Search results for: 'research area metabolism hsp'
-
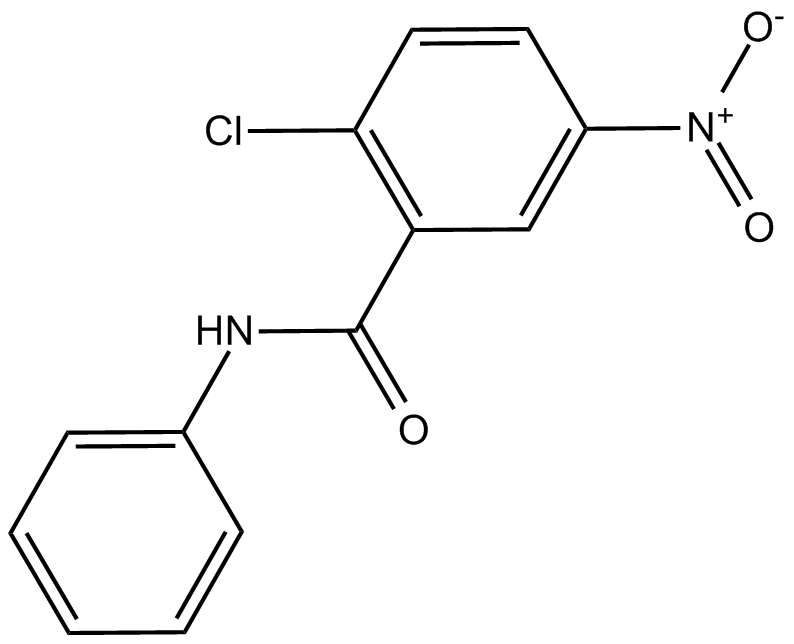 A4300 GW96627 CitationSummary: PPARγ antagonist
A4300 GW96627 CitationSummary: PPARγ antagonist -
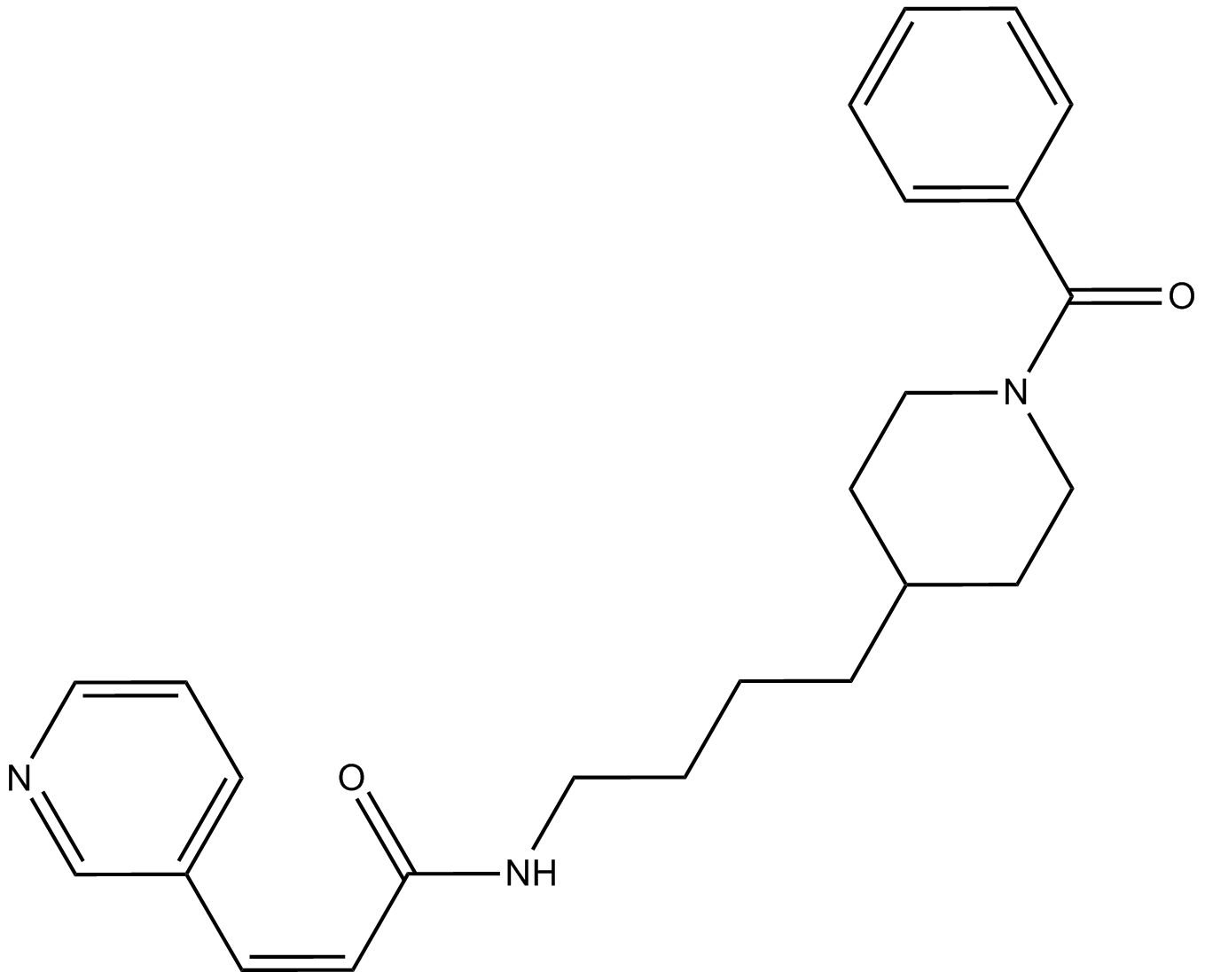 A4381 FK866 (APO866)11 CitationTarget: NamptSummary: NAMPT inhibitor, non-competitive, highly specific
A4381 FK866 (APO866)11 CitationTarget: NamptSummary: NAMPT inhibitor, non-competitive, highly specific -
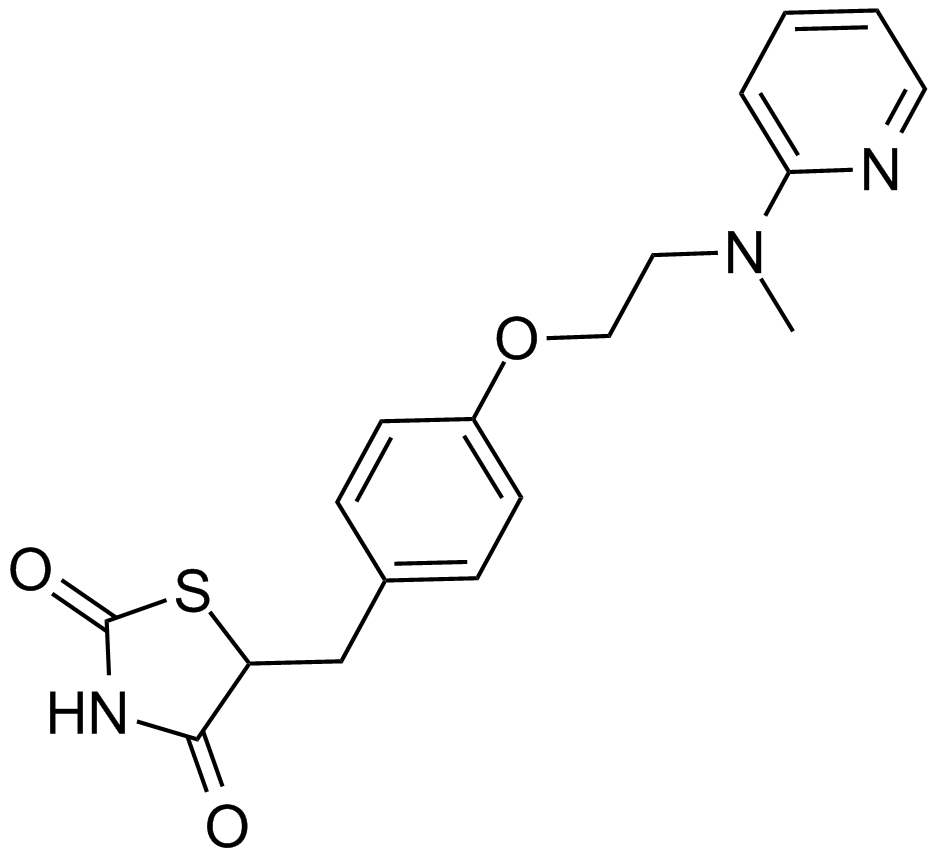 A4304 Rosiglitazone5 CitationSummary: Potent PPARγ agonist
A4304 Rosiglitazone5 CitationSummary: Potent PPARγ agonist -
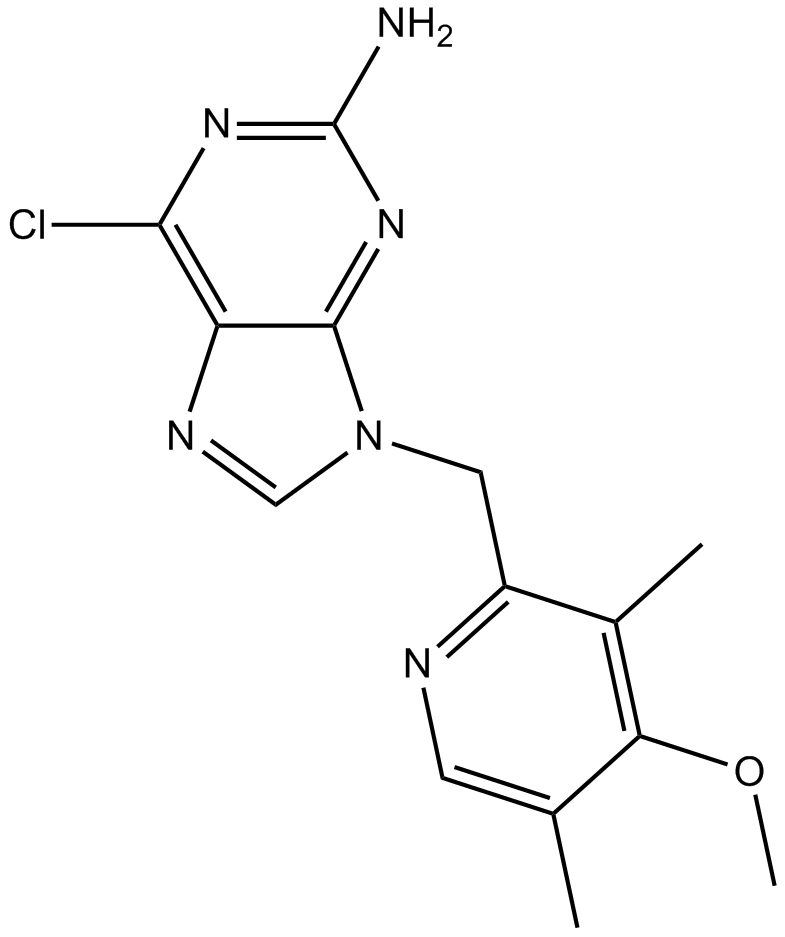 A4058 BIIB021Target: HSP90Summary: Hsp90 inhibitor,selective and competitive
A4058 BIIB021Target: HSP90Summary: Hsp90 inhibitor,selective and competitive -
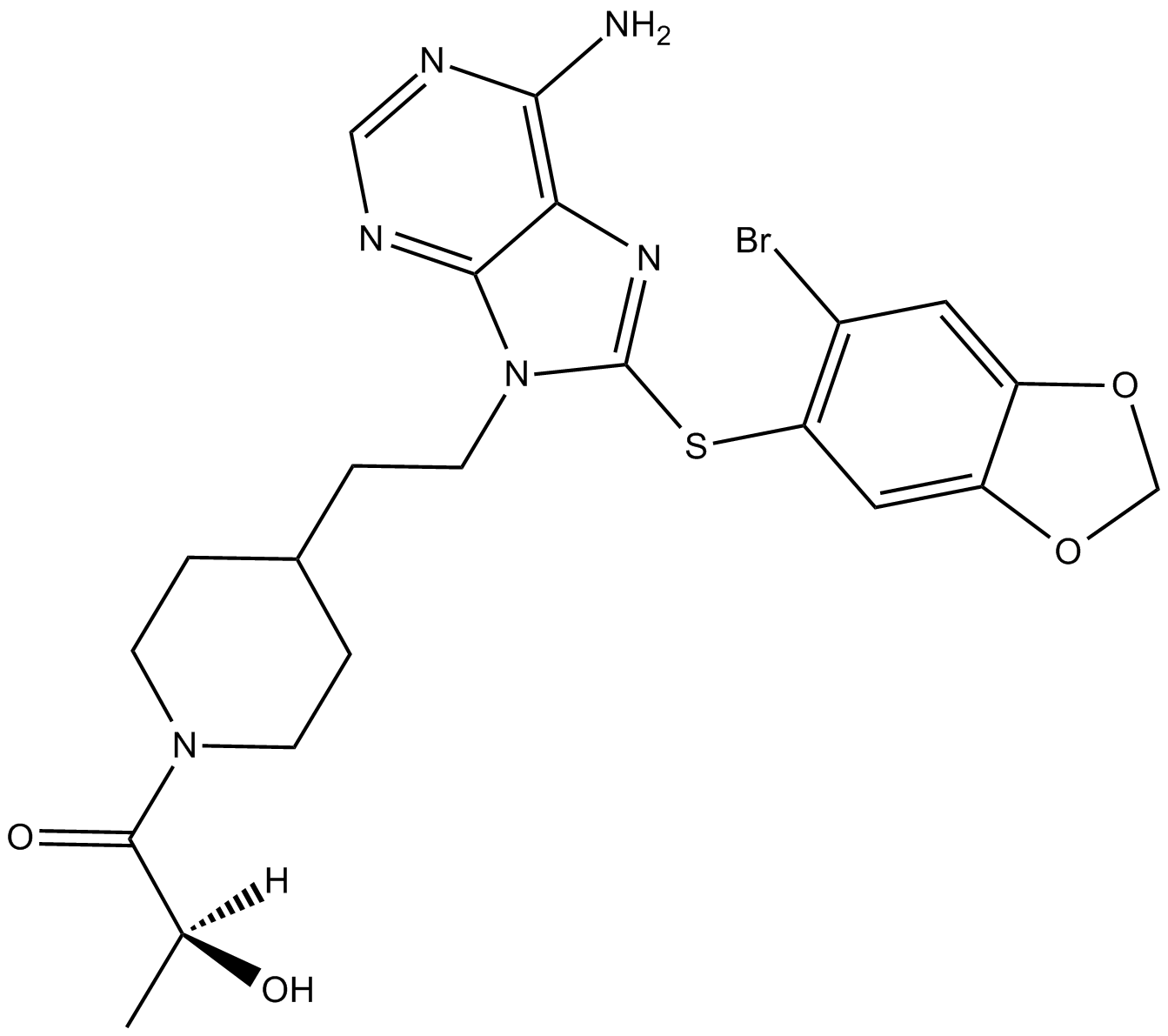 A4063 MPC-3100Summary: Hsp90 inhibitor
A4063 MPC-3100Summary: Hsp90 inhibitor -
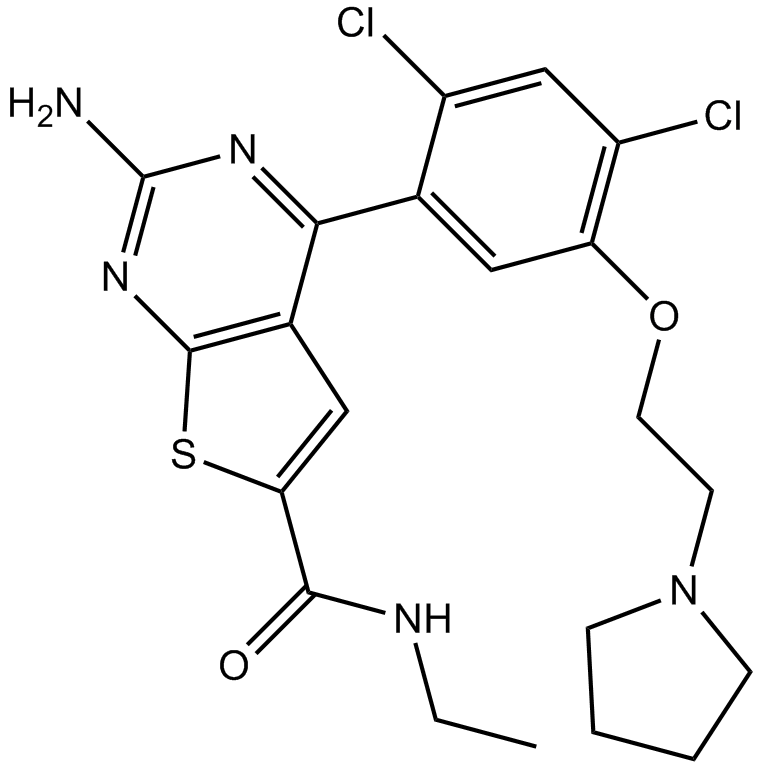 A4064 NVP-BEP800Target: HSP90Summary: Oral Hsp90β inhibitor, novel, fully synthetic
A4064 NVP-BEP800Target: HSP90Summary: Oral Hsp90β inhibitor, novel, fully synthetic -
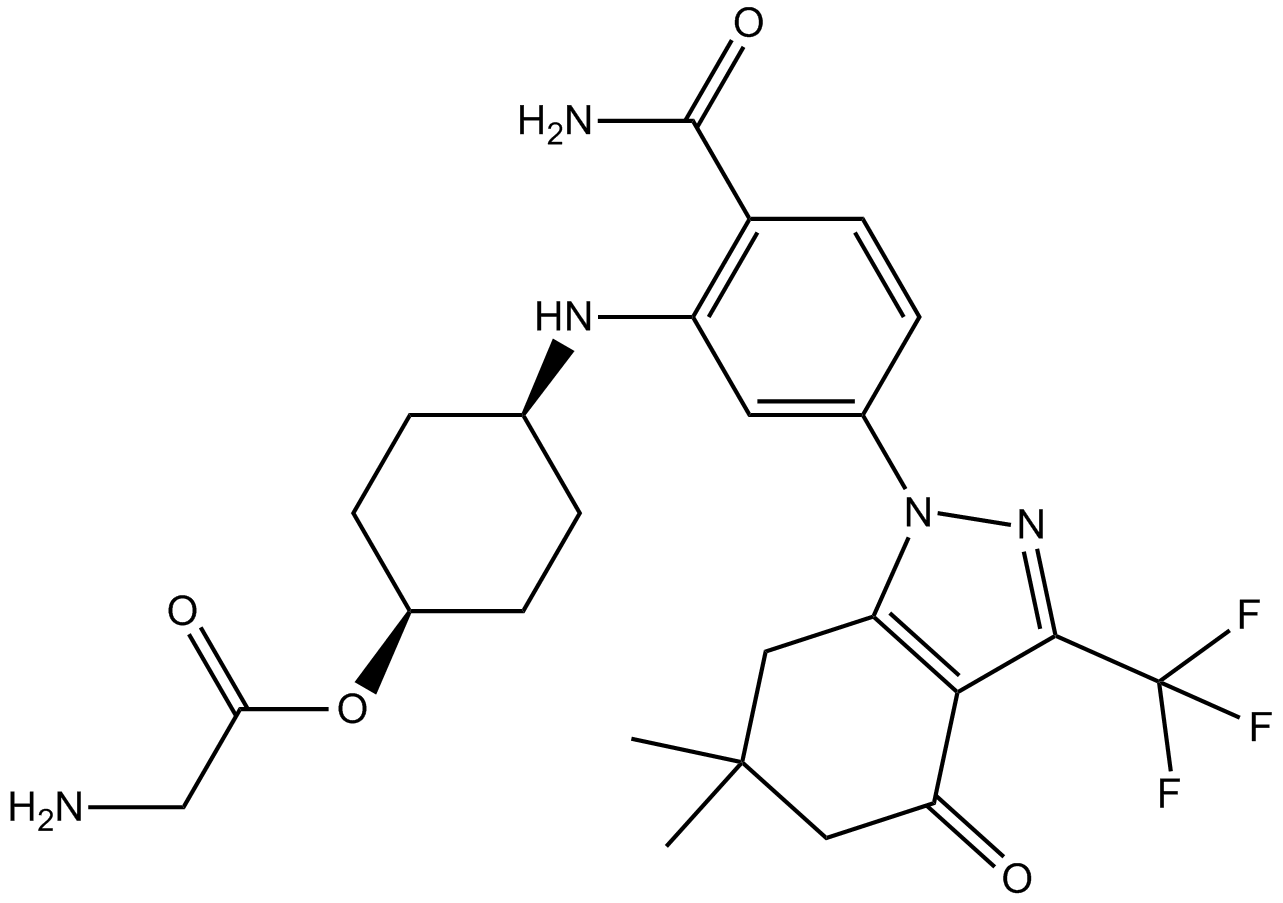 A4065 PF-04929113 (SNX-5422)Target: HSP90Summary: Hsp90 inhibitor,potent and selective
A4065 PF-04929113 (SNX-5422)Target: HSP90Summary: Hsp90 inhibitor,potent and selective -
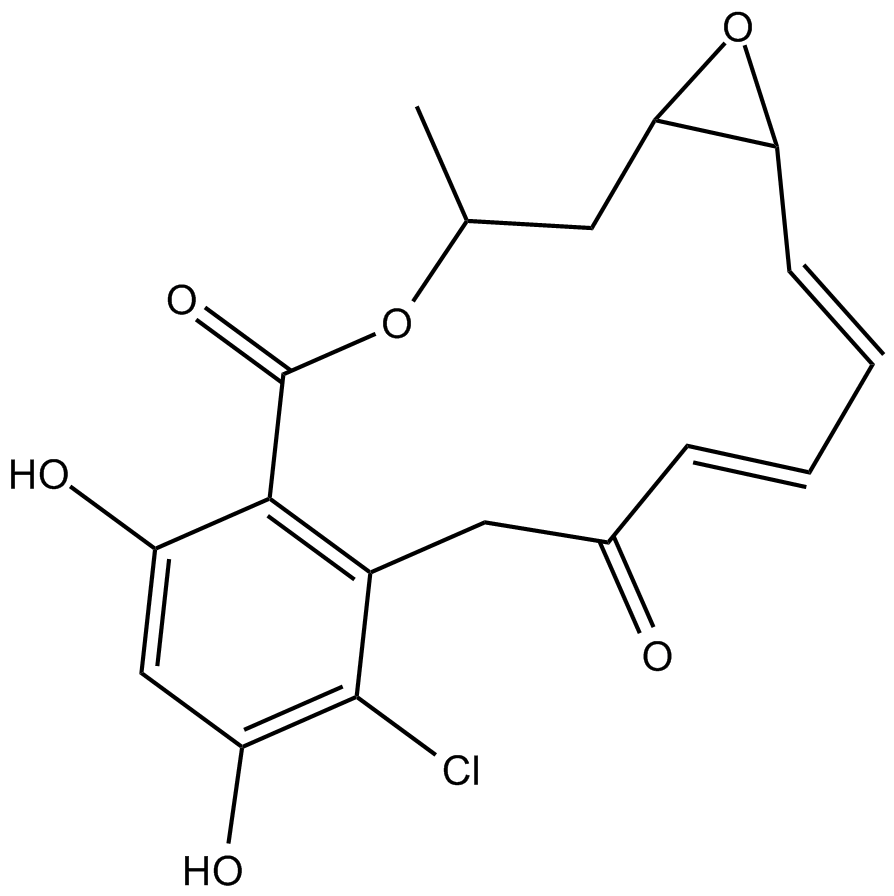 A4067 Radicicol3 CitationSummary: ATPase/kinase inhibitor
A4067 Radicicol3 CitationSummary: ATPase/kinase inhibitor -
 L1032 DiscoveryProbe™ Metabolism-related Compound Library1 CitationSummary: A unique collection of 493 metabolism-related compounds for metabolism research.
L1032 DiscoveryProbe™ Metabolism-related Compound Library1 CitationSummary: A unique collection of 493 metabolism-related compounds for metabolism research. -
 L1032P DiscoveryProbe™ Metabolism-related Compound Library PlusSummary: A unique collection of 2065 metabolism-related compounds for metabolism research.
L1032P DiscoveryProbe™ Metabolism-related Compound Library PlusSummary: A unique collection of 2065 metabolism-related compounds for metabolism research.


