LY2835219
LY2835219 is a selective and orally available dual cyclin-dependent kinases 4/6 (CDK4/6) inhibitor that potently inhibits the activities of CDK4 and CDK6 with the half maximal inhibition concentration IC50 values of 2 nM and 10 nM respectively [1].
LY2835219 has also been found to inhibit Rb phosphorylation both in vivo and in vitro leading to specific cell arrest at G1 phase as well as the inhibition of tumor growth [1].
Since the blood brain barrier (BBB) is a major obstacle for the effective treatment of primary brain tumors and brain metastases, LY2835219, which is able to cross the BBB, has the potential to inhibit intracranial tumor growth alone or in combination with other agents [1].
References:
[1] Concepcion Sanchez-Martinez, Lawrence M. Gelbert, Harlan Shannon, Alfonso De Dios, Brian A. Staton, Rose T. Ajamie, Geri Sawada, Graham N. Wishart and Thomas J. Raub. LY2835219, a potent oral inhibitor of the cyclin-dependent kinases 4 and 6 (CDK4/6) that crosses the blood-brain barrier and demonstrates in vivo activity against intracranial human brain tumor xenografts [abstract]. In: Proceedings of the AACR-NCI-EORTC International Conference: Molecular Targets and Cancer Therapeutics; 2011 Nov 12-16; San Francisco, CA. Philadelphia (PA): AACR; Mol Cancer Ther 2011;10(11 Suppl):Abstract nr B234.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 602.7 |
| Cas No. | 1231930-82-7 |
| Formula | C27H32F2N8·CH4O3S |
| Solubility | ≥30.15 mg/mL in DMSO; ≥55.2 mg/mL in H2O; ≥6.61 mg/mL in EtOH with ultrasonic |
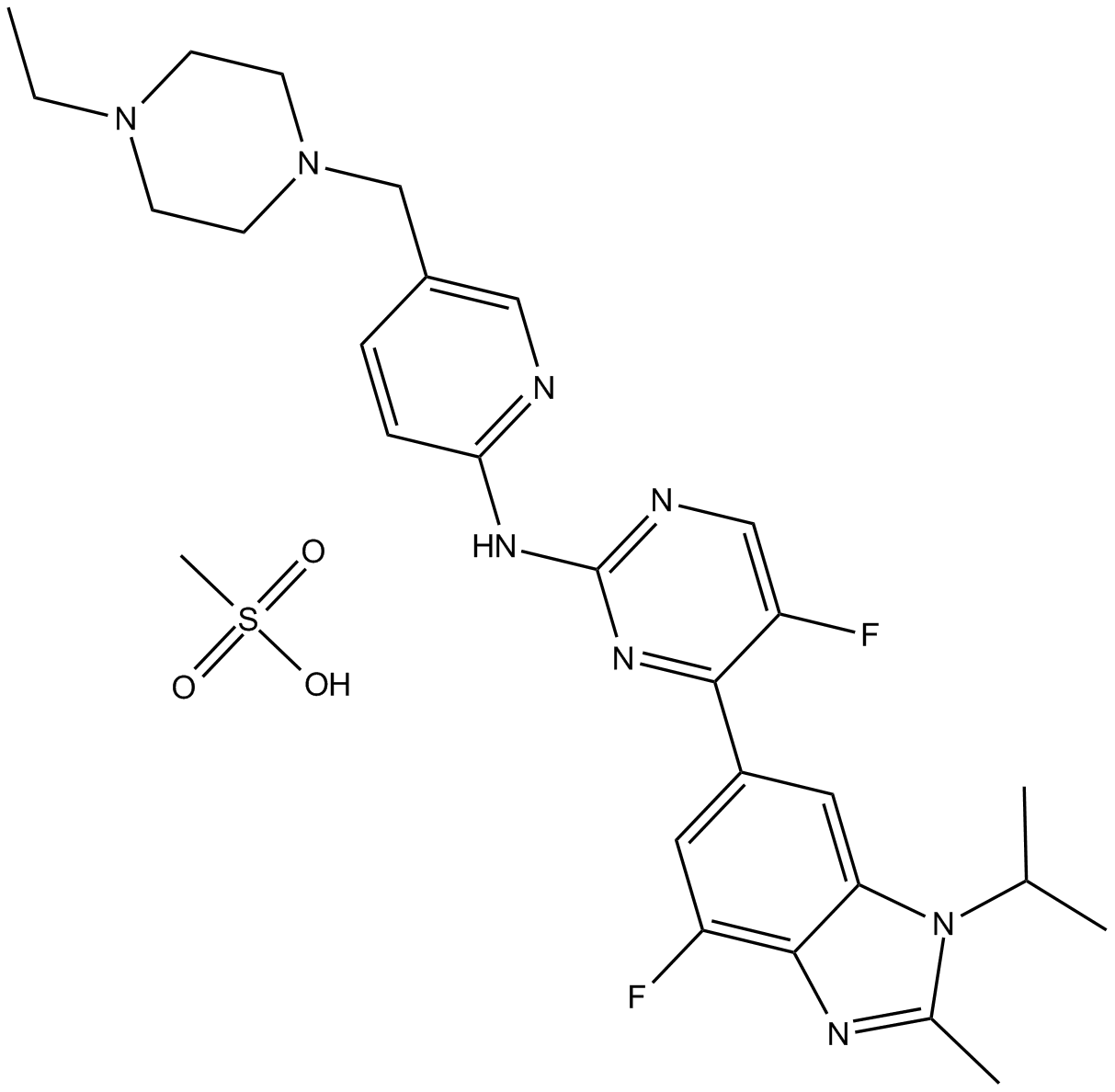
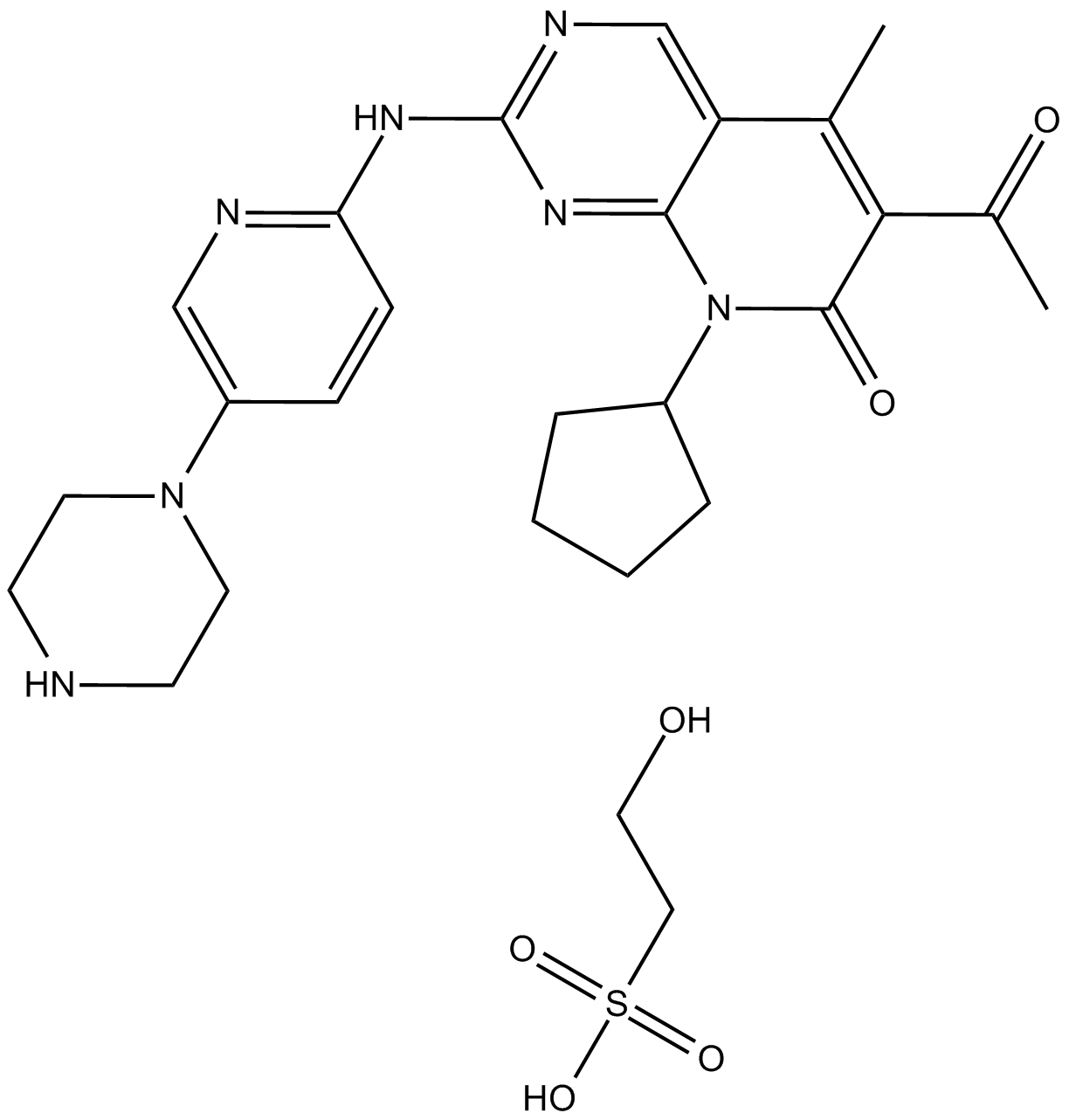
| Chemical Name | N-[5-[(4-ethylpiperazin-1-yl)methyl]pyridin-2-yl]-5-fluoro-4-(7-fluoro-2-methyl-3-propan-2-ylbenzimidazol-5-yl)pyrimidin-2-amine;methanesulfonic acid |
| SDF | Download SDF |
| Canonical SMILES | CCN1CCN(CC1)CC2=CN=C(C=C2)NC3=NC=C(C(=N3)C4=CC5=C(C(=C4)F)N=C(N5C(C)C)C)F.CS(=O)(=O)O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Kinase experiment [1]: | |
|
Binding assays |
CDK4 and CDK6 activity was determined by radiometric-filter binding assay using a c-terminus fragment of the human Rb protein (containing amino acids 773 to 928) as a substrate. Human CDK4/cyclin D1 and CDK6/cyclin D1 complexes were expressed in insect cells and purified as described. LY2835219 were serially diluted 1:3 in 20 % DMSO to create a 10-point curve at a starting concentration of 20 μM. 20 % DMSO buffer alone was employed as a control; 500 mM EDTA was used to determine the level of background in the absence of enzyme activity. A 4-parameter logistic curve fit was used to generate the IC50 values using ActivityBase software (IDBS). For kinetic analysis, a range of ATP concentrations was used and Ki for both CDK4/cyclin D1 and CDK6/cyclin D1 complexes was determined by fitting to the Michaelis-Menten equation for a competitive inhibitor using GraphPad Prism. |
| Cell experiment [1]: | |
|
Cell lines |
Colo-205 colorectal cells, MDA-MB-361 and MCF10A breast cancer cell lines, MV4-11 AML cells. |
|
Preparation method |
The solubility of this compound in DMSO is >10 mM. General tips for obtaining a higher concentration: Please warm the tube at 37 ℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reaction Conditions |
24 h |
|
Applications |
LY2835219 is a selective and orally available dual cyclin-dependent kinases 4/6 (CDK4/6) inhibitor. LY2835219 (6000 nM) inhibits Rb phosphorylation with IC50 value of 120 nM and a corresponding arrest of cells in G1 (2 N DNA content) with EC50 value of 72 nM. |
| Animal experiment [1]: | |
|
Animal models |
Mice bearing colo-205 xenograft tumors. |
|
Dosage form |
12.5 mg/kg, 25 to 100mg/kg |
|
Preparation method |
Formulated in 1 % hydroxyethyl cellulose + 0.1 % antifoam in 25 mM PB pH 2 and administered orally by gavage (final volume 0.2 mL). |
|
Applications |
LY2835219 mediates CDK4/6 inhibition, cell-cycle arrest and tumor growth inhibition (TGI) in colo-205 and inhibits Rb phosphorylation by CDK4/6. LY2835219 significantly inhibits tumor growth, doses up to 100 mg/kg are well tolerated with no loss of body weight or other signs of toxicity during or after treatment. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Gelbert LM, Cai S, Lin X, et al. Preclinical characterization of the CDK4/6 inhibitor LY2835219: in-vivo cell cycle-dependent/independent anti-tumor activities alone/in combination with gemcitabine. Invest New Drugs, 2014, 32(5): 825-837. |
|
| Description | LY2835219 is a potent and selective inhibitor of CDK4 and CDK6 with IC50 value of 2 nM and 10 nM, respectively. | |||||
| Targets | CDK4 | CDK6 | ||||
| IC50 | 2 nM | 10 nM | ||||
Quality Control & MSDS
- View current batch:
Chemical structure