Search results for: 'signaling pathways proteases aminopeptidase'
-
 MA3695 Anti-Methionine Aminopeptidase 2 Rabbit Monoclonal AntibodySummary: Anti-Methionine Aminopeptidase 2 Rabbit Monoclonal Antibody
MA3695 Anti-Methionine Aminopeptidase 2 Rabbit Monoclonal AntibodySummary: Anti-Methionine Aminopeptidase 2 Rabbit Monoclonal Antibody -
 L1026 DiscoveryProbe™ Neuronal Signaling Library1 CitationSummary: A unique collection of 556 neuronal signaling-related small molecules for neuroscience reasearch.
L1026 DiscoveryProbe™ Neuronal Signaling Library1 CitationSummary: A unique collection of 556 neuronal signaling-related small molecules for neuroscience reasearch. -
 L1044 DiscoveryProbe™ NF-κB Signaling LibrarySummary: A unique collection of 73 NF-κB inhibitors for NF-κB signaling pathway research.
L1044 DiscoveryProbe™ NF-κB Signaling LibrarySummary: A unique collection of 73 NF-κB inhibitors for NF-κB signaling pathway research. -
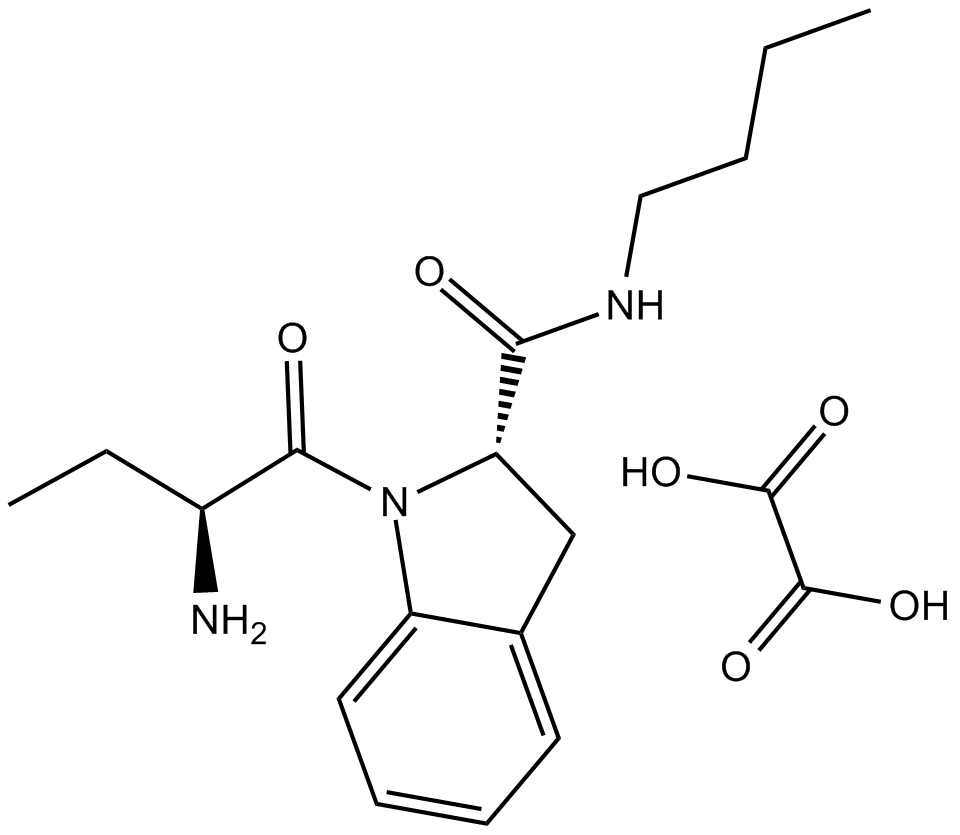 B6689 Butabindide oxalateSummary: CCK-inactivating serine protease (tripeptidyl peptidase II) inhibitor
B6689 Butabindide oxalateSummary: CCK-inactivating serine protease (tripeptidyl peptidase II) inhibitor -
 BA7971 M8891Summary: M8891 is an orally active, reversible and blood-brain barrier-crossing inhibitor of methionine aminopeptidase 2.
BA7971 M8891Summary: M8891 is an orally active, reversible and blood-brain barrier-crossing inhibitor of methionine aminopeptidase 2. -
 P1578 Recombinant Aeromonas AminopeptidaseSummary: Recombinant Aeromonas Aminopeptidase (E.coli, Tag Free, Lyophilized)
P1578 Recombinant Aeromonas AminopeptidaseSummary: Recombinant Aeromonas Aminopeptidase (E.coli, Tag Free, Lyophilized) -
 L1044P DiscoveryProbe™ NF-κB Signaling Compound Library PlusSummary: A unique collection of 178 NF-κB inhibitors for NF-κB signaling pathway research.
L1044P DiscoveryProbe™ NF-κB Signaling Compound Library PlusSummary: A unique collection of 178 NF-κB inhibitors for NF-κB signaling pathway research. -
 L1026P DiscoveryProbe™ Neuronal Signaling Compound Library PlusSummary: A unique collection of 948 neuronal signaling-related small molecules for neuroscience reasearch.
L1026P DiscoveryProbe™ Neuronal Signaling Compound Library PlusSummary: A unique collection of 948 neuronal signaling-related small molecules for neuroscience reasearch. -
 BA6270 STING-IN-6Summary: STING-IN-6 is a potent inhibitor.
BA6270 STING-IN-6Summary: STING-IN-6 is a potent inhibitor. -
 BA9168 TGFβ-IN-5Summary: TGFβ-IN-5 is a TGFβ inhibitor.
BA9168 TGFβ-IN-5Summary: TGFβ-IN-5 is a TGFβ inhibitor.


