PD168393
PD168393 is an irreversible kinase inhibitor of epidermal growth factor receptor (EGFR) with IC50 value of 0.70±0.09 nM and continued to have suppressed kinase activity after 8 hr in compound-free medium.[1]
The epidermal growth factor receptor (EGFR; ErbB-1; HER1 in humans) is the cell-surface receptor for members of the epidermal growth factor family (EGF-family) of extracellular protein ligands. Upon activation by its growth factor ligands, EGFR undergoes a transition from an inactive monomeric form to an active homodimer.[2] In addition to forming homodimers after ligand binding, EGFR may pair with another member of the ErbB receptor family to create an activated heterodimer. EGFR dimerization stimulates its intrinsic intracellular protein-tyrosine kinase activity. [3] As a result, autophosphorylation of several tyrosine (Y) residues in the C-terminal domain of EGFR occurs which elicits downstream activation and signaling by several other proteins that associate with the phosphorylated tyrosines through their own phosphotyrosine-binding SH2 domains. These downstream signaling proteins initiate several signal transduction cascades, principally the MAPK, Akt and JNK pathways, leading to DNA synthesis and cell proliferation.[4]
Mutations that lead to EGFR overexpression or overactivity have been associated with a number of cancers, thus many therapeutic approaches are aimed at the EGFR now. PD 168393 can poss a high specificity toward the EGFr with Cys-773 which inhibit the EGFR tyrosine kinase, which is on the cytoplasmic side of the receptor. Without kinase activity, EGFR is unable to activate itself, which is a prerequisite for binding of downstream adaptor proteins. Ostensibly by halting the signaling cascade in cells that rely on this pathway for growth, tumor proliferation and migration is diminished.[5]
PD168393 can enhance paclitaxel-induced DNA fragmentation, sub-G1 fraction accumulation, mitochondrial membrane dysfunction, cytochrome C release, caspase-3 activation and eventually apoptosis in vitro by MTT assay and median-effect analysis. In conclusion, the combination of paclitaxel and PD168393 produced a profound synergistic growth inhibition of AIPC cells,resulting in clinical benefits and warrants further investigation.[6]
References:
1.David W. Fry. et al. Specific, irreversible inactivation of the epidermal growth factor receptor and erbB2, by a new class of tyrosine kinase inhibitor. Pharmacology. Proc Natl Acad Sci U S A. 1998, 95(20): 12022–12027.
2.Yosef Yarden and Joseph Schlessinger. "Epidermal Growth-Factor Induces Rapid, Reversible Aggregation of the Purified Epidermal Growth-Factor Receptor". Biochemistry 1987,26 (5): 1443–1451.
3.Downward J, Parker P, Waterfield MD. "Autophosphorylation sites on the epidermal growth factor receptor". Nature 1984,311 (5985): 483–5.
4.Oda K, Matsuoka Y, Funahashi A, Kitano H. "A comprehensive pathway map of epidermal growth factor receptor signaling". Mol. Syst. Biol,2005,1 (1).
5.Olive DM ."Quantitative methods for the analysis of protein Phosphorylation In drug development". Expert Rev Proteomics2004, 1(3):327–41.
6.Pu YS1, Hsieh MW, et al. Epidermal growth factor receptor inhibitor (PD168393) potentiates cytotoxic effects of paclitaxel against androgen-independent prostate cancer cells. Biochem Pharmacol.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 369.22 |
| Cas No. | 194423-15-9 |
| Formula | C17H13BrN4O |
| Solubility | insoluble in H2O; ≥1 mg/mL in EtOH with gentle warming and ultrasonic; ≥18.45 mg/mL in DMSO |
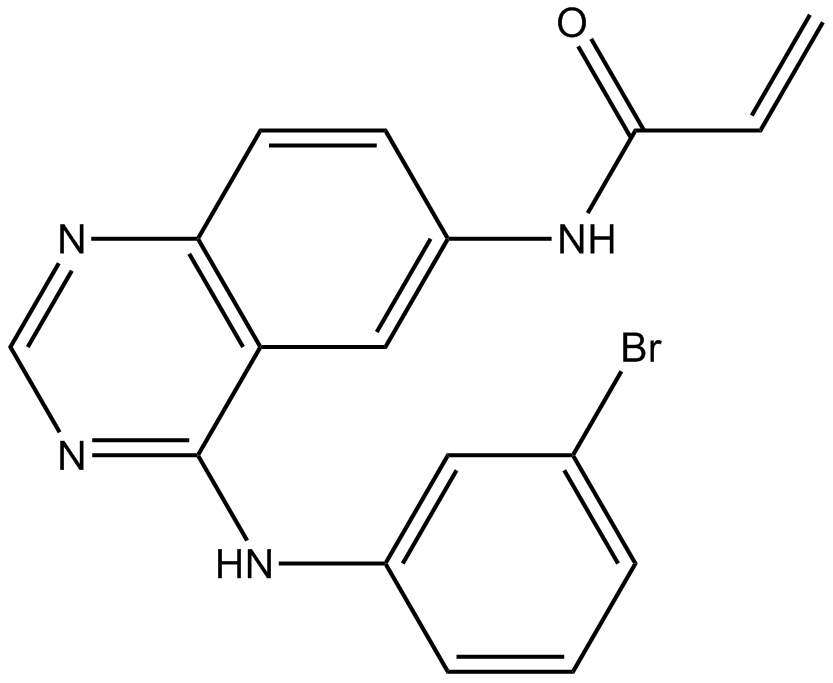
| Chemical Name | N-[4-(3-bromoanilino)quinazolin-6-yl]prop-2-enamide |
| SDF | Download SDF |
| Canonical SMILES | C=CC(Nc(cc1)cc2c1ncnc2Nc1cccc(Br)c1)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1-3]: | |
|
Cell lines |
A431 cells, MDA-MB-453 cells, HS-27 human fibroblasts, 3T3-Her2 cells |
|
Preparation method |
The solubility of this compound in DMSO is > 18.5 mg/mL. General tips for obtaining a higher concentration: Please warm the tube at 37 ℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reacting condition |
2 μM, 1 hr |
|
Applications |
PD168393 completely suppressed EGF-dependent receptor autophosphorylation in A431 cells, with continous suppression even after 8 hr in compound-free medium. PD168393 inhibited heregulin-induced tyrosine phosphorylation in MDA-MB-453 cells with IC50 of 5.7 nM. PD 168393 inhibited EGFr autophosphorylation in A431 human epidermoid carcinoma cells. PD168393 inhibited EGF-mediated tyrosine phosphorylation in HS-27 human fibroblasts with IC50 of 1-6 nM. PD168393 potently inhibited Her2-induced tyrosine phosphorylation with IC50 of ~100 nM in 3T3-Her2 cells. PD168393 inhibited phosphorylation of PLCγ1/Stat1/Dok1/δ-catenin in 3T3-Her2 cells. PD168393 completely inhibited AKT and ERK phosphorylation at concentrations as low as 0.03 umol/L. PD168393 induced apoptosis and inhibited cell growth in ErbB2 positive lung and breast cancer cell lines. |
| Animal experiment [1,4]: | |
|
Animal models |
Nude mice bearing A431 human epidermoid carcinoma xenograft |
|
Dosage form |
Intraperitoneal injection, 58 mg/kg on days 10–14, 17–21, and 24–28 |
|
Application |
PD 168393 (58 mg/kg, i.p.) produced tumor growth inhibition of 115%. PD 168393 reduced the phosphotyrosine content of EGFr l by 50% 24 hr after injection. In the rat model of CIBP, PD168393 (10 μg, intrathecal injection, 9 days) significantly reduced the mRNA expressions of Akt-1 and P38MAPK and the protein levels of p-Akt-1 and p-P38MAPK in spinal cord tissues of rats. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Fry D W, Bridges A J, Denny W A, et al. Specific, irreversible inactivation of the epidermal growth factor receptor and erbB2, by a new class of tyrosine kinase inhibitor. Proceedings of the National Academy of Sciences, 1998, 95(20): 12022-12027. [2]. Bose R, Molina H, Patterson A S, et al. Phosphoproteomic analysis of Her2/neu signaling and inhibition[J]. Proceedings of the National Academy of Sciences, 2006, 103(26): 9773-9778. [3]. Li G et al. Modulation of ErbB2 blockade in ErbB2-positive cancers: the role of ErbB2 Mutations and PHLDA1. PLoS One. 2014 Sep 19;9(9):e106349. [4]. Jiang J, Zhang J, Yao P, et al. Activation of spinal neuregulin 1-ErbB2 signaling pathway in a rat model of cancer-induced bone pain[J]. International journal of oncology, 2014, 45(1): 235-244. | |
| Description | PD168393 is a potent, cell-permeable, irreversible inhibitor of EGFR with IC50 value of 700 pM. | |||||
| Targets | EGFR | |||||
| IC50 | 700 pM | |||||
Quality Control & MSDS
- View current batch:
-
Purity = 98.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
- Datasheet
Chemical structure