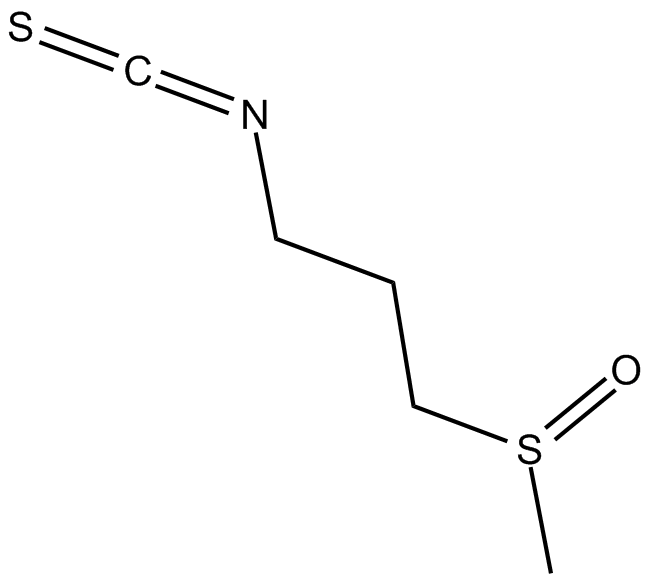
Iberin
Iberin is a phase II detoxification enzyme inducer.
Phase II enzymes are found to protect against chemical carcinogenesis, and the selectivity of isothiocyanates in inducing such enzymes is of interest in view of recent epidemiological studies showing a decreased incidence of cancer.
In vitro: Previous study fhound that the treatment of neuroblastoma cells with iberin led to a dose- and time-dependent growth inhibition, increased cytotoxicity, and G1 or G2 cell cycle arrest. The iberin-induced cell cycle arrest was related to the inhibition of Cdk2, Cdk4, and Cdk6 protein expression. DNA-staining pattern analyses revealed an increase in apoptotic cell death in iberin-treated cells, and FLICA staining founf that iberin could induce apoptosis, which was associated with the activation of PARP, caspase-9, and caspase-3 [1].
In vivo: In animal study, the ability of iberin was tested to increase tissue levels of the phase II enzymes quinone reductase (QR) and glutathione S-transferase (GST). At the low dose level employed (40 μmol/kg/day), cheirolin was without effect in any tissue. Results showed that iberin was able to increase the activities of GST and QR in the forestomach, duodenum, and/or the urinary bladder of the rats, with the greatest effects being observed in the urinary bladder. However, little difference was observed in the inductive activity of iberin and its various isothiocyanate analogs [2].
Clinical trial: Up to now, Iberin is still in the preclinical development stage.
References:
[1] Jadhav U, Ezhilarasan R, Vaughn SF, Berhow MA, Mohanam S. Iberin induces cell cycle arrest and apoptosis in human neuroblastoma cells. Int J Mol Med. 2007 Mar;19(3):353-61.
[2] Munday R, Munday CM. Induction of phase II detoxification enzymes in rats by plant-derived isothiocyanates: comparison of allyl isothiocyanate with sulforaphane and related compounds. J Agric Food Chem. 2004 Apr 7;52(7):1867-71.
| Physical Appearance | A solution in ethanol. To change the solvent, simply evaporate the ethanol under a gentle stream of nitrogen and immediately add the solvent of choice. |
| Storage | Store at -20°C |
| M.Wt | 163.3 |
| Cas No. | 505-44-2 |
| Formula | C5H9NOS2 |
| Synonyms | NSC 321801 |
| Solubility | ≤11mg/ml in ethanol;16mg/ml in DMSO;16mg/ml in dimethyl formamide |
| Chemical Name | 1-isothiocyanato-3-(methylsulfinyl)-propane |
| Canonical SMILES | CS(CCCN=C=S)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure