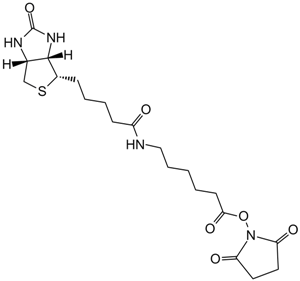
Iodoacetyl-LC-Biotin
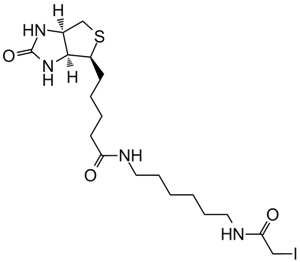
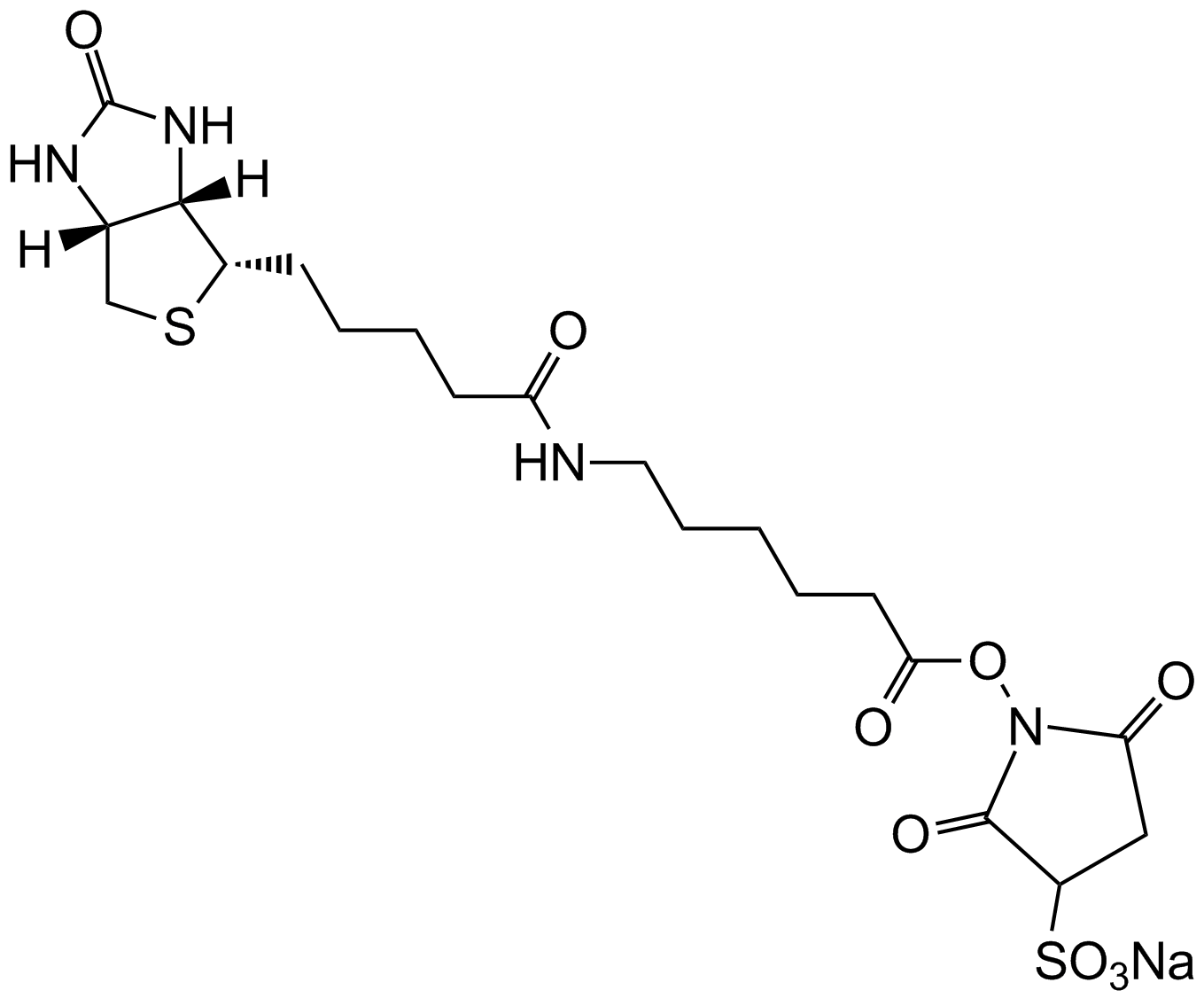
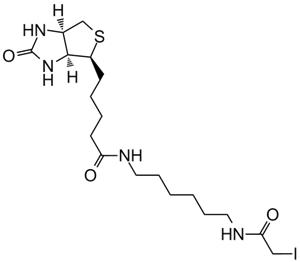
Iodoacetyl-LC-Biotin (N-iodoacetyl-N-biotinylhexylenediamine), a sulfhydryl-reactive biotinylation agent, is a water-insoluble reagent that requires the dissolution of suitable solvents, including dimethyl sulfoxide (DMSO) and dimethylformamide (DMF), prior to the addition into aqueous reactions. Iodoacetyl-LC-Biotin consists of a bicyclic biotin rings structure, a 1,6-diaminohexane spacer group attached to the valeric acid side chain of biotin and an iodoacetyl group at the end of the spacer providing sulfhydryl reactivity. The iodoacetyl group at the end of Iodoacetyl-LC-Biotin is able to react with sulfhydryl groups on proteins and other molecules forming a nonreversible bond. The long spacer arm of Iodoacetyl-LC-Biotin enables the modified molecule to better bind to the avidin or streptavidin probes.
Reference
Bioconjugate Techniques , 2nd ed. By Greg T.Hermanson (Pierce Biotechnology, Thermo Fisher Scientific, Rockford, IL). Academic Press (an imprint of Elsevier): London, Amsterdam, Burlington, San Diego . 2008. ISBN 978-0-12-370501-3.
• Protein labelingbiotinylate antibodies or other proteins for use in protein methods
• Intracellular labeling—membrane-permeable, can be used to label inside cells
• Thiol-reactive—reacts with sulfhydryls (-SH), such as the side-chain of cysteine (C)
• Iodoacetyl-activated—perform reactions in the dark at pH 7.5 to 8.5 in Tris or borate buffer
• Irreversible—forms permanent amide bonds; spacer arm cannot be cleaved
• Solubility—water insoluble, must be dissolved in DMSO or DMF before further dilution in aqueous buffers
• Medium length—spacer arm is 27.1 angstroms; contains hexylenediamine extension
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 510.43 |
| Cas No. | 93285-75-7 |
| Formula | C18H31IN4O3S |
| Synonyms | N-Biotinyl-N'-(iodoacetyl)-1,6-hexanediamine,Iaa-biotin |
| Solubility | ≥51 mg/mL in DMSO with gentle warming; insoluble in EtOH; insoluble in H2O |
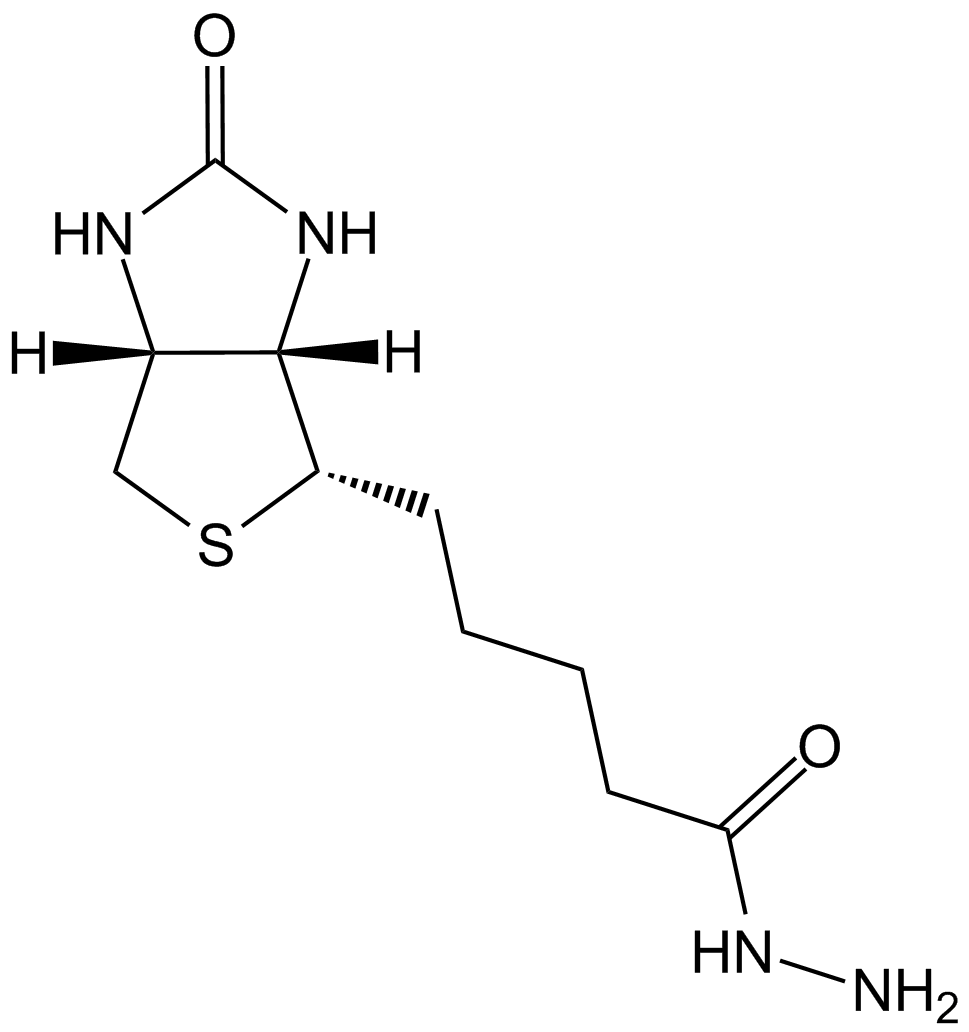
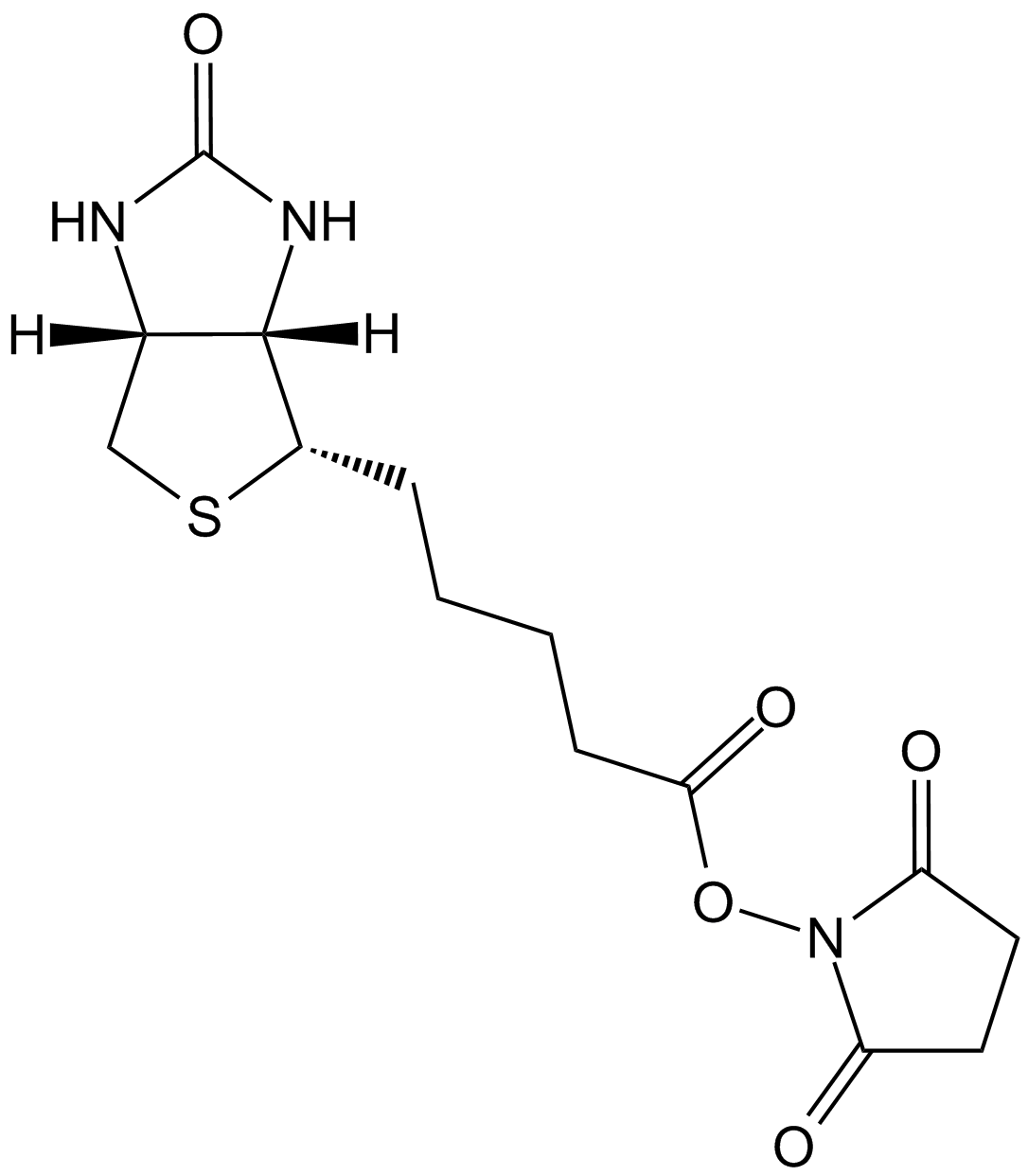
| Chemical Name | 5-[(3aS,4S,6aR)-2-oxo-1,3,3a,4,6,6a-hexahydrothieno[3,4-d]imidazol-4-yl]-N-[6-[(2-iodoacetyl)amino]hexyl]pentanamide |
| Canonical SMILES | C1C2C(C(S1)CCCCC(=O)NCCCCCCNC(=O)CI)NC(=O)N2 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Biotinylation method [1]: | |
|
Sample |
Immunoglobulin G |
|
Preparation method |
Soluble in DMSO or DMF. |
|
Reaction Conditions |
4mM, room temperature for 90min |
|
Applications |
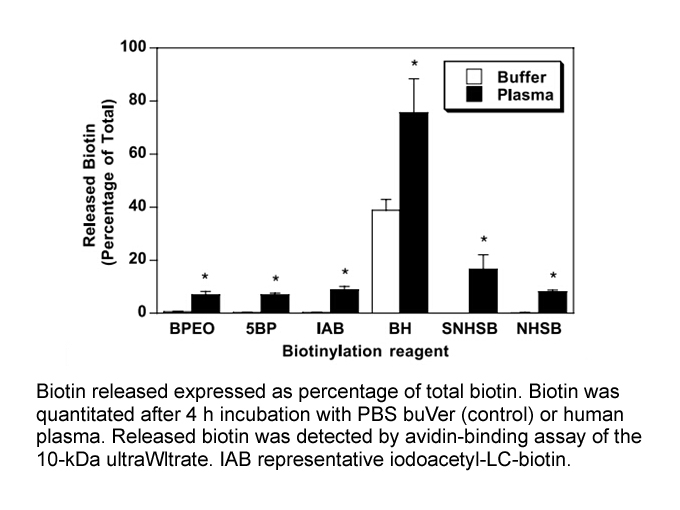
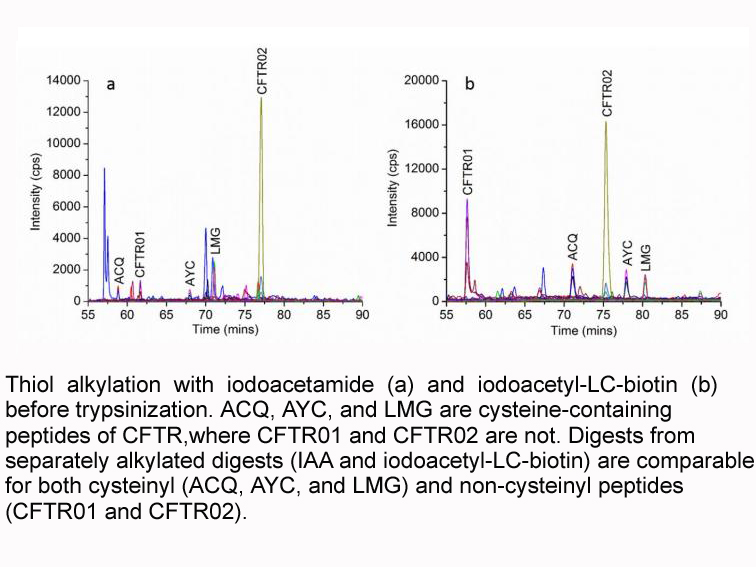
Immunoglobulin G (26.7 nmol) was dissolved in 1ml of 0.1M sodium phosphate, 5Mm EDTA buffer, pH 6. To this solution, DTT was added to produce a 50mM final concentration and incubation for 90min at 37 °C, the solution was allowed to cool to room temperature. Excess DTT was removed by molecular sieve chromatography using 50mM Tris, pH 8.3, with 5mM EDTA as the elution buffer. The IgG with reduced sulfhydryl groups was then biotinylated; 30 μl of 4mM iodoacetyl-LC-biotin in Me2SO was added and the mixture was incubated for 90min at room temperature in the dark. Finally, residual biotinylating reagent and free biotin were removed by dialysis against 0.1mM PBS, pH 7.2, for 72h. Protein concentrations were determined by BCA protein assay. Total biotin covalently bound to IgG was determined by an avidin-binding assay. |
|
References: [1]. Anna Bogusiewicz,Nell I. Mock, and Donald M. Mock. Instability of the biotin–protein bond in human plasma. Analytical Biochemistry 327 (2004) 156–161. | |
| Description | Iodoacetyl-LC-Biotin is a haloacetyl-activated sulfhydryl-reactive biotinylation reagent. | |||||
| Targets | protein cysteines | molecules containing sulfhydryl groups | ||||
| IC50 | ||||||
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data
Related Biological Data