Canertinib (CI-1033)
Canertinib (also known as CI-1033), a 3-chloro, 4-fluoro, 4-anilinoquinazoline, is an orally available, potent and irreversible Pan-erbB tyrosine kinase inhibitor that inhibits EGFR, HER2 and HER4 in vitro with the half maximal inhibition concentration IC50 of 0.8 nM, 19 nM and 7 nM respectively [1].
Canertinib irreversibly binds into the ATP pocket within the TK domain of all erbB family members, where the acrylamide side-chain at position C6 of canertinib is brought into close proximity with cysteines of erbB members, followed by the rapid formation of a covalent bond, which permanently inactivates the catalytically active erB1, erB2 and erB4 family members and effectively inhibits erbB3-dependent signaling [2].
References:
[1] Michelle Arkin, Mark M. Moasser. HER2 directed small molecule antagonists. Curr Opin Investig Drugs. Author manuscript; available in PMC 2011 February 1. Published in final edited form as: Curr Opin Investig Drugs. 2008 December; 9(12): 1264–1276.
[2] Calvo E, Tolcher AW, Hammond LA, Patnaik A, de Bono JS, Eiseman IA, Olson SC, Lenehan PF, McCreery H, Lorusso P, Rowinsky EK. Administration of CI-1033, an irreversible pan-erbB tyrosine kinase inhibitor, is feasible on a 7-day on, 7-day off schedule: a phase I pharmacokinetic and food effect study. Clin Cancer Res. 2004 Nov 1;10(21):7112-20.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 485.94 |
| Cas No. | 267243-28-7 |
| Formula | C24H25ClFN5O3 |
| Solubility | insoluble in H2O; ≥10.1 mg/mL in EtOH; ≥12.15 mg/mL in DMSO with gentle warming |
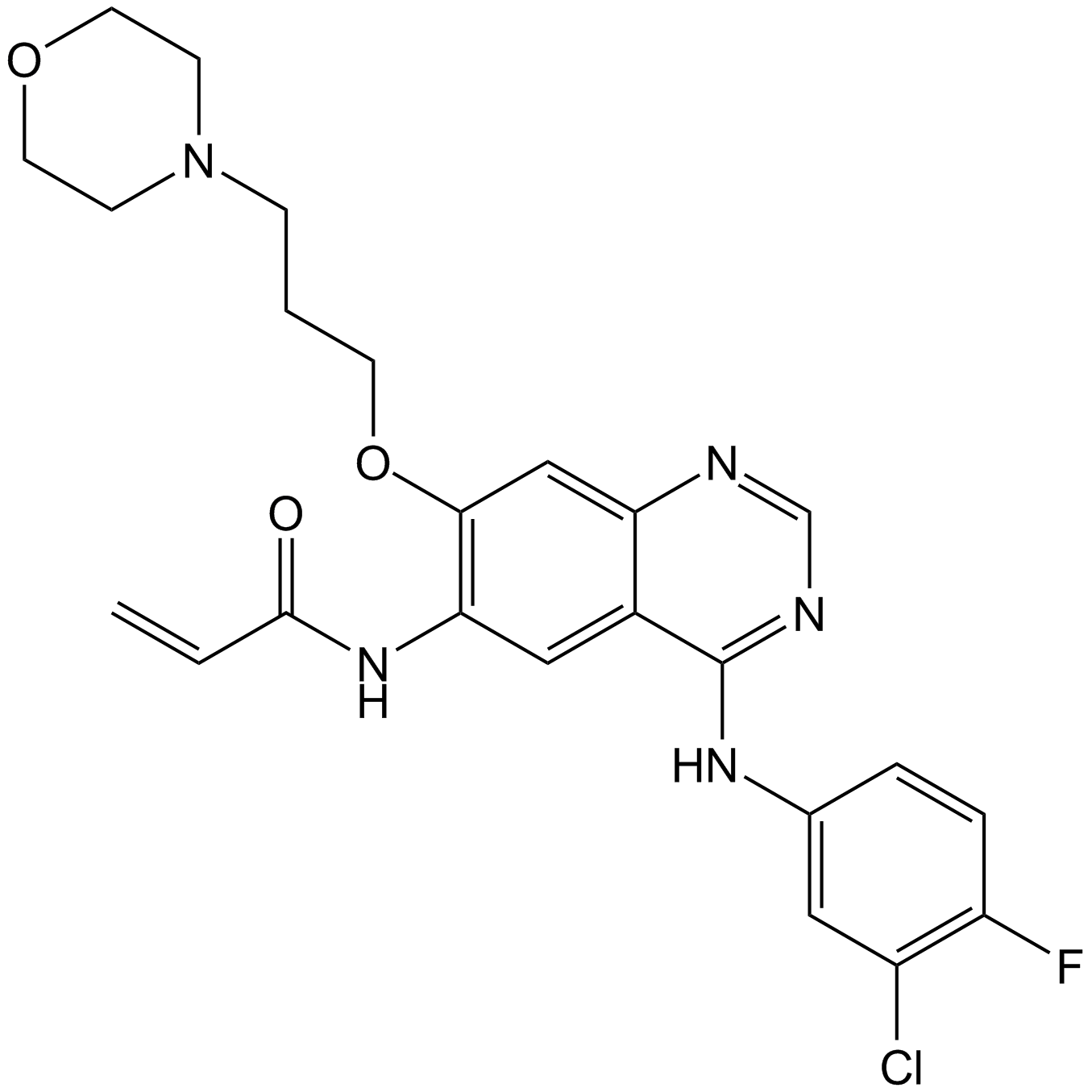
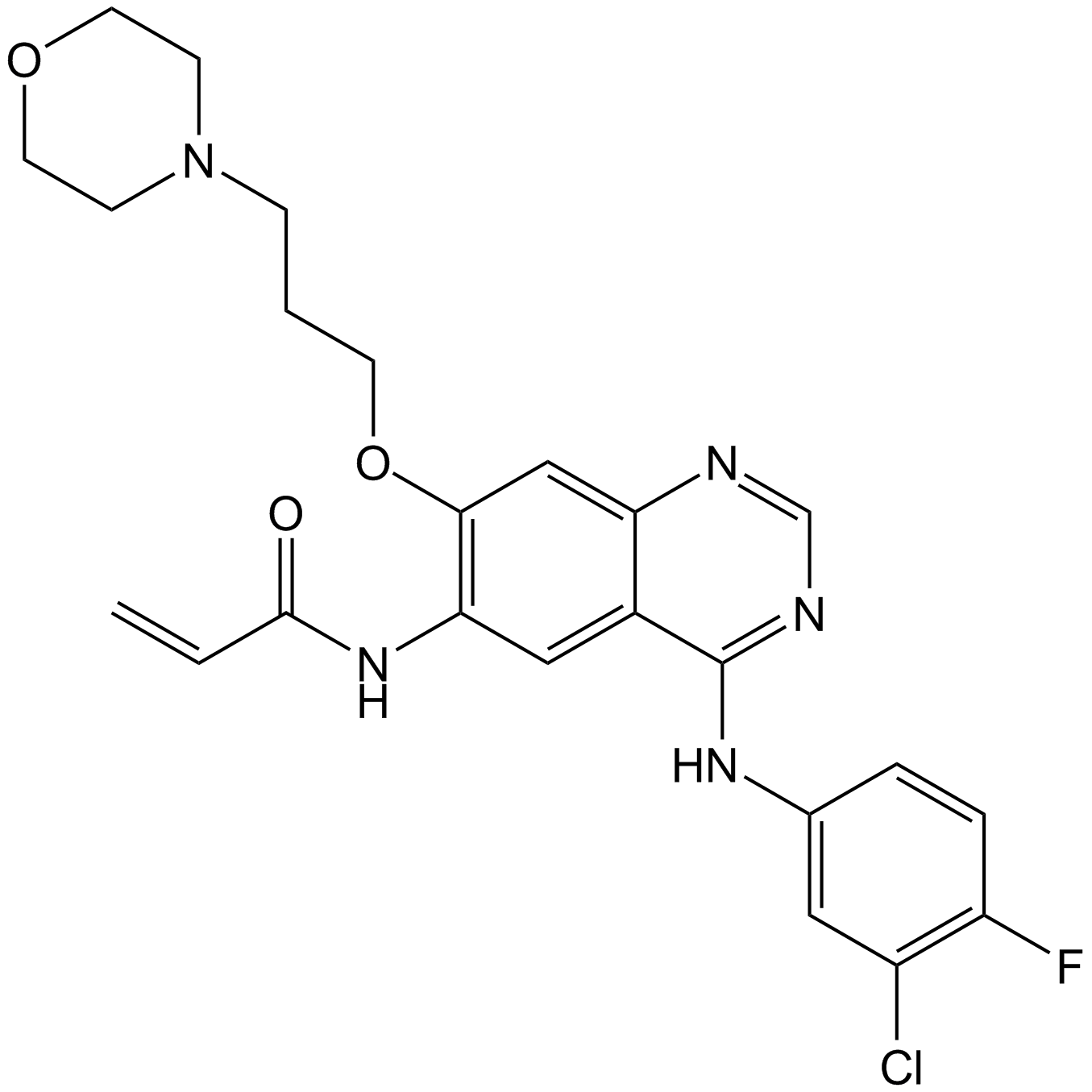
| Chemical Name | N-[4-(3-chloro-4-fluoroanilino)-7-(3-morpholin-4-ylpropoxy)quinazolin-6-yl]prop-2-enamide |
| Canonical SMILES | C=CC(Nc(c(OCCCN1CCOCC1)c1)cc2c1ncnc2Nc(cc1)cc(Cl)c1F)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Description | Canertinib is an irreversible quinazoline-based inhibitor of HER family tyrosine kinases with IC50 values of 0.8, 19 and 7 nM for EGFR, HER2 and HER4, respectively. | |||||
| Targets | EGFR | HER2 | HER4 | |||
| IC50 | 0.8 nM | 19 nM | 7 nM | |||
Quality Control & MSDS
- View current batch:
Chemical structure