations, each pair of which differed by 3- or 10-fold dilutions in 200 μl complete medium with triplicates. After 48 h, total RNA was extracted from replicon cells using the TRIzol reagent, and viral RNAs were quantified by realtime reverse transcription-PCR (RT-PCR). First-strand cDNA synthesis used 1g of total RNA along with Moloney murine leukemia virus and 4 M randomized 9-nucleotide (nt) primer mix. RT-PCR used the Bio-Rad IQ SYBR green kit, and primers were HCV 5’-UTRsense (5’-AGC CAT GGC GTT AGT ATG AGT GTC-3’) and 5’-UTRanti (5’-ACA AGG CCT TTC GCG ACC CAA C-3’). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was detected using the sense and antisense oligonucleotides 5’-GAGTCAACGGATTTG GTC GT-3’ and 5’-TGG GAT TTC CAT TGA TGA CA-3’, respectively. All reaction mixtures were heated to 95℃ for 10 min, followed by 40 cycles of PCR of 15 s at 95℃, 20 s at 55℃, and 30 s at 72℃. The fold change and percent change of each group were compared to values for controls. The effective VX-222 concentration that reduced HCV RNA replicon level by 50% (EC50) was calculated with GraphPad Prism software by nonlinear regression analysis with log curve fitting. |
| Cell experiment [2]: | |
|
Cell lines |
HCV genotype 1b (HCV-1b) mADE replicon cells. |
|
Preparation method |
Soluble in DMSO > 10 mM. General tips for obtaining a higher concentration: Please warm the tube at 37℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reaction Conditions |
1 μM; 6 days. |
|
Applications |
In HCV replicon cells, VX-222 dose-dependently increases IFN-β promoter activity by 5.0-fold and inhibits HCV activity with EC50 and EC90 values of 0.3 and 12 nM, respectively. Also, VX-222 rescues the Sendai virus-activated Rig-I pathway due to the inhibition of viral replication. |
| Human experiment [3]: | |
|
Patients |
Patients with genotype 1 hepatitis C virus infection. |
|
Dosage form |
100 or 400 mg twice daily; VX-222+telaprevir ('DUAL' regimen), with ribavirin ('TRIPLE' regimen), or with peginterferon+ribavirin ('QUAD' regimen); 12 weeks. |
|
Applications |
VX-222 (100 or 400 mg twice daily) is well tolerated. Patients exhibit sustained virologic response by 67%, 79% and 90% for TRIPLE (VX-222 400 mg twice daily) and QUAD (VX-222 100 and 400 mg twice daily), respectively. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Yi G, Deval J, Fan B, et al. Biochemical study of the comparative inhibition of hepatitis C virus RNA polymerase by VX-222 and filibuvir. Antimicrob Agents Chemother, 2012, 56(2): 830-837. [2]. Kalkeri G, Lin C, Gopilan J, et al. Restoration of the activated Rig-I pathway in hepatitis C virus (HCV) replicon cells by HCV protease, polymerase, and NS5A inhibitors in vitro at clinically relevant concentrations. Antimicrob Agents Chemother, 2013, 57(9): 4417-4426. [3]. Di Bisceglie AM, Sulkowski M, Gane E, et al. VX-222, a non-nucleoside NS5B polymerase inhibitor, in telaprevir-based regimens for genotype 1 hepatitis C virus infection. Eur J Gastroenterol Hepatol, 2014, 26(7): 761-773. |
|
| Description | VX-222 is a potent and selective inhibitor of HCV RNA-dependent RNA polymerase with IC50 value of 0.94-1.2 μM. | |||||
| Targets | HCV RNA-dependent RNA polymerase | |||||
| IC50 | 0.94-1.2 μM | |||||
Quality Control & MSDS
- View current batch:
Chemical structure