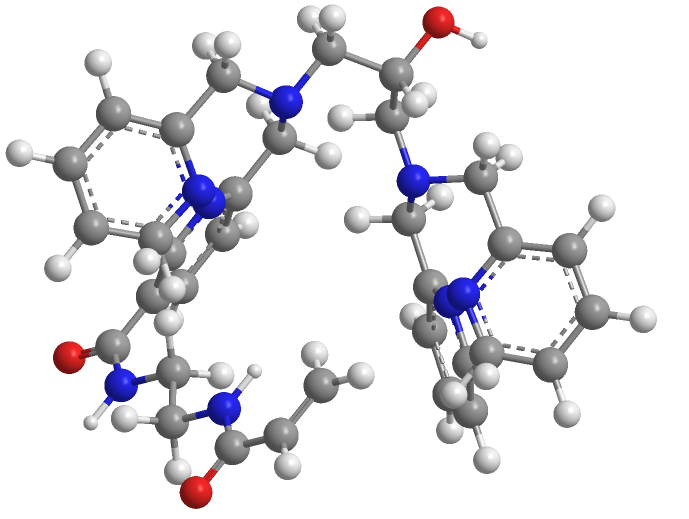
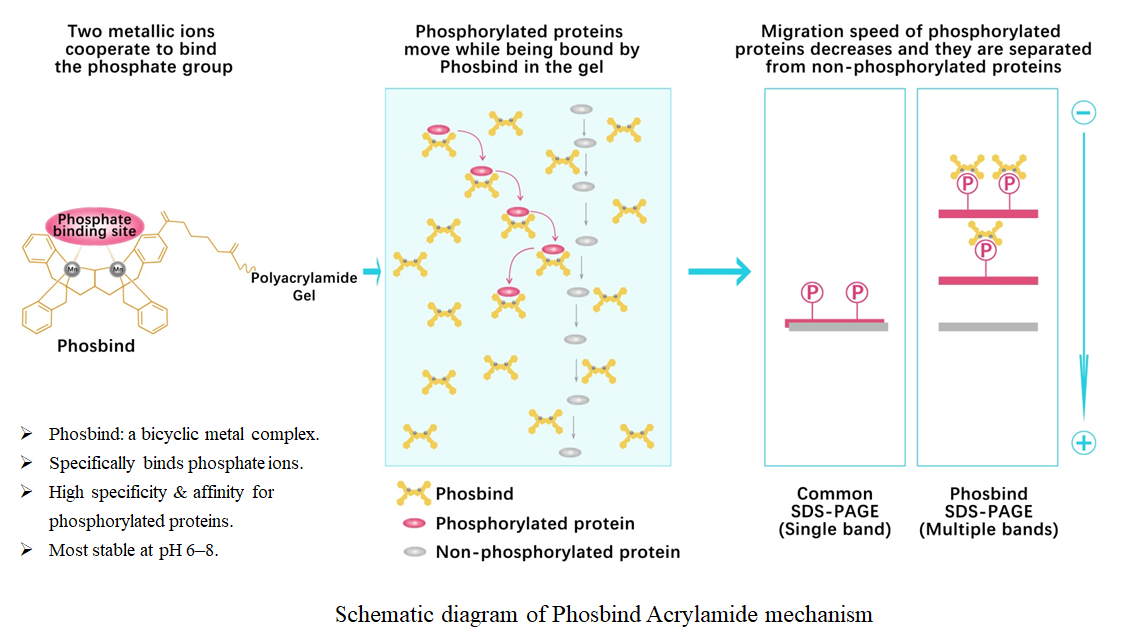
Phos binding reagent (Phosbind) acrylamide
Phosbind Acrylamide is an innovative phosphate-binding reagent. It appears as a yellow viscous oil and is an acrylamide derivative functionalized with a Phosbind ligand. It is also a bicyclic metal complex, specifically designed for the electrophoretic separation and detection of phosphorylated and non-phosphorylated proteins. In the presence of divalent metal ions such as Mn²⁺ or Zn²⁺, the reagent specifically recognizes and binds to phosphate groups, with optimal reaction conditions at neutral physiological pH 6–8.
The mechanism of Phosbind Acrylamide relies on its specific interaction with phosphate groups. After the reagent is added to a conventional SDS-PAGE separating gel together with divalent metal ions such as Mn²⁺ or Zn²⁺, the Phosbind Acrylamide–metal ion complex immobilized in the gel specifically binds to phosphate groups on proteins. This reduces the electrophoretic mobility of phosphorylated proteins, while the migration of non-phosphorylated proteins remains unaffected, thereby achieving separation between the two forms. Moreover, the higher the phosphorylation level of a protein, the slower its migration rate. The typical molar ratio of Phosbind Acrylamide to metal ions is 1:2, although exceptions may occur in practical applications.
This product has a wide range of applications. It can be used for qualitative and quantitative detection of phosphorylation levels in in vitro kinase and phosphatase reaction systems, analysis of endogenous protein phosphorylation, plant protein phosphorylation studies, phosphorylation status identification, and phosphoproteomics research. It also enables fine separation of phosphorylated isoforms in two-dimensional electrophoresis. Additionally, it is suitable for drug screening and target validation, studies of cell signaling pathways (e.g., GPCR, Wnt, MAPK), as well as fundamental research areas including neuroscience, metabolic regulation, apoptosis, and DNA damage repair.
This product is suitable for detecting proteins in the range of 5–350 kDa, with no limitations regarding species (human, animal, plant, microorganism, etc.) or sample type (purified protein, cell lysate, tissue homogenate, in vitro kinase reaction system, etc.). It is compatible with various downstream analysis methods, including CBB staining, Western blotting (WB), two-dimensional electrophoresis, and mass spectrometry (MS). Compared with traditional phosphorylation detection methods, this technology allows direct visualization of phosphorylation-dependent electrophoretic mobility shifts without the need for specific phospho-antibodies. For optimal experimental results, the use of the following recommended accessories is advised: Prestained Protein Marker (F4005), Protease Inhibitor Cocktail (K1007), Phosphatase Inhibitor Cocktail (K1015), and Tris-Gly Fixed-Concentration Gel Preparation Kits (K4131–K4135, 6%/8%/10%/12%/15%).
This product consists of Phosbind Acrylamide and a separately packaged tube of MnCl₂. Using a standard Mini PAGE gel (8.3 cm × 7.3 cm, 1 mm thickness) with a separating gel volume of 5 mL and a final Phosbind Acrylamide concentration of 50 μM as an example, one 5 mg vial of the product can prepare approximately 33 gels. The number of gels that can be prepared varies depending on the concentration of Phosbind Acrylamide used; higher concentrations yield fewer gels.
- 1. Lizhu Ma, Wei Fu, et al. "FGF4 initiates X-chromosome inactivation via activating YY1 and prompting loss of pluripotency factors." EMBO J. 2026 Mar 25. PMID: 41882225
- 2. Ken-ichi Nakajima, Shakur Mohibi, et al. "Nuclear Translocation of Mitochondrial Ferredoxin Reductase Is Regulated by AKT-Mediated Phosphorylation." Biochem J. 2026 May 6;483(5):685-698. PMID: 41860465
- 3. Zemin Ma, Xuanlin Gao, et al. "A leucine‐rich‐repeat receptor‐like kinase SERL1 phosphorylates and stabilizes OsALDH2B1 to promote alkaline tolerance and grain size in rice." J Integr Plant Biol. 2026 Mar 4. PMID: 41782191
- 4. Katie Culhane, Shiv Kumar, et al. "Protocol for purifying recombinant HBV capsids from HepG2 cells and comparative phosphorylation analysis by phosphate-affinity SDS-PAGE." STAR Protoc. 2026 Feb 25;7(1):104403. PMID: 41746804
- 5. Xueying Li, Xiumin Nie, et al. "SmCPK20 phosphorylates SmWRKY2 to regulate jasmonate‐mediated tanshinone synthesis in Salvia miltiorrhiza." J Integr Plant Biol. 2026 Feb 24. PMID: 41736258
- 6. Lina Qiu, Jie Yang, et al. "The MdOST1-MdCNGC1C-MdCaM7. 1 module fine-tunes cold-induced calcium signaling in apple." Plant Cell. 2026 Mar 3;38(3):koag032. PMID: 41715950
- 7. Shuyao Jiang, Zixuan Jia, et al. "HSPA1A and DNAJB1 regulate NELF condensate dynamics to safeguard transcriptional recovery under heat stress." Mol Cell. 2026 Feb 19;86(4):674-692.e10. PMID: 41653920
- 8. Ruilin Li, Sisi Xia, et al. "HCMV encoded UL84 hijacks FHL2 to suppress type I interferon production and enhance viral replication." PLoS Pathog. 2026 Jan 26;22(1):e1013895. PMID: 41587224
- 9. Tingting Pei, Minghui Zhan, et al. "LysM receptor‐like kinase MdLYK3 destabilizes the ABA receptors PYR/PYLs to suppress ABA signaling in apple." New Phytol. 2026 Jan 22. PMID: 41572932
- 10. Damayantee Das, Shawn M. Lamothe, et al. "Regulation of Kv2.1 biogenesis and gating by candidate disease-linked Kv6.1 variants." J Biol Chem. 2026 Jan;302(1):110943. PMID: 41241096
- 11. Yunzi Mao, Mei Cui, et al. "AMPKα2 signals amino acid insufficiency to inhibit protein synthesis." Cell Metab. 2026 Jan 6;38(1):192-207.e8. PMID: 41061696
- 12. Chen‐xi Feng, Mei Wang, et al. "FAM20C Modulates Neuronal Differentiation in Hypoxic–Ischemic Brain Damage via KAP1 Phosphorylation and LINE1 RNA m6A ‐Dependent H3K9me3 Regulation." Cell Prolif. 2026 Jan;59(1):e70073. PMID: 40511628
- 13. Xueying Li, Bolun Li, et al. "Genome-wide identification of the calcium-dependent kinase CPK gene family in Salvia miltiorrhiza reveals that SmCPK24 promotes tanshinone biosynthesis." Industrial Crops and Products Volume 239, January 2026, 122418
- 14. Golnoush Akhtari, Milad Falahat Chian, et al. "Rhythmicity of TOR (Target of Rapamycin) activity supports circadian function in Neurospora crassa." J Biol Rhythms. 2025 Dec 30:7487304251393577. PMID: 41472420
- 15. Zhen Chen, Zhen He, et al. "Histidine kinase Hik33 plays a crucial role in UV-B-induced photosynthetic acclimation in cyanobacteria." Plant Physiol. 2025 Dec 18:kiaf661. PMID: 41411299
- 16. Yi Kan, Xiao-Rui Mu, et al. "A stepwise decoding mechanism for heat sensing in plants connects lipid remodeling to a nuclear signaling cascade." Cell 188, 1–19, December 24, 2025. PMID: 41338194
- 17. Meng Qi, Dong Meng, et al. "Hyperoside, a flavonoid, induces lnc187 and lnc999 to enhance pigeon pea pollen tube growth and seed production." Sci Adv. 2025 Nov 14;11(46):eadz3499. PMID: 41237233
- 18. Hang Yin, Yutong Tian, et al. "Phosphorylation of EF-P aggravates Streptococcus suis-induced blood–brain barrier damage by enhancing serine protease production." Vet Res. 2025 Nov 7;56(1):215. PMID: 41204333
- 19. Yi Lu, Xiaoying Yu, et al. "Antimicrobial peptide CATH-1 combats Streptococcus suis by targeting the serine/threonine kinase." Int J Antimicrob Agents. 2025 Oct 27:107656. PMID: 41161580
- 20. Shu Wang, Xin Xin, et al. "Plant-specific BLISTER modulates miRNA biogenesis by regulating MIR transcription, HYL1 phosphorylation, and nuclear transport in Arabidopsis." Nucleic Acids Res. 2025 Sep 23;53(18):gkaf1012. PMID: 41063343
- 21. Ningning Li, Hongyu Hou, et al. "PPP2/PP2A-mediated dephosphorylation of LC3B links PINK1-PRKN/Parkin-mediated mitophagy to SCA12 pathogenesis." Autophagy. 2025 Oct 8. PMID: 41059761
- 22. Yuan Luo, Mengqi Sun, et al. "IRE1α translational suppression potentiates STING-dependent chemoresistance in pancreatic cancer." Cell Death Dis. 2025 Oct 6;16(1):680. PMID: 41053142
- 23. Xuecui Yin, Shuang Liu, et al. "Rosmarinic Acid Synthase 1 Phosphorylation by SmMAPK3 Is Required for Salicylic Acid-Induced Salvianolic Acid Accumulation in Salvia miltiorrhiza Hairy Roots." Plant Biotechnol J. 2025 Sep 23. PMID: 40985286
- 24. Ruiheng Luo, Mingliang Ma, et al. "GSK461364 Inhibits NLRP3 Inflammasome by Targeting NEK7 Phosphorylation." Adv Sci (Weinh). 2025 Nov;12(44):e04816. PMID: 40948397
- 25. Lior Almagor, William I. Weis, et al. "Polarity protein Par6 facilitates the processive phosphorylation of Lgl via a dynamic interaction with aPKC." Commun Biol. 2025 Jul 1;8:967. PMID: 40595028
- 26. Hongrui Cao, Rongxiu Cui, et al. "Ca2+-dependent cytoplasmic and nuclear phosphorylation of STOP1 by CPK21 and CPK23 confers ALMT1-dependent aluminum resistance." Nat Commun. 2025 Jun 5;16(1):5225. PMID: 40473618
- 27. Siqi Ge, Sai Wang, et al. "Calcium - responsive phosphorylation of SlLHP1b epigenetically suppresses auxin synthesis to control drought - induced flower drop in tomato." Dev Cell. 2025 May 27:S1534-5807(25)00296-5. PMID: 40460834
- 28. Bin Tian, Yanming Tian, et al. "Duck plague virus US3 kinase phosphorylates and induces STING degradation to inhibit innate immune responses." Poult Sci. 2025 May 26;104(8):105336. PMID: 40446687
- 29. Qian Qian, Weiguo Hu, et al. "The Cytokinin Oxidase/Dehydrogenase TaCKX11 - D Positively Regulates Grain Size in Wheat." J Exp Bot. 2025 May 13:eraf196. PMID: 40357640
- 30. Liang Xu, Ruiyi Zhou, et al. "Social hierarchy modulates drug reinforcement and protein phosphorylation in the nucleus accumbens." Front Pharmacol. 2025 Apr 11:16:1537131. PMID: 40290427
- 31. Yu Fu, Xiaoyan Kang, et al. "The low-dose CHK1 inhibitor prexasertib triggers VDAC1 dephosphorylation to activate mtDNA-STING signaling and synergize immunotherapy." Cell Rep. 2025 May 27;44(5):115605. PMID: 40249707
- 32. Huangzhuo Xiao, Jiaqi Hou, et al. "The receptor-like cytoplasmic kinase OsSTRK1 regulates brassinosteroid signaling by phosphorylating OsGSK2." Cell Rep. 2025 Apr 22;44(4):115569. PMID: 40220291
- 33. Zhi-Man Zhu, Fu-Chun Huo, et al. "METTL3-mediated m6A modification of PAK6 drives cervical cancer progression through activating MAPK14." Int J Biol Macromol. 2025 May;309(Pt 2):142880. PMID: 40194572
- 34. Kai Li, Rui Liu, et al. "Marek's disease virus protein kinase US3 inhibits DNA-sensing antiviral innate immunity via abrogating activation of NF-κB." Microbiol Spectr. 2025 Apr;13(4):e0234724. PMID: 40042340
- 35. Sapir Nachum-Raines, Noy Gamliel, et al. "Cdk1 and PP2A constitute a molecular switch controlling orderly degradation of atypical E2Fs." bioRxiv [Preprint]. 2025 Feb 23:2025.02.23.639703. PMID: 40027684
- 36. Liangyi Zhao, Hua Cassan‐Wang, et al. "Calcium-dependent protein kinase PpCDPK29-mediated Ca2+-ROS signal and PpHSFA2a phosphorylation regulate postharvest chilling tolerance of peach fruit." Plant Biotechnol J. 2025 Jun;23(6):1938-1953. PMID: 40014693
- 37. Zhicheng Huang, Qing Wang, et al. "A multilayered regulatory network mediated by protein phosphatase 4 controls carbon catabolite repression and de-repression in Magnaporthe oryzae." Commun Biol. 2025 Jan 28;8(1):130. PMID: 39875495
- 38. David M. Renner, Nicholas A. Parenti, et al. "Betacoronaviruses Differentially Activate the Integrated Stress Response to Optimize Viral Replication in Lung-Derived Cell Lines." Viruses. 2025 Jan 16;17(1):120. PMID: 39861909
- 39. Yongliang Liu, Jizhe Shi, et al. "Transcriptional Reprogramming Deploys a Compartmentalized ‘Timebomb’ in Catharanthus roseus to Fend Off Chewing Herbivores." Plant Cell Environ. 2025 May;48(5):3236-3256. PMID: 39718032
- 40. Lior Almagor,William Weis, et al. "An Efficient Off-Membrane Switch: Par6 Facilitates Processive Phosphorylation of Lgl's Serine Sites via a Dynamic Interaction with aPKC." Research Square. 12 Jan, 2025
- 41. Xiang-Yang Hao, Tai-Fei Yu, et al. "Somatic embryogenetic receptor kinase TaSERL2 regulates heat stress tolerance in wheat by influencing TaBZR2 protein stability and transcriptional activity." Plant Biotechnol J. 2025 Apr 4
- 42. Yonghong Xie, Kaichong Teng, et al. "The E3 ubiquitin ligase OsPUB9 modulates the abscisic acid signaling complex to respond cold stress in rice." Authorea. June 02, 2025
- 43. Rosa Eskandari, Maryam Fayyazi, et al. "EGO-like complex regulates TOR (Target of Rapamycin) activity and localization in Neurospora." bioRxiv. December 09, 2025
- 44. David M Renner, Nicholas A Parenti, et al. "BETACORONAVIRUSES DIFFERENTIALLY ACTIVATE THE INTEGRATED STRESS RESPONSE TO OPTIMIZE VIRAL REPLICATION IN LUNG DERIVED CELL LINES." bioRxiv. 2024 Sep 26:2024.09.25.614975. PMID: 39386680
- 45. Jihuan Hou, Kunrong Mei, et al. "TGM1/3-mediated transamidation of Exo70 promotes tumor metastasis upon LKB1 inactivation." Cell Rep. 2024 Aug 14;43(8):114604. PMID: 39146185
- 46. Tahrima Saiha Huq, Jean Luo, et al. "Naturally occurring hyperactive variants of human parkin." Commun Biol. 2024 Aug 8;7(1):961. PMID: 39117722
- 47. Shanshan Liu, Yidan Shao, et al. "SepM mutation in Streptococcus mutans clinical isolates and related function analysis." BMC Oral Health. 2024 Jun 25;24(1):730. PMID: 38918777
- 48. Yangkun Li, Lei Mu, et al. "Golgi dispersal in cancer stem cells promotes chemoresistance of colorectal cancer via the Golgi stress response." Cell Death Dis. 2024 Jun 15;15(6):417. PMID: 38879509
- 49. Zhongwei Xu, Kaiyuan Fan, et al. "The application of proteomics and phosphoproteomics to reveal the molecular mechanism of salidroside in ameliorating myocardial hypoxia." Heliyon. 2024 Apr 28;10(9):e30433. PMID: 38737233
- 50. Cancan Zhang, Yinyin Xu, et al. "Phosphorylation of FOXK2 at Thr13 and Ser30 by PDK2 sustains glycolysis through a positive feedback manner in ovarian cancer." Oncogene. 2024 Jun;43(26):1985-1999. PMID: 38734828
- 51. Ananya Acharya, Hélène Bret, et al. "Mechanism of DNA unwinding by MCM8-9 in complex with HROB." Nat Commun. 2024 Apr 27;15(1):3584. PMID: 38678026
- 52. Yuqing Yan, Hui Wang, et al. "OsATL32 ubiquitinates the reactive oxygen species-producing OsRac5–OsRbohB module to suppress rice immunity." J Integr Plant Biol. 2024 Apr 17. PMID: 38629772
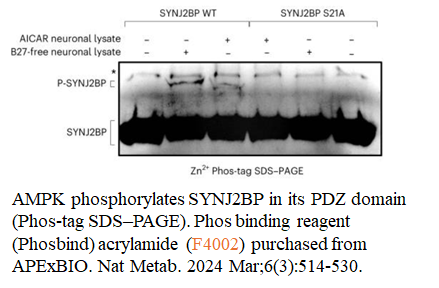
- 53. J Tabitha Hees, Simone Wanderoy, et al. "Insulin signalling regulates Pink1 mRNA localization via modulation of AMPK activity to support PINK1 function in neurons." Nat Metab. 2024 Mar 19;6(3):514–530. PMID: 38504131
- 54. Tiefeng Jin, Lingling Ding, et al. "BUB1/KIF14 complex promotes anaplastic thyroid carcinoma progression by inducing chromosome instability." J Cell Mol Med. 2024 Apr;28(7):e18182. PMID: 38498903
- 55. Deyue Yu, et al. "The Myb73-GDPD2-GA2ox1 transcriptional regulatory module confers phosphate deficiency tolerance in soybean." Plant Cell. 2024 Feb 12:koae041. PMID: 38345432
- 56. Hongliang Wang, et al. "N-terminal acetyltransferase 6 facilitates enterovirus 71 replication by regulating PI4KB expression and replication organelle biogenesis." J Virol. 2024 Feb 20;98(2):e0174923. PMID: 38189249
- 57. Hui Jiang, et al. "A mitophagy sensor PPTC7 controls BNIP3 and NIX degradation to regulate mitochondrial mass." Mol Cell. 2024 Jan 18;84(2):327-344.e9. PMID: 38151018
- 58. Eugenia Wulff-Fuentes, Jeffrey Boakye, et al. "O-GlcNAcylation regulates OTX2's proteostasis." iScience. 2023 Oct 12;26(11):108184. PMID: 38026167
- 59. Anna C Howes, Olga Perisic, et al. "Structural insights into the activation of ataxia-telangiectasia mutated by oxidative stress." Sci Adv. 2023 Sep 29;9(39):eadi8291. PMID: 37756394
- 60. Yunran Zhang, Yunyun Wei, et al. "Cand2 Inhibits CRL-mediated Ubiquitination and Suppresses Autophagy to Facilitate Pathogenicity of Phytopathogenic Fungi." Plant Commun. 2023 Sep 16:100720. PMID: 37718510
- 61. Pengfei Cheng, Yueqiao Wang, et al. "Molecular hydrogen positively regulates nitrate uptake and seed size by targeting nitrate reductase." Plant Physiol. 2023 Aug 25;kiad474. PMID: 37625793
- 62. Heting Wang, Xuya Pan, et al. "CRISPR screen identifies the role of RBBP8 in mediating unfolded protein response induced liver damage through regulating protein synthesis." Cell Death Dis. 2023 Aug 18;14(8):531. PMID: 37591836
- 63. Ananya Acharya, Hélène Bret, et al. "Mechanism of DNA unwinding by hexameric MCM8-9 in complex with HROB." Res Sq. 2023 Jun 26:rs.3.rs-3054483. PMID: 37461676
- 64. Jiahui Liu, Xiaoyun Wu, et al. "A plant RNA virus inhibits NPR1 sumoylation and subverts NPR1-mediated plant immunity." Nat Commun. 2023 Jun 16;14(1):3580. PMID: 37328517
- 65. Kai Niu, Yu Meng, et al. "Phosphorylation of GntR reducesStreptococcus suisoxidative stress resistance and virulence by inhibiting NADH oxidase transcription." PLoS Pathog. 2023 Mar 13;19(3):e1011227. PMID: 36913374
- 66. Yunwen Yang, Suwen Liu, et al. "DNA-dependent protein kinase catalytic subunit (DNA-PKcs) drives chronic kidney disease progression in male mice." Nat Commun. 2023 Mar 11;14(1):1334. PMID: 36906617
- 67. Sun Q, Ma H, et al. "A Self‐Sustaining Antioxidant Strategy for Effective Treatment of Myocardial Infarction." Adv Sci (Weinh) 2023 Feb;10(5) PMID: 36567266
- 68. Wang B, Stevenson EL, et al. "Functional analysis of 110 phosphorylation sites on the circadian clock protein FRQ identifies clusters determining period length and temperature compensation." G3 (Bethesda) 2023 Feb 09;13(2) PMID: 36537198
- 69. J. Tabitha Hees, Angelika B. Harbauer, et al. "Insulin signaling regulates Pink1 mRNA localization via modulation of AMPK activity to support PINK1 function in neurons." bioRvix. February 06, 2023
- 70. Akhtari, Golnoush, et al. "The Relationship Between TOR Activity and the Circadian Clock of Neurospora crassa." Communities & Collections. Aug 04, 2023
- 71. Yan Xiong, Jaiprakash Sharma, et al. "TFEB degradation is regulated by an IKK/β-TrCP2 phosphorylation-ubiquitination cascade." bioRvix. October 19, 2023
- 72. Meng-Yu Liu, Li-Xiao Sun, et al. "De NovoPurine Nucleotide Biosynthesis Pathway Is Required for Development and Pathogenicity inMagnaporthe oryzae." J Fungi (Basel). 2022 Aug 29;8(9):915. PMID: 36135640
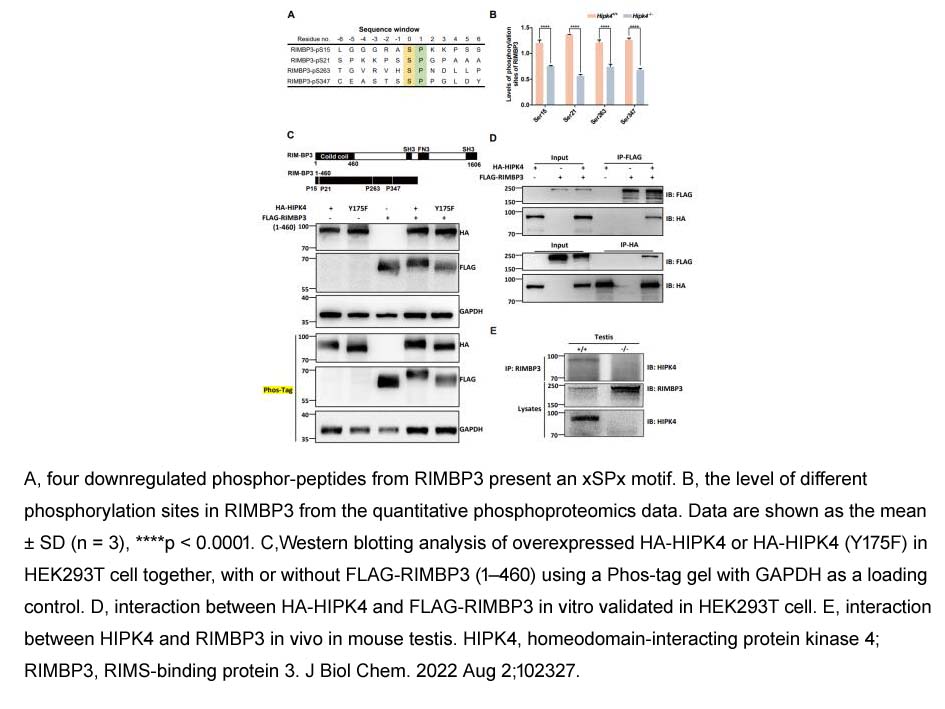
- 73. Xiaofei Liu, Chunyan Zang, et al. "Homeodomain-interacting protein kinase HIPK4 regulates phosphorylation of manchette protein RIMBP3 during spermiogenesis." J Biol Chem. 2022 Aug 2;102327. PMID: 35931115
- 74. Jian Huang, Luxin Wang, et al. "CDC-like kinase 4 deficiency contributes to pathological cardiac hypertrophy by modulating NEXN phosphorylation." Nat Commun. 2022 Jul 30;13(1):4433. PMID: 35907876
- 75. Huilin Li, Jiali Ou, et al. "ISX-9 potentiates CaMKIIδ-mediated BMAL1 activation to enhance circadian amplitude." Commun Biol. 2022 Jul 28;5(1):750. PMID: 35902736
- 76. Jia Yuan, Qingchen Zhu, et al. "Ezh2 competes with p53 to license lncRNA Neat1 transcription for inflammasome activation." Cell Death Differ. 2022 May 14. PMID: 35568718
- 77. Hong Gao, Liangyu Jiang, et al. "GmMKK4‐activated GmMPK6 stimulates GmERF113 to trigger resistance to Phytophthora sojae in soybean." Plant J. 2022 Jul;111(2):473-495. PMID: 35562858
- 78. Gang Xiang, Shuxuan Wang, et al. "UBR5 targets tumor suppressor CDC73 proteolytically to promote aggressive breast cancer." Cell Death Dis. 2022 May 12;13(5):451. PMID: 35551175
- 79. Guangli Wang, Chan Fan, et al. "Type VI secretion system-associated FHA domain protein TagH regulates the hemolytic activity and virulence of Vibrio cholerae." Gut Microbes. 2022 Jan-Dec;14(1):2055440. PMID: 35383540
- 80. Yajing Song, Ruofan Niu, et al. "OsSLA1 functions in leaf angle regulation by enhancing the interaction between OsBRI1 and OsBAK1 in rice." Plant J. 2022 Mar 11. PMID: 35275421
- 81. Zhao-Huan Zhang, Jian-Mei Xiong, et al. "WNK3-PER1 interactions regulate the circadian rhythm in the suprachiasmatic nucleus in rats." Am J Transl Res. 2022 Feb 15;14(2):1001-1009. PMID: 35273702
- 82. Guo-Xiang Zhou, Wei-Bo Liu, et al. "Environmental cadmium impairs blood-testis barrier via activating HRI-responsive mitochondrial stress in mice." Sci Total Environ. 2021 Dec 9;810:152247. PMID: 34896485
- 83. Weiwei Fu, Megan T Zhao, et al. "NUAK family kinase 2 is a novel therapeutic target for prostate cancer." Mol Carcinog. 2021 Nov 24. PMID: 34818445
- 84. Qing Ye, Yingying Zhang, et al. "The important roles and molecular mechanisms of annexin A2 autoantibody in children with nephrotic syndrome." Ann Transl Med. 2021 Sep;9(18):1452. PMID: 34734004
- 85. Qian Zhang, Tao Song, et al. "OsANN4 modulates ROS production and mediates Ca2+ influx in response to ABA." BMC Plant Biol. 2021 Oct 18;21(1):474. PMID: 34663209
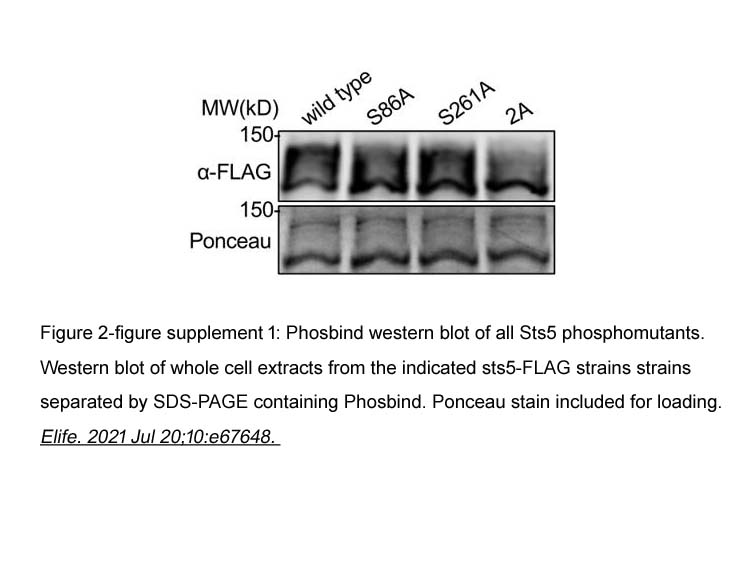
- 86. Joseph O Magliozzi, James B Moseley. "Pak1 kinase controls cell shape through ribonucleoprotein granules." Elife. 2021 Jul 20;10:e67648. PMID: 34282727
- 87. Fan XJ, Wang YL, et al. "NONO phase separation enhances DNA damage repair by accelerating nuclear EGFR-induced DNA-PK activation." Am J Cancer Res. 2021 Jun 15;11(6):2838-2852. PMID: 34249431
- 88. Yamin Meng, Xing Zhang, et al. "A Novel Nitrogen and Carbon Metabolism Regulatory Cascade Is Implicated in Entomopathogenicity of the Fungus Metarhizium robertsii." mSystems. 2021 Jun 29;6(3):e0049921. PMID: 34156296
- 89. Wirth M, Mouilleron S, et al. "Phosphorylation of the LIR Domain of SCOC Modulates ATG8 Binding Affinity and Specificity." J Mol Biol. 2021 Jun 25;433(13):166987. PMID: 33845085
- 90. Haijing Guo, Jen-Hsuan Wei, et al. "Importin α phosphorylation promotes TPX2 activation by GM130 to control astral microtubules and spindle orientation." J Cell Sci. 2021 Feb 19;134(4):jcs258356. PMID: 33526712
- 91. Amina Merwa LEKEHAL. "altFUS, the second protein encoded in the FUS gene, inhibits mitochondrial recruitment of PARKIN." Mars, 2021
- 92. Li kaihuai, Gaoge Xu, et al. "A predatory soil bacterium reprograms a quorum sensing signal system to regulate antifungal weapon production in a cyclic-di-GMP-independent manner." Research Square. March 11th, 2021
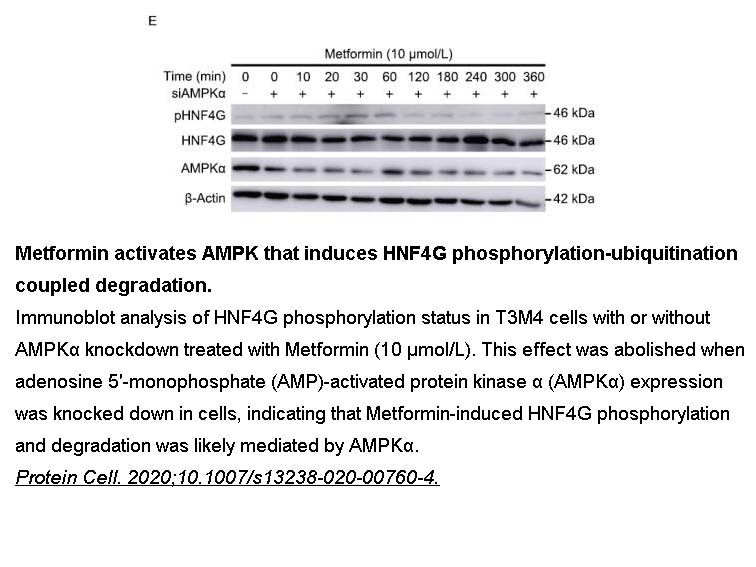
- 93. Chengcheng Wang, Taiping Zhang, et al. "Metformin inhibits pancreatic cancer metastasis caused by SMAD4 deficiency and consequent HNF4G upregulation." Protein & Cell (2020);31 July 2020. PMID: 32737864
- 94. Wen-Jie Shu, Runfa Chen, et al. "Rph1 coordinates transcription of ribosomal protein genes and ribosomal RNAs to control cell growth under nutrient stress conditions." Nucleic Acids Res. 2020 Sep 4;48(15):8360-8373. PMID: 32619236
- 95. Domagoj Bareti_x005f_x0001_, Michael Jenkyn-Bedford, et al. "Cryo-EM Structure of the Fork Protection Complex Bound to CMG at a Replication Fork." Mol Cell. 2020 Apr 29;S1097-2765(20)30254-9. PMID: 32369734
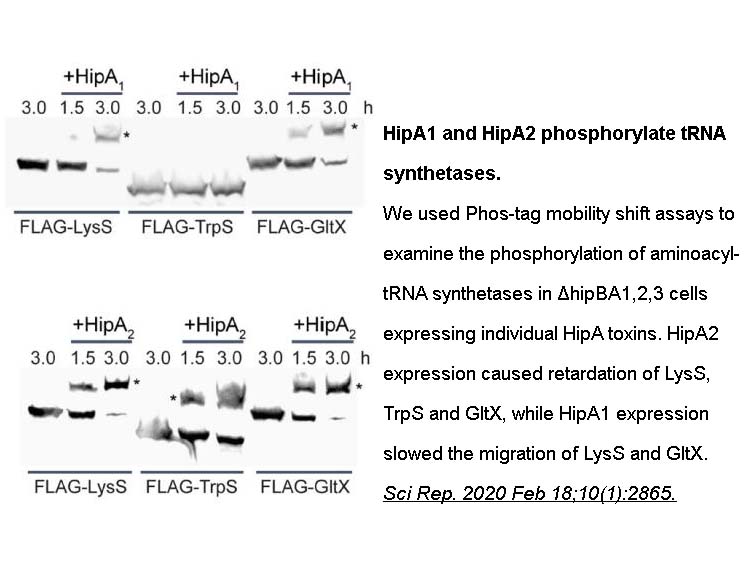
- 96. Huang CY, Gonzalez-Lopez C, et al. "hipBA toxin-antitoxin systems mediate persistence in Caulobacter crescentus." Sci Rep. 2020;10(1):2865. PMID: 32071324
- 97. Haijing Guo, Jen-Hsuan Wei, et al. "TPX2 activation by GM130 controls astral microtubule formation and spindle orientation." bioRxiv. March 17, 2020
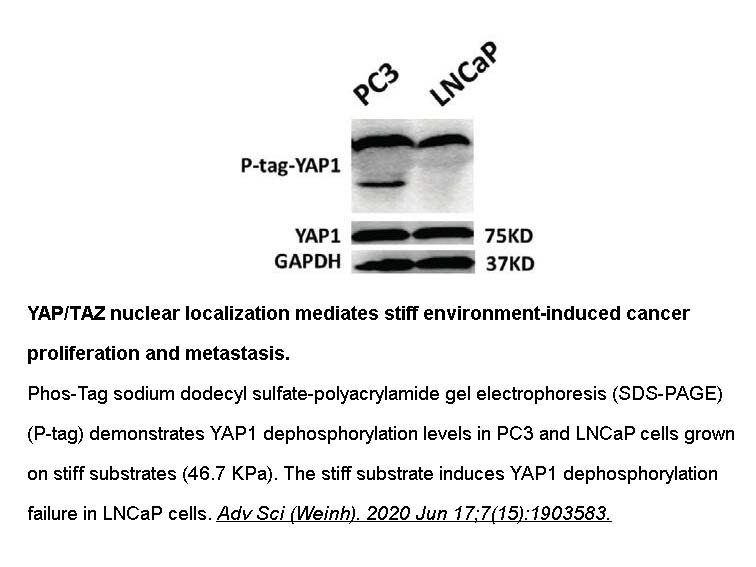
- 98. Zhixiao Liu, Liujun Wang, et al. "Heterogeneous Responses to Mechanical Force of Prostate Cancer Cells Inducing Different Metastasis Patterns." Adv. Sci. 2020, 1903583
- 99. Xie Y, Lv X, et al. "HPD degradation regulated by the TTC36-STK33-PELI1 signaling axis induces tyrosinemia and neurological damage." Nat Commun. 2019 Sep 19;10(1):4266. PMID: 31537781
- 100. Jing ZF, Bi JB, et al. "miR-19 promotes the proliferation of clear cell renal cell carcinoma by targeting the FRK-PTEN axis." Onco Targets Ther. 2019 Apr 10;12:2713-2727. PMID: 31043790
- 101. Wang B, Kettenbach AN, et al. "The Phospho-Code Determining Circadian Feedback Loop Closure and Output in Neurospora." Mol Cell. 2019 Mar 26. pii: S1097-2765(19)30177-7. PMID: 30954403
- 102. Jordann Alyse Smak. "Cell Cycle-Specific Functions and Regulation of the DNA Nuclease SNM1B." University of Michigan. 2019
- 103. Li Zhang, Ruci Wang, et al. "Separable regulation of POW1 in TAF2-mediated grain development and BR-mediated leaf angle formation in rice." bioRxiv. 2019 November 04
- 104. Yu J, Deng T, et al. "Structural basis for protein phosphatase 1 recruitment by glycogen-targeting subunits." FEBS J. 2018 Dec;285(24):4646-4659. PMID: 30422398
- 105. Yin Y, Wu S, et al. "The MAPK kinase BcMkk1 suppresses oxalic acid biosynthesis via impeding phosphorylation of BcRim15 by BcSch9 in Botrytis cinerea." PLoS Pathog. 2018 Sep 13;14(9):e1007285. PMID: 30212570
- 106. Sauvé V, Sung G, et al. "Mechanism of parkin activation by phosphorylation." Nat Struct Mol Biol. 2018 Jul;25(7):623-630. PMID: 29967542
·No phospho-antibody required: Particularly suitable for studying phosphorylation sites lacking commercial antibodies, and offers a more cost-effective solution.
·Excellent separation performance: Enables fine separation and simultaneous detection of non-phosphorylated proteins and proteins with different phosphorylation levels on the same membrane.
·Complete kit: Supplied with sufficient high-quality MnCl₂; no additional purchase required.
·High affinity: Exhibits high specificity and high affinity for any phosphorylated group on proteins.
·High safety: Free of radioactive elements, chemical labels, and fluorescent markers.
·Broad applicability: No species or sample type restrictions; recognizes all phosphorylation forms at Ser, Thr, and Tyr residues.
·High sensitivity: Detection limit reaches the ng level, which can be further enhanced by using high-sensitivity chemiluminescent substrates.
·Easy operation: The workflow is essentially the same as conventional SDS-PAGE, requiring no additional specialized equipment.
·Strong compatibility: Compatible with various downstream analyses, including CBB staining, Western blotting, two-dimensional electrophoresis, and mass spectrometry (MS).
[1] M. J. Hubbard, P. Cohen. On target with a new mechanism for the regulation of protein phosphorylation. Trends Biochem. Sci. 18: 172 (1993).
[2] B. Agnew et al. Compositions and methods for detection and isolation of phosphorylated molecules. US Patent # 7,102,005. (September 5, 2006).
| Storage | 2-10℃ |
| M.Wt | 594.71 |
| Formula | C33H38N8O3 |
| Solubility | >29.7mg/mL in DMSO |
| Chemical Name | N-(2-acrylamidoethyl)-6-(((3-(bis(pyridin-2-ylmethyl)amino)-2-hydroxypropyl)(pyridin-2-ylmethyl)amino)methyl)nicotinamide |
| Canonical SMILES | O=C(C1=CN=C(CN(CC2=NC=CC=C2)CC(O)CN(CC3=NC=CC=C3)CC4=NC=CC=C4)C=C1)NCCNC(C=C)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Related Biological Data
Related Biological Data
Related Biological Data
Related Biological Data
Related Biological Data
Related Biological Data
Related Biological Data

Related Biological Data
Q: Product (F4002) – How many gels can be prepared?
A: Based on a Mini PAGE gel (8.3 cm × 7.3 cm, 1 mm thick) with a 50 μM Phosbind separating gel (5 mL/gel), 10 mg yields 3.3 mL of stock solution. Using 0.05 mL of Phosbind per gel at 50 μM, approximately 66 gels can be prepared; 5 mg yields about 33 gels. The number varies with Phosbind concentration — higher concentration, fewer gels.
Q: Storage conditions for product (F4002)
A: Phosbind acrylamide (F4002) is a yellow oil. Store at 4°C protected from light for up to 1 year. Prepared stock solutions can be stored at 4°C protected from light for at least 3 months. Mn²⁺-Phosbind gels should be freshly prepared.
Q: What are the applications of Phosbind?
A: Suitable for purified proteins, cell lysates, in vitro kinase reactions, and various sample types (animal, plant, microbial). Compatible with Mn²⁺ or Zn²⁺ systems. Enables phosphoprotein detection without specific antibodies, supporting downstream analyses such as CBB staining, Western blotting, 2D electrophoresis, and mass spectrometry. Works with standard electrophoresis, transfer, and imaging equipment.
Q: Is Phosbind (F4002) suitable for tagged proteins (e.g., His, GFP), in vitro kinase assays, multi-band proteins (e.g., Erk), and mutant phosphoprotein detection?
A: Fully suitable. In vitro kinase reaction mixtures can be directly loaded without special purification.
Q: What is the molecular weight range for Phosbind?
A: Works well for proteins in the 30–130 kDa range, with reported successful separation of proteins up to 350 kDa.
Q: What are the advantages of Phosbind over traditional phosphodetection methods (e.g., phospho-antibodies, radiolabeling)?
A: ① Non-radioactive, safe; ② No phospho-specific antibodies needed, detects all phosphorylation sites; ③ Resolves differentially phosphorylated isoforms; ④ Simple, compatible with standard SDS-PAGE workflows.
Q: Which amino acid phosphorylations can Phosbind detect?
A: Phosbind specifically binds phosphate groups on all phosphorylated amino acids (pSer, pThr, pTyr, etc.), independent of amino acid type or sequence.
Q: What is the detection sensitivity of Phosbind?
A: ng-level sensitivity; can be further enhanced with high-sensitivity chemiluminescent substrates.
Q: Can Phosbind distinguish the number of phosphate groups on a protein?
A: Yes, it distinguishes different phosphorylation levels — more phosphate groups result in slower migration, producing multiple resolved bands. However, it cannot determine the exact number of phosphorylation sites per protein; mass spectrometry is required for that.
Q: Can Phosbind be used for DNA analysis?
A: Yes, for detecting and separating DNA phosphorylation sites (e.g., 5'-end phosphorylated DNA, phosphorylated oligonucleotides), based on the same phosphate-binding principle as for proteins.
Q: What are the main Phosbind series products and their applications?
A: Three main products: Phosbind acrylamide (F4002) and Phosbind Biotin series (F4001, F4004). F4002 is used to prepare SDS-PAGE gels for separating phosphorylated from non-phosphorylated proteins, followed by detection with non-phospho primary antibodies. The Biotin series (F4001/F4004) are used after transfer to PVDF membranes with streptavidin-HRP for antibody-free detection. F4004 has a longer linker for higher sensitivity; F4001 has better solubility — F4001 is recommended first.
Q: How to choose between Mn²⁺-Phosbind and Zn²⁺-Phosbind systems?
A: ① Mn²⁺: Compatible with standard Tris-Glycine buffers, easier to use, more widely reported — first choice. ② Zn²⁺: Neutral Bis-Tris gel system, higher binding affinity, suitable for difficult-to-resolve or high-molecular-weight proteins (>200 kDa).
Q: What quality control guarantees are there for Phosbind products?
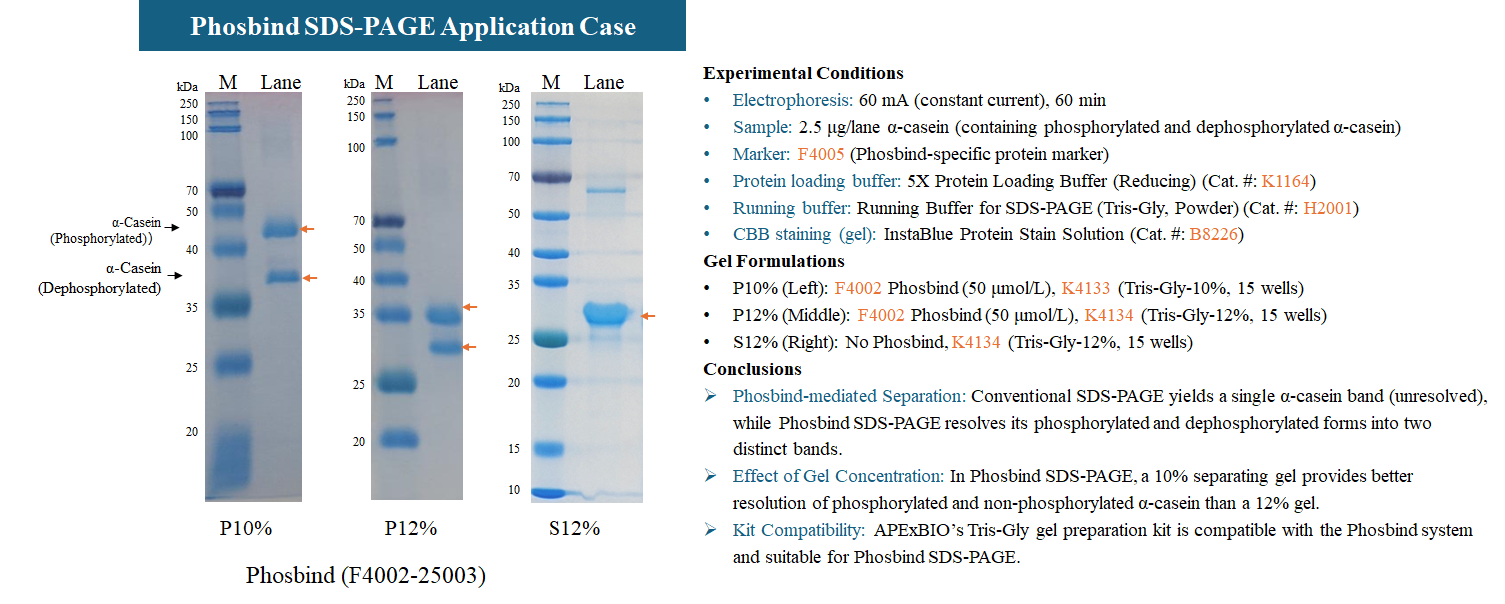
A: Every batch undergoes strict QC: ① Purity testing; ② Activity validation using standard samples (e.g., dephosphorylated α-casein); ③ Stability testing under different storage conditions; ④ QC report included with each batch for traceability and reproducibility.
Q: What are the key steps and precautions for preparing Phosbind SDS-PAGE gels?
A: ① Add Phosbind acrylamide and metal ions (Mn²⁺ or Zn²⁺, typically 1:2 molar ratio) to the separating gel. Try Phosbind concentrations from 20–50–100 μM. ② Mn²⁺ gels should be freshly prepared; Zn²⁺ gels can be stored at 4°C protected from light for up to 3 months. ③ Avoid EDTA, high concentrations of detergents (e.g., SDS), and inorganic salts.
Q: How to optimize sample loading and gel concentration?
A: ① Loading: 1–5 μg for purified protein, 10–30 μg for cell/tissue lysates (adjust based on expression). ② Gel concentration: 8% for <60 kDa, 6% for >60 kDa; for >200 kDa, add 3–5% agarose to increase gel strength. ③ Phosbind concentration: Titrate from 20 μM up to 50–100 μM; use the lowest concentration that gives good separation.
Q: Cloudiness when preparing Phosbind stock solution — is it normal? Can water be used directly?
A: Cloudiness is normal due to methanol; let it sit to clear. For stock preparation, dissolve in methanol first, then add water. Using water alone makes dissolution difficult and slow. If insoluble material remains, centrifuge and use the supernatant.
Q: How to choose a protein marker? Can it clearly indicate molecular weights?
A: Use an EDTA-free protein marker (e.g., F4005) to avoid band distortion. The marker in Phosbind results only indicates transfer efficiency; it cannot be used to accurately infer sample protein molecular weights due to phosphorylation-induced mobility shifts.
Q: Does ATP used in in vitro phosphorylation reactions affect electrophoresis results?
A: ATP itself carries phosphate groups. At 2.0 mM, it has no special effect. Concentrations above 5 mM may compete for Phosbind binding or interfere with gel polymerization. Dialysis or precipitation to remove ATP is recommended before loading.
Q: What are the special steps for Phosbind Western blot transfer?
A: After electrophoresis, the gel must be washed with transfer buffer or running buffer containing 1–10 mM EDTA for 10 minutes, 1–3 times, to remove metal ions and improve transfer efficiency. After transfer, follow standard WB procedures. Wet transfer and PVDF membranes are recommended.
Q: What causes band distortion with Phosbind and how to fix it?
A: ① Sample contains EDTA/salts/detergents: TCA precipitation or dialysis. ② Viscous sample: Boil thoroughly or dilute. ③ Acidic sample (yellow/orange after loading buffer): Neutralize with Tris to blue-purple. ④ Prestained marker contains EDTA: Switch to EDTA-free marker (e.g., F4005). ⑤ Empty lanes: Add equal volume of loading buffer to empty lanes to avoid gradient artifacts.
Q: What causes smearing or severe trailing and how to fix it?
A: Protein degradation (inactive protease inhibitors), incomplete solubilization (particulates), insufficient SDS in running buffer, uneven gel polymerization, excessive Phosbind or Mn²⁺ concentration, or high temperature/fast electrophoresis. Solutions: fully lyse and centrifuge samples, use fresh inhibitors and running buffer, optimize conditions with cooling, and titrate Phosbind/Mn²⁺ concentrations.
Q: What causes weak or no phosphoprotein signal and how to fix it?
A: Low phosphorylation levels (often reversible), insufficient sample amount, inadequate sample protection (insufficient or inactive phosphatase inhibitors), poor transfer efficiency, inappropriate blocking conditions, or poor antibody quality. Solutions: increase sample amount, optimize sample handling and pre-transfer washing, use high-quality antibodies and appropriate blocking. Ensure thorough EDTA treatment before transfer.
Q: What causes difficult transfer and how to fix it?
A: ① Mn²⁺ not removed before transfer: Wash thoroughly with EDTA-containing transfer/running buffer. ② Gel too stiff: Reduce acrylamide concentration or methanol concentration.
Q: What causes high background or multiple nonspecific bands and how to fix it?
A: Similar to standard Western blot — excessive primary/secondary antibody, insufficient blocking, inadequate washing, poor antibody specificity, or improper PVDF membrane handling. Solutions: reduce antibody concentrations, optimize blocking and washing conditions, use highly specific antibodies.
Q: What causes multiple bands and what do you suggest?
A: May result from different phosphorylation levels or protein degradation. Run a parallel SDS-PAGE gel without Phosbind to rule out degradation.
Q: What causes phosphoprotein bands not shifting as expected or showing little difference from regular gels, and how to fix it?
A: ① EDTA treatment omitted or insufficient, preventing efficient transfer. ② Phosbind or Mn²⁺ concentration too low to cause a clear mobility shift. ③ Target protein has few phosphorylation sites or low modification levels. ④ Gel concentration too low for proper separation. Solutions: ① Thorough EDTA pre-treatment before transfer. ② Optimize Phosbind/Mn²⁺ concentrations. ③ Increase loading or stimulate cells to enhance phosphorylation. ④ Use appropriate gel concentration.
Q: Phosbind gels are fragile, especially for high-molecular-weight proteins — what to do?
A: Phosbind gels are more fragile than regular gels due to the added Phosbind and MnCl₂. For low-concentration gels (e.g., for high-MW proteins), add 0.5% agarose to increase mechanical strength.
Q: How to verify poor separation between phosphorylated and non-phosphorylated proteins?
A: ① Use a positive control (e.g., mixture of phosphorylated and dephosphorylated α-casein) to confirm system performance. ② Run a parallel regular SDS-PAGE gel to rule out degradation. ③ Adjust Phosbind concentration or switch from Mn²⁺ to Zn²⁺ system. ④ Treat samples with λ protein phosphatase — if the shifted bands disappear, it strongly confirms phosphorylation.
Positive control reference:

Q: What are the suggested experimental controls?
A:
|
Design |
Control Type |
Treatment |
Purpose / Explanation |
|
Experimental Design |
Control |
Untreated |
Baseline control |
|
|
Model / Negative control |
Stimulated |
Induce phosphorylation changes |
|
|
Treatment group |
Stimulated + drug treatment |
Assess drug effect on phosphorylation |
|
PAGE Control |
Positive control |
α-Casein |
Validate Phosbind gel system |
|
|
Phosphatase-treated / Dephosphorylation control |
λ protein phosphatase treatment |
Confirm phosphorylation. Disappearance of shifted bands on Phosbind gel proves mobility shift is due to phosphorylation |
|
|
Internal control |
Run parallel standard SDS-PAGE gel, probe with same non-phospho antibody |
Confirm band separation on Phosbind gel is due to phosphorylation, not degradation or other modifications |
Q: Can Phosbind (F4002) be used with a pre-made gel kit (Tris-Glycine system)?
A: Yes, it can be used with our Tris-Glycine fixed-concentration gel kits (K4131–K4135: 6%/8%/10%/12%/15%). For other brands of gel kits, we recommend testing compatibility.
Q: What are the related products for Phosbind experiments?
|
Category |
Product/Description |
|
Phosbind Series |
Phosbind Gel Method (Add during resolving gel preparation, requires non-phosphorylated control) • Phos binding reagent (Phosbind) acrylamide (F4002, separates phosphorylated and non-phosphorylated proteins) Phosbind Biotin Method (Incubate PVDF membrane after transfer, only detects phosphorylated proteins, no primary antibody needed) • Phosbind Biotin BTL-104 (F4001, high solubility, recommended first choice) • Phosbind Biotin BTL-105 (F4004, long linker, high sensitivity) |
|
Control Setup |
• Positive control: α-casein (different phosphorylation levels) • Phosphatase-treated control/dephosphorylation control (treatment enzyme): λ Protein Phosphatase (RNase-free) (K1102) |
|
Sample Preparation |
Animal Tissue & Cell Lysate • RIPA Lysis Buffer (Strong, with inhibitors) (K1020) Protein Protection Inhibitor Cocktail (EDTA-free, universal type) • Protease Inhibitor Cocktail: K1007 (classic multi-literature version), K4002 (K1007 upgraded version) • Phosphatase Inhibitor Cocktail: K1015 (more comprehensive and well-documented, A+B tube), K4004 (single tube, aqueous) • Deacetylase Inhibitor Cocktail (K1017) • Protease & Phosphatase Inhibitor Cocktail (K4006, single tube, aqueous) Protein Extraction Kits • Cellular Compartment Extraction: Nuclear Extraction Kit (K1137), Mitochondrial Extraction Kit (K1138) • Active Protein Extraction: Kits (Mammalian-K4061, Insect Cell-K4062, Bacteria-K4063) |
|
Protein Quantitation |
BCA Kit (compatible with RIPA lysis buffer) • BCA Protein Assay Kit (K4101, classic version, 50-2000 μg/mL range) • Micro BCA Protein Assay Kit (K4102, low protein concentration version, 0.5~20 μg/mL range) Bradford Kit (detergent-compatible) • Detergent Compatible Bradford Protein Assay Kit (K4104, detergent-compatible version, 0.1-1.5 mg/mL range) |
|
Protein Electrophoresis |
Gel Casting Kits • Tris-Gly fixed-concentration gel casting kits (K4131-K4135, 6%-15%) Loading Buffer • Reducing: 5X Protein Loading Buffer (Reducing) (K1164) • Non-reducing: 5X Protein Loading Buffer (No Reducing Buffer) (K1165) Protein Marker (compatible with Phosbind products, highly recommended) • Prestained Protein Marker (Triple color, EDTA free, 10-250 kDa) (F4005) Running Buffer (Tris-Gly system) • 10X Running Buffer (K1196) |
|
Western Transfer |
Transfer Buffer (Tris-Gly system) • 10X Transfer Buffer (K1198) • EDTA (B7197, 0.5M product, use concentration 1~10 mM) |
|
Blocking & Antibody Incubation |
Blocking Buffer • SwiftBlock™ Blocking Buffer for Western Blot (K4129, non-protein); BSA Blocking Buffer (K4106) Antibody Diluent • Primary Antibody Diluent (K4140), Secondary Antibody Diluent (K4662) Antibodies • Primary antibody: rich primary antibody products • Secondary antibody: HRP class (e.g., Goat Anti-Rabbit IgG (H+L)-K1223, Goat Anti-Rabbit IgG (H+L)-K1221) Antibody Stripping Buffer • Neutral K4111 (first choice), Acidic K4111, Weak Base K4113, Strong Base K4114 |
|
Protein Detection |
Gel Detection • Protein Fast Stain (CBB staining method, B8226) Chemiluminescence Detection • Ultra-sensitive ECL (K1231) |
|
Antibody Stripping |
Antibody Stripping/Removal Buffer (Stripping buffer) • Specialized antibody stripping buffers: Neutral K4112 (first choice), Acidic K4111, Weak Base K4113, Strong Base K4114 |