Search results for: 'signaling pathways apoptosis bax'
-
 A4459 Bax channel blockerSummary: Inhibitor of Bax-mediated mitochondrial cytochrome c release
A4459 Bax channel blockerSummary: Inhibitor of Bax-mediated mitochondrial cytochrome c release -
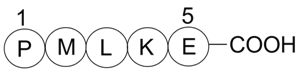 A4460 Bax inhibitor peptide P5Summary: Bax inhibitor
A4460 Bax inhibitor peptide P5Summary: Bax inhibitor -
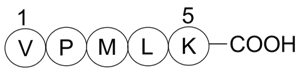 A4461 Bax inhibitor peptide V5Target: BaxSummary: Bax inhibitor
A4461 Bax inhibitor peptide V5Target: BaxSummary: Bax inhibitor -
 A4016 Apoptosis Activator 2Summary: Indoledione caspase activator, cell-permeable
A4016 Apoptosis Activator 2Summary: Indoledione caspase activator, cell-permeable -
 K2295 Annexin V-APC/PI Apoptosis KitSummary: Annexin V-APC/PI apoptosis detection kit, which can detect apoptosis& necrosis within 15-30 minutes
K2295 Annexin V-APC/PI Apoptosis KitSummary: Annexin V-APC/PI apoptosis detection kit, which can detect apoptosis& necrosis within 15-30 minutes -
 K2718 Apoptosis Inducer Set (Ready-to-use)Summary: A set of apoptosis inducers
K2718 Apoptosis Inducer Set (Ready-to-use)Summary: A set of apoptosis inducers -
 K2711 Apoptosis Inducer KitSummary: A ready-to-use apoptosis inducer reagent, which consists of recombinant human TNF-α and SM-164
K2711 Apoptosis Inducer KitSummary: A ready-to-use apoptosis inducer reagent, which consists of recombinant human TNF-α and SM-164 -
 K2255 Annexin V-Cy5/DAPI Apoptosis Kit1 CitationSummary: Annexin V-Cy5/DAPI apoptosis detection kit, which can detect apoptosis& necrosis within 10-20 minutes
K2255 Annexin V-Cy5/DAPI Apoptosis Kit1 CitationSummary: Annexin V-Cy5/DAPI apoptosis detection kit, which can detect apoptosis& necrosis within 10-20 minutes -
 K2294 Annexin V-APC/DAPI Apoptosis KitSummary: Annexin V-APC/DAPI apoptosis detection kit, which can detect apoptosis& necrosis within 15-30 minutes
K2294 Annexin V-APC/DAPI Apoptosis KitSummary: Annexin V-APC/DAPI apoptosis detection kit, which can detect apoptosis& necrosis within 15-30 minutes -
 K2297 Annexin V-APC/7-AAD Apoptosis Kit1 CitationSummary: Annexin V-APC/7-AAD apoptosis detection kit, which can detect apoptosis& necrosis within 15-30 minutes
K2297 Annexin V-APC/7-AAD Apoptosis Kit1 CitationSummary: Annexin V-APC/7-AAD apoptosis detection kit, which can detect apoptosis& necrosis within 15-30 minutes


