Search results for: 'signaling pathways metabolism hsp'
-
 L1026 DiscoveryProbe™ Neuronal Signaling Library1 CitationSummary: A unique collection of 556 neuronal signaling-related small molecules for neuroscience reasearch.
L1026 DiscoveryProbe™ Neuronal Signaling Library1 CitationSummary: A unique collection of 556 neuronal signaling-related small molecules for neuroscience reasearch. -
 L1044 DiscoveryProbe™ NF-κB Signaling LibrarySummary: A unique collection of 73 NF-κB inhibitors for NF-κB signaling pathway research.
L1044 DiscoveryProbe™ NF-κB Signaling LibrarySummary: A unique collection of 73 NF-κB inhibitors for NF-κB signaling pathway research. -
 M1078 beta-Alanine metabolismSummary: An intermediate metabolite in the pyrimidine metabolism pathway, involved in β-alanine production.
M1078 beta-Alanine metabolismSummary: An intermediate metabolite in the pyrimidine metabolism pathway, involved in β-alanine production. -
 L1044P DiscoveryProbe™ NF-κB Signaling Compound Library PlusSummary: A unique collection of 178 NF-κB inhibitors for NF-κB signaling pathway research.
L1044P DiscoveryProbe™ NF-κB Signaling Compound Library PlusSummary: A unique collection of 178 NF-κB inhibitors for NF-κB signaling pathway research. -
 L1026P DiscoveryProbe™ Neuronal Signaling Compound Library PlusSummary: A unique collection of 948 neuronal signaling-related small molecules for neuroscience reasearch.
L1026P DiscoveryProbe™ Neuronal Signaling Compound Library PlusSummary: A unique collection of 948 neuronal signaling-related small molecules for neuroscience reasearch. -
 M1074 Cysteine and methionine metabolismSummary: A biomarker that can be used to indicate oxidative stress in vivo.
M1074 Cysteine and methionine metabolismSummary: A biomarker that can be used to indicate oxidative stress in vivo. -
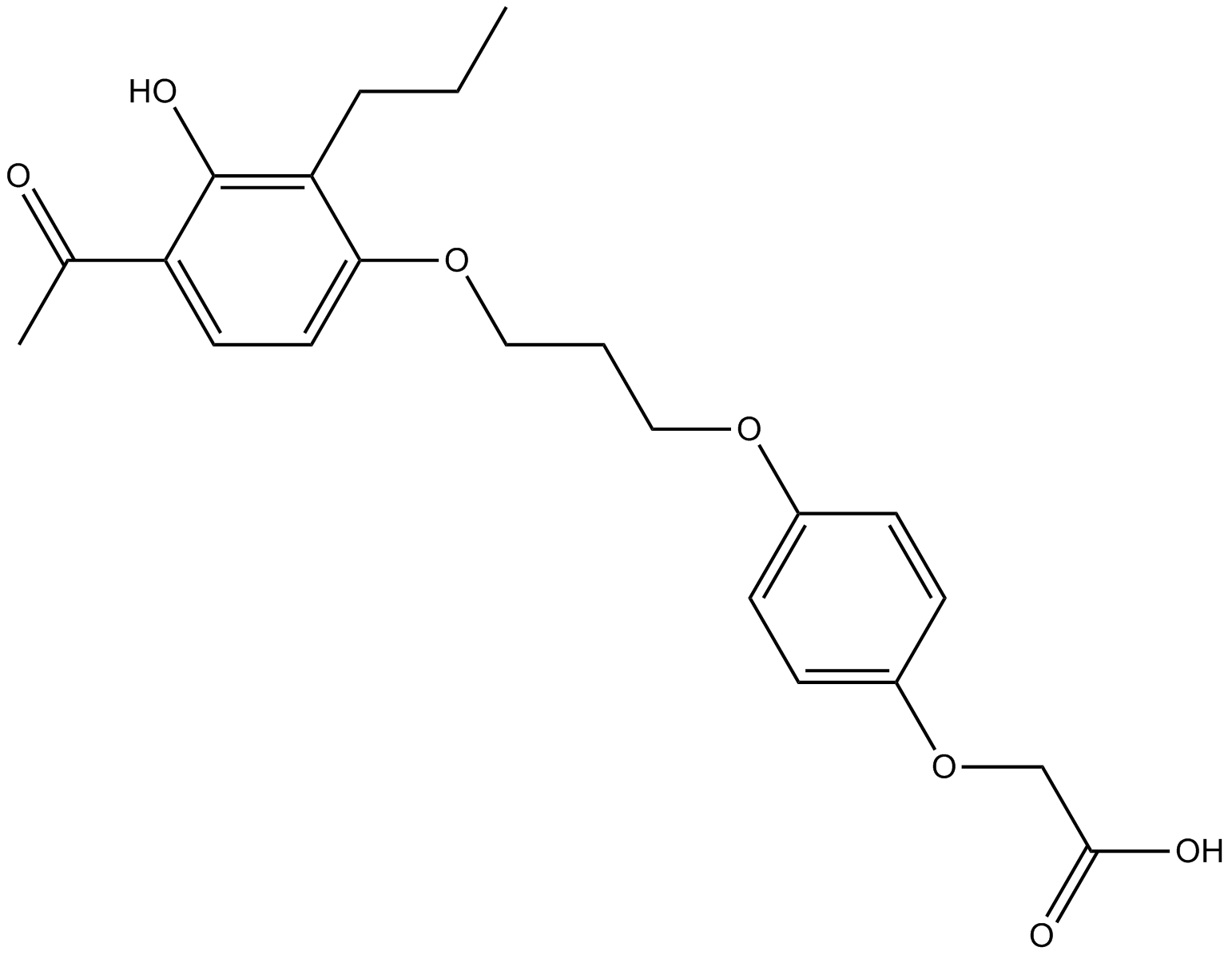 A3536 L-165041Target: PPARSummary: PPARβ/δ agonist,cell permeable,potent and selective
A3536 L-165041Target: PPARSummary: PPARβ/δ agonist,cell permeable,potent and selective -
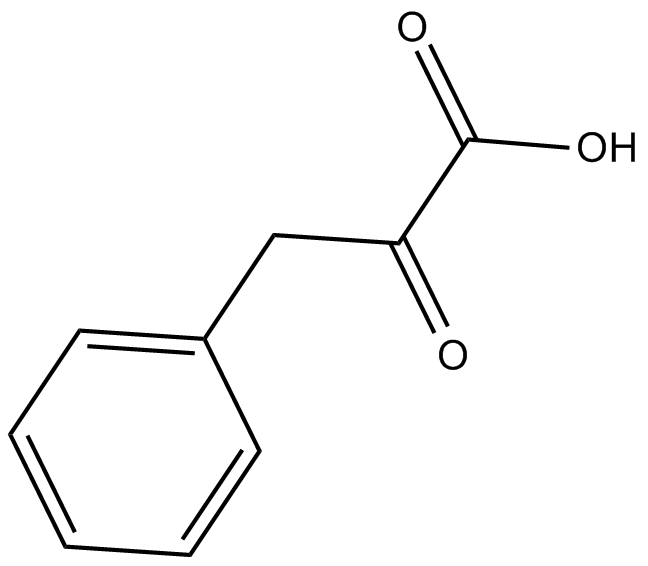 M1118 Phenylalanine & tyrosine metabolismSummary: A metabolic intermediate that can be synthesized into 3-phenyllactic acid via lactate dehydrogenase.
M1118 Phenylalanine & tyrosine metabolismSummary: A metabolic intermediate that can be synthesized into 3-phenyllactic acid via lactate dehydrogenase. -
 M1091 Glycine, serine and threonine metabolismSummary: A short-chain keto acid derivative produced by enzymatic cleavage of cystathionine.
M1091 Glycine, serine and threonine metabolismSummary: A short-chain keto acid derivative produced by enzymatic cleavage of cystathionine. -
 BA6713 KL-11743Summary: KL-11743 is a potent, orally active inhibitor of glucose-competitive class I glucose transporter proteins.
BA6713 KL-11743Summary: KL-11743 is a potent, orally active inhibitor of glucose-competitive class I glucose transporter proteins.


