Search results for: 'signaling pathways ubiquitination proteasome proteasome'
-
 K2096 Proteasome Activity Fluorometric Assay KitSummary: Specifically detects proteasome activity
K2096 Proteasome Activity Fluorometric Assay KitSummary: Specifically detects proteasome activity -
 K2242 Fluorometric Proteasome 20S Activity Assay KitSummary: A kit for the detection of proteasome 20S activity by the fluorescent substrate LLVY-R110
K2242 Fluorometric Proteasome 20S Activity Assay KitSummary: A kit for the detection of proteasome 20S activity by the fluorescent substrate LLVY-R110 -
 L1049 DiscoveryProbe™ Ubiquitination Compound LibrarySummary: A collection of 144 ubiquitination compounds supplied as lyophilized powder or pre-dissolved DMSO solutions
L1049 DiscoveryProbe™ Ubiquitination Compound LibrarySummary: A collection of 144 ubiquitination compounds supplied as lyophilized powder or pre-dissolved DMSO solutions -
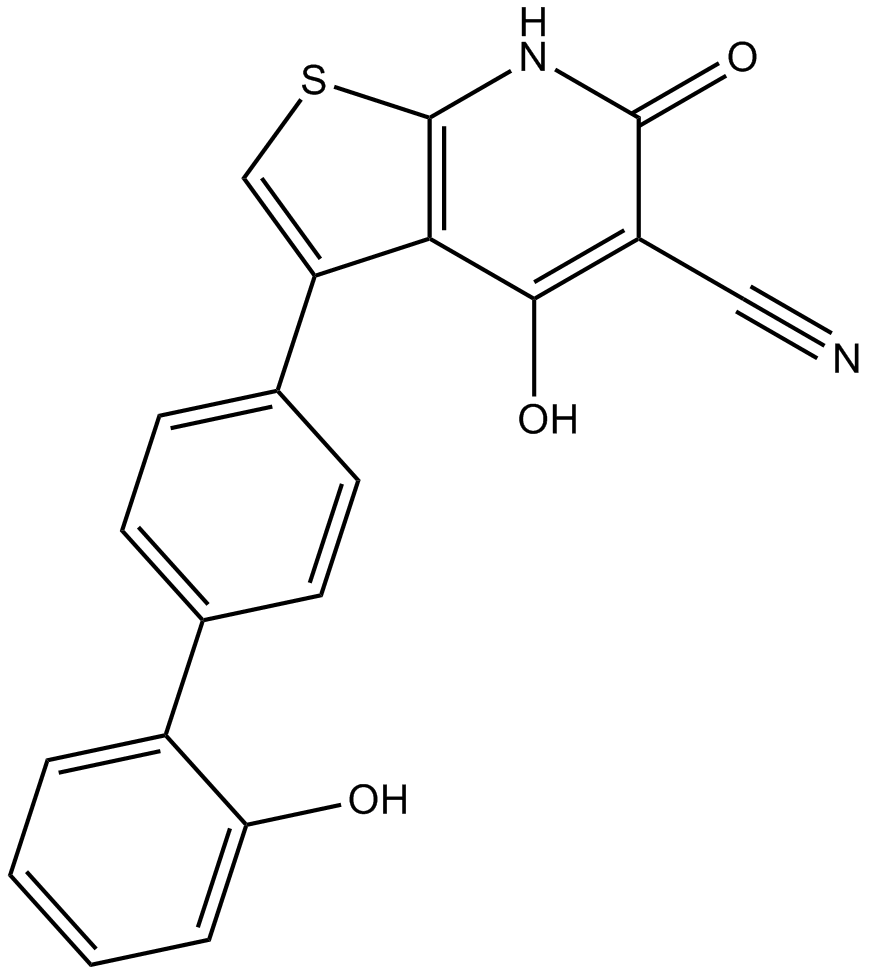 A3963 A-7696624 CitationSummary: AMPK activator,potent and reversible
A3963 A-7696624 CitationSummary: AMPK activator,potent and reversible -
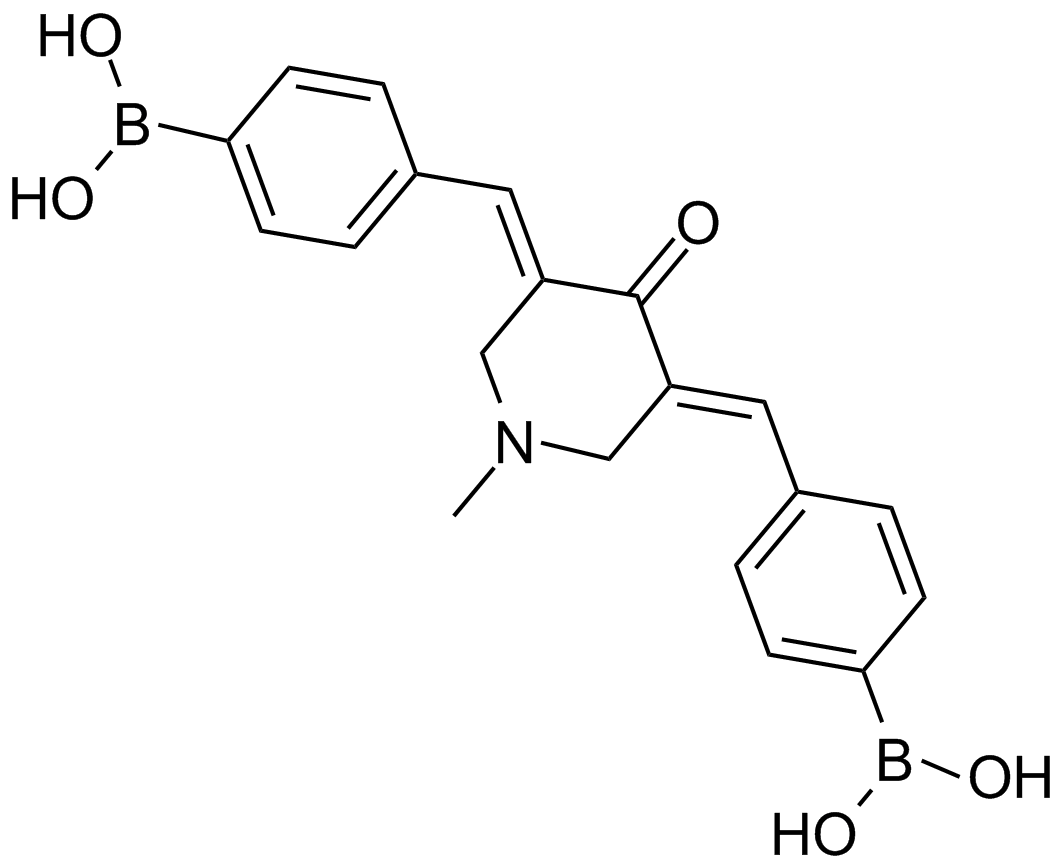 A8163 AM 1142 CitationTarget: ProteasomeSummary: 20S proteasome inhibitor
A8163 AM 1142 CitationTarget: ProteasomeSummary: 20S proteasome inhibitor -
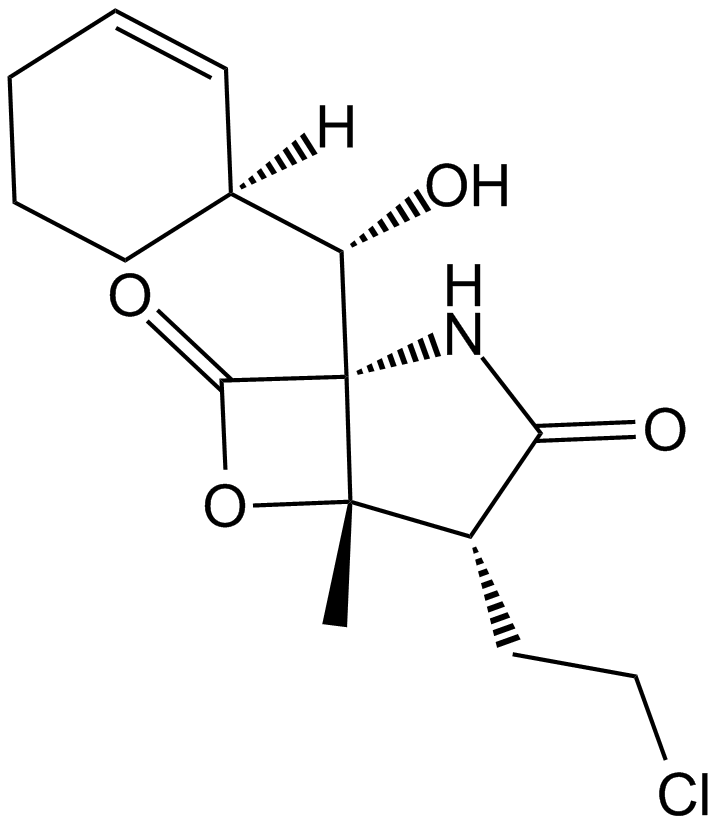 A4010 Salinosporamide A (NPI-0052, Marizomib)Target: ProteasomeSummary: 20S proteasome inhibitor
A4010 Salinosporamide A (NPI-0052, Marizomib)Target: ProteasomeSummary: 20S proteasome inhibitor -
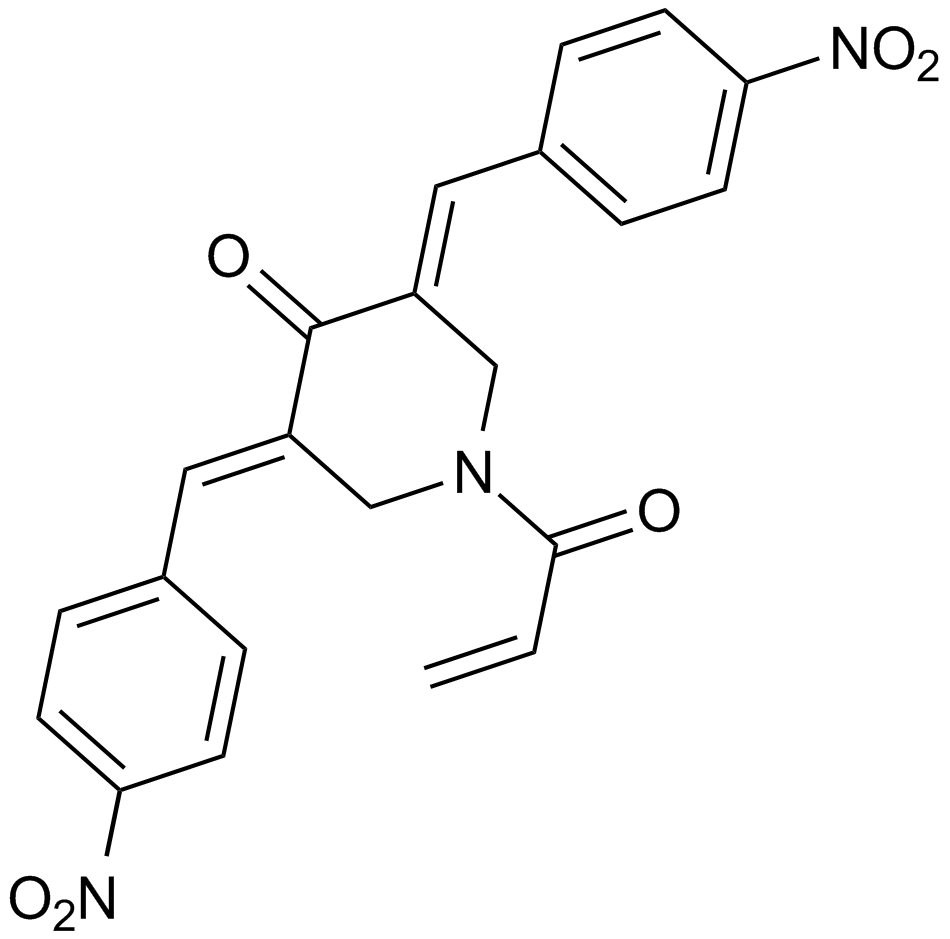 A4453 NSC 687852 (b-AP15)Target: Deubiquitinating enzymes (DUBs)Summary: 19S regulatory particle Inhibitor
A4453 NSC 687852 (b-AP15)Target: Deubiquitinating enzymes (DUBs)Summary: 19S regulatory particle Inhibitor -
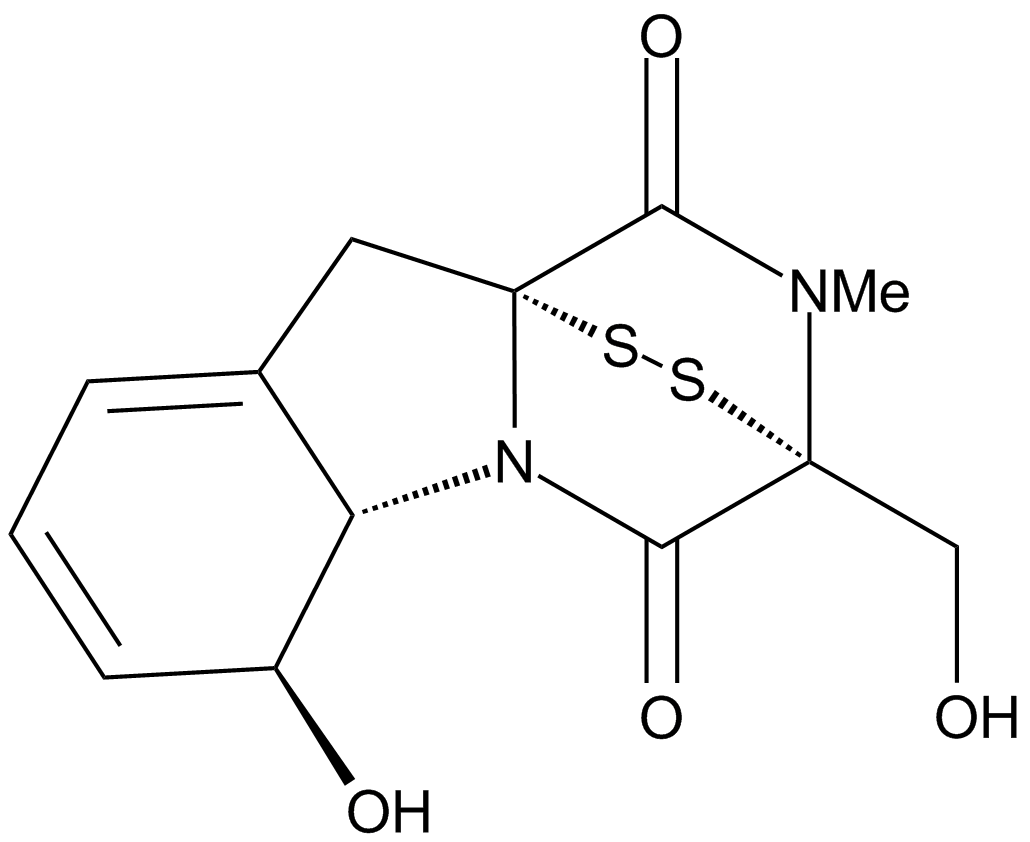 A4443 Gliotoxin1 CitationTarget: 20S proteasomal chymotrypsin|Geranylgeranyltransferase I|FarnesyltransferaseSummary: 20S proteasome inhibitor
A4443 Gliotoxin1 CitationTarget: 20S proteasomal chymotrypsin|Geranylgeranyltransferase I|FarnesyltransferaseSummary: 20S proteasome inhibitor -
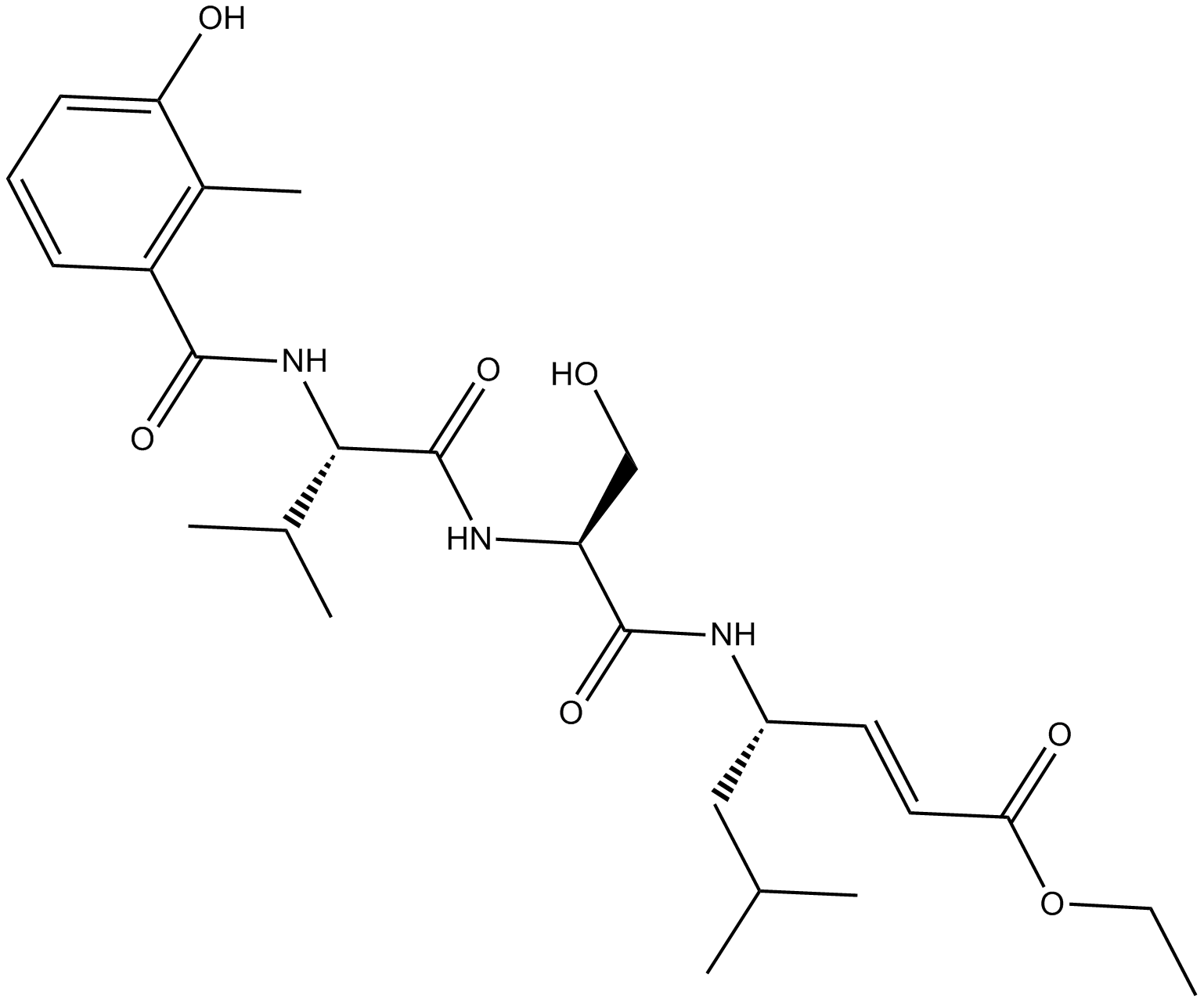 C4090 HMB-Val-Ser-Leu-VESummary: inhibitor of the trypsin-like activity of the 20S proteasome
C4090 HMB-Val-Ser-Leu-VESummary: inhibitor of the trypsin-like activity of the 20S proteasome -
 BA8291 Ac-Nle-Pro-Nle-Asp-AMCSummary: Ac-Nle-Pro-Nle-Asp-AMC is a specific substrate for the 26S proteasome.
BA8291 Ac-Nle-Pro-Nle-Asp-AMCSummary: Ac-Nle-Pro-Nle-Asp-AMC is a specific substrate for the 26S proteasome.


