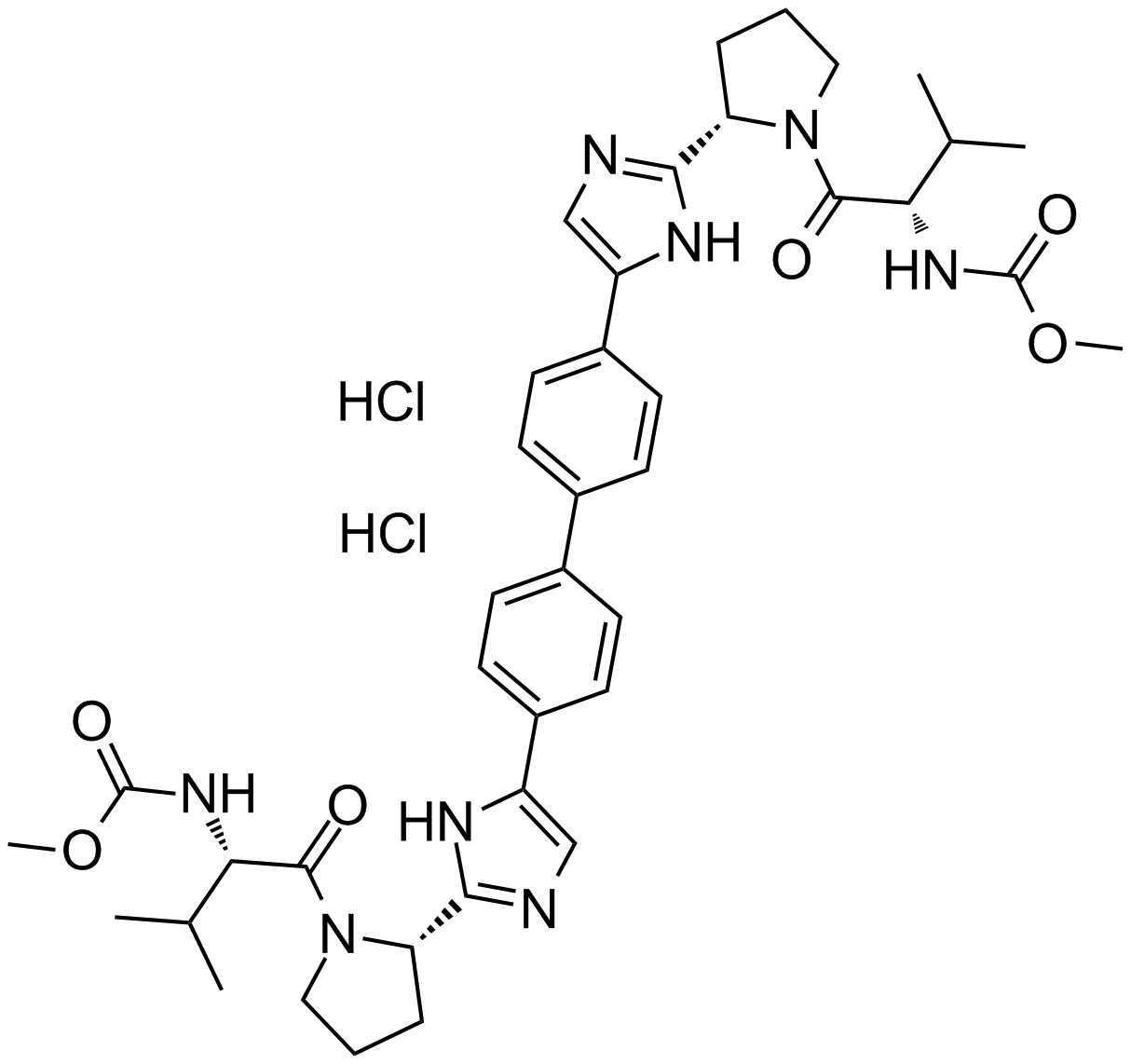
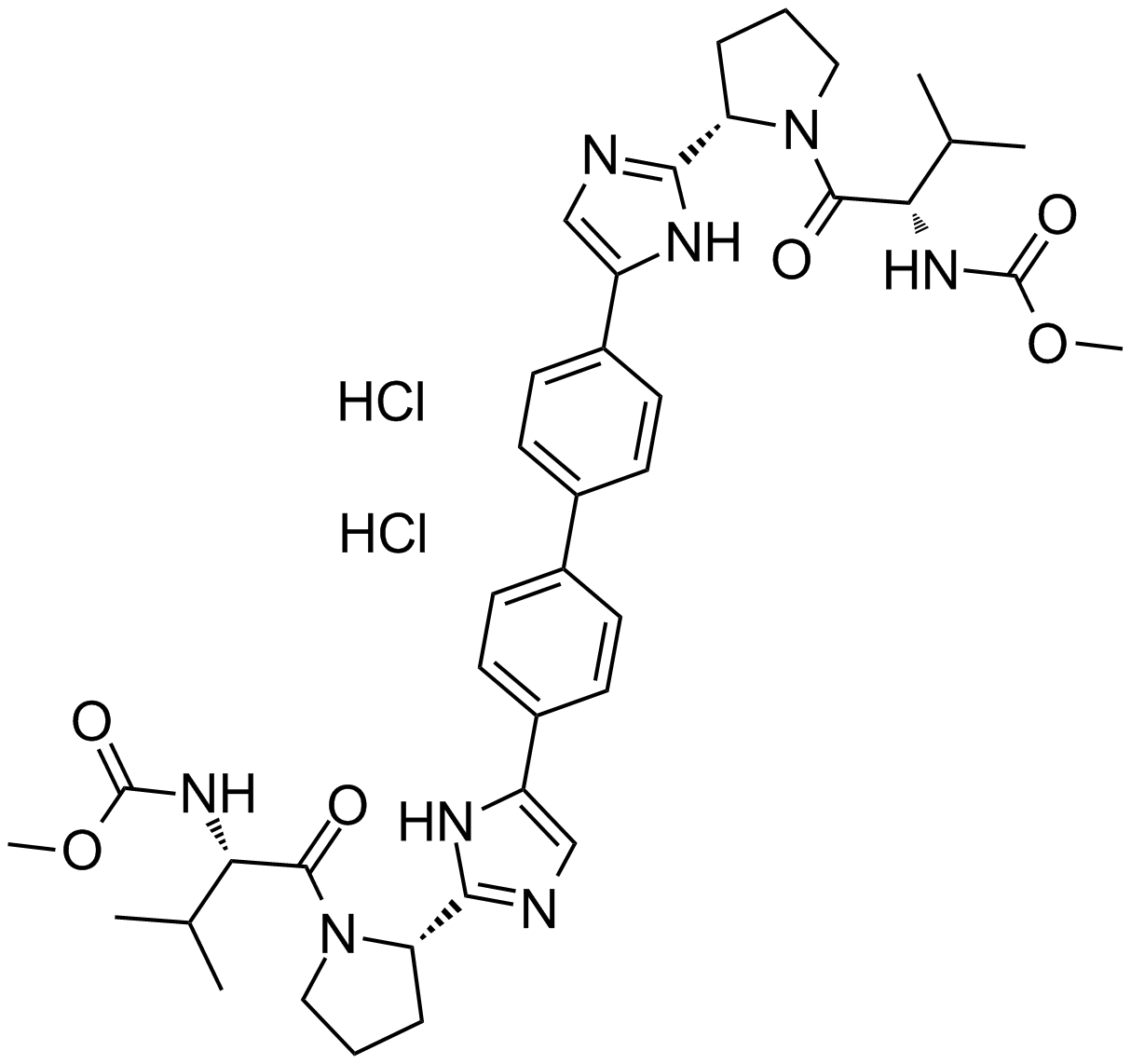
BMS-790052 dihydrochloride
BMS-790052 is a potent inhibitor of HCV NS5A with EC50 value of 9-50 pM [1].
NS5A (nonstructural protein 5A) is a zinc-binding protein and plays a pivotal role in HCV RNA replication. NS5A involves in mediating the host cell function and HCV infection, therefore, it is regarded as a promising target for HCV treatment [2].
BMS-790052 is a selective HCV NS5A inhibitor and has the most potent ability to inhibit HCV replication reported so far. When using FRET assays to determine the efficiay of BMS-790052 on HCV NS5A, replicon cells were cultured in 96-well plates and after 12 hours BMS-790052 was added to test the replication activity and cytotoxicity, the results showed that the mean values of BMS-790052 for genotype 1a and 1b were 50 and 9 pM, respectively [1]. In stable replication cell lines with GT-5a hybrid replicons (GT-5a-6, GT-5a-7, and GT-5a-9), BMS-790052 treatment showed effective antiviral activity with EC50 values range from 3 to 7 pM [3].
It has been shown that oral administration of BMS-790052 was safe and well tolerated up to 200 mg when tested with healthy people, and it was safe and well tolerated at the dose of 100 mg when tested with HCV-infected patients [1].
References:
[1]. Gao, M., et al., Chemical genetics strategy identifies an HCV NS5A inhibitor with a potent clinical effect. Nature, 2010. 465(7294): p. 96-100.
[2]. Eyre, N.S. and M.R. Beard, HCV NS5A inhibitors disrupt replication factory formation: a novel mechanism of antiviral action. Gastroenterology, 2014. 147(5): p. 959-62.
[3]. Wang, C., et al., Comparison of daclatasvir resistance barriers on NS5A from hepatitis C virus genotypes 1 to 6: implications for cross-genotype activity. Antimicrob Agents Chemother, 2014. 58(9): p. 5155-63.
| Storage | Store at -20°C |
| M.Wt | 811.8 |
| Cas No. | 1009119-65-6 |
| Formula | C40H52Cl2N8O6 |
| Synonyms | Daclatasvir dihydrochloride;BMS790052 dihydrochloride;BMS 790052 dihydrochloride |
| Solubility | Soluble in DMSO |
| Chemical Name | dimethyl ((2S,2'S)-((2S,2'S)-2,2'-(5,5'-([1,1'-biphenyl]-4,4'-diyl)bis(1H-imidazole-5,2-diyl))bis(pyrrolidine-2,1-diyl))bis(3-methyl-1-oxobutane-2,1-diyl))dicarbamate dihydrochloride |
| Canonical SMILES | CC(C)[C@H](NC(OC)=O)C(N1[C@H](C2=NC=C(C3=CC=C(C4=CC=C(C5=CN=C([C@@H]6CCCN6C([C@@H](NC(OC)=O)C(C)C)=O)N5)C=C4)C=C3)N2)CCC1)=O.Cl.Cl |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure