Search results for: 'signaling pathways tyrosine kinase'
-
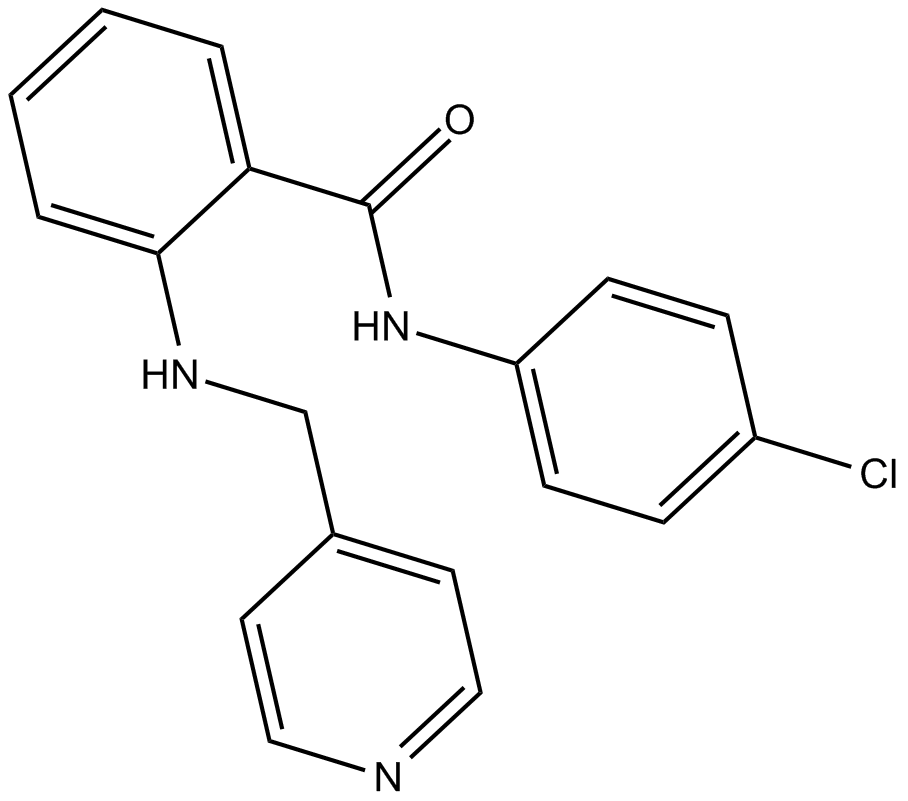 C4603 VEGFR Tyrosine Kinase Inhibitor II1 CitationSummary: VEGFR inhibitor
C4603 VEGFR Tyrosine Kinase Inhibitor II1 CitationSummary: VEGFR inhibitor -
 L1028 DiscoveryProbe™ Tyrosine Kinase Inhibitor LibrarySummary: A unique collection of 369 tyrosine kinase inhibitors for high throughput screening (HTS) and high content screening (HCS).
L1028 DiscoveryProbe™ Tyrosine Kinase Inhibitor LibrarySummary: A unique collection of 369 tyrosine kinase inhibitors for high throughput screening (HTS) and high content screening (HCS). -
 P1330 Recombinant Rhesus Macaque Fms-related Tyrosine Kinase 3 LigandSummary: Recombinant Rhesus Macaque FLT3L (E.coli, Tag Free, Lyophilized)
P1330 Recombinant Rhesus Macaque Fms-related Tyrosine Kinase 3 LigandSummary: Recombinant Rhesus Macaque FLT3L (E.coli, Tag Free, Lyophilized) -
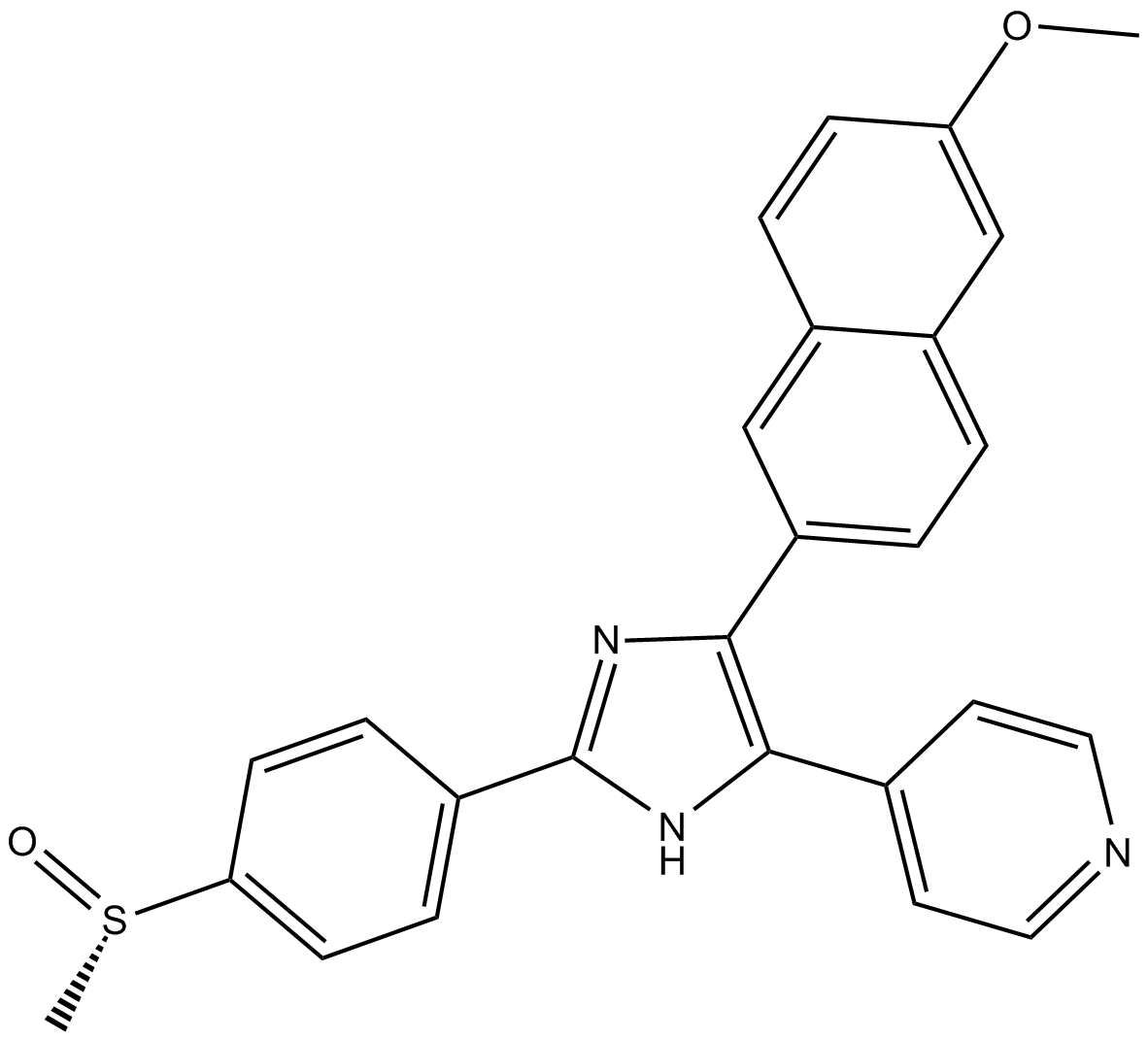 A5979 Tie2 kinase inhibitor1 CitationTarget: Tie-2Summary: Tie-2(Tie2 ) inhibitor
A5979 Tie2 kinase inhibitor1 CitationTarget: Tie-2Summary: Tie-2(Tie2 ) inhibitor -
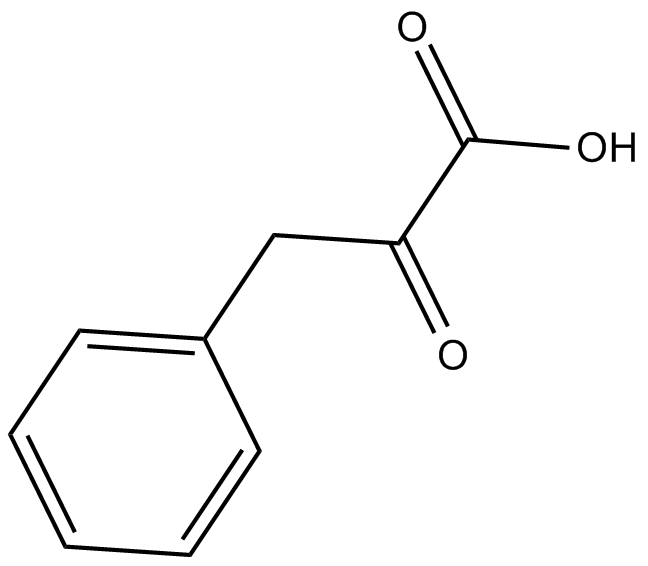 M1118 Phenylalanine & tyrosine metabolismSummary: A metabolic intermediate that can be synthesized into 3-phenyllactic acid via lactate dehydrogenase.
M1118 Phenylalanine & tyrosine metabolismSummary: A metabolic intermediate that can be synthesized into 3-phenyllactic acid via lactate dehydrogenase. -
 BA8810 HG-7-86-01Summary: HG-7-86-01 is a type II tyrosine kinase inhibitor.
BA8810 HG-7-86-01Summary: HG-7-86-01 is a type II tyrosine kinase inhibitor. -
 BA8889 BAY-474Summary: BAY-474 is a tyrosine protein kinase inhibitor.
BA8889 BAY-474Summary: BAY-474 is a tyrosine protein kinase inhibitor. -
 BA9011 eCF506Summary: eCF506 is a potent, orally active inhibitor of non-receptor tyrosine kinases.
BA9011 eCF506Summary: eCF506 is a potent, orally active inhibitor of non-receptor tyrosine kinases. -
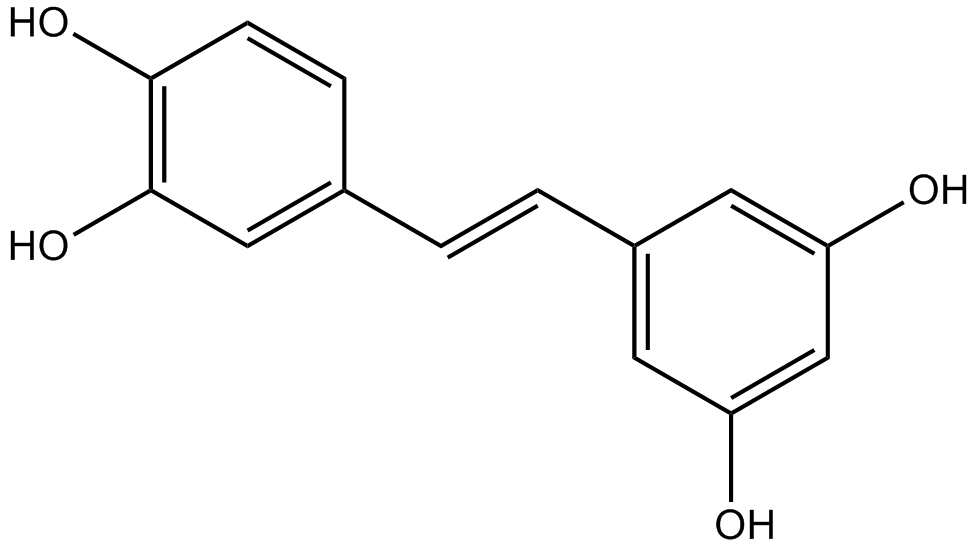 N2031 PiceatannolSummary: p56lck/Syk inhibitor
N2031 PiceatannolSummary: p56lck/Syk inhibitor -
 C5282 Adenosine Kinase Inhibitor (hydrate)Summary: inhibitor of adenosine kinase
C5282 Adenosine Kinase Inhibitor (hydrate)Summary: inhibitor of adenosine kinase


