
How to Request a Quote

Form
Please download and fill in the

Email
And then send the form to [email protected]

Feedback
We will contact you by email as soon as possible, please make sure your Email address is correct and check your email in time.
1. Overview
Untargeted metabolomics, or discovery metabolomics, focuses on global detection and relative quantitation of small molecules in a sample without prejudice. APExBIO uses LC-MS to collect the metabolite spectra of samples, compares the content of metabolites in different groups of samples, finds different metabolites, and explores the metabolic pathways between different metabolites. It can be used for biomarker discovery, disease diagnosis, etc. Targeted metabolomics, or quantitative metabolomics, focuses on measuring well-defined groups of metabolites, with opportunities for absolute quantitation. APExBIO uses UHPLC-QQQ-MS (Agilent 1290 UHPLC-6470 MS) to quickly and accurately determine the content of target small molecules in samples, and perform multivariate statistics and KEGG metabolic pathway enrichment analysis.
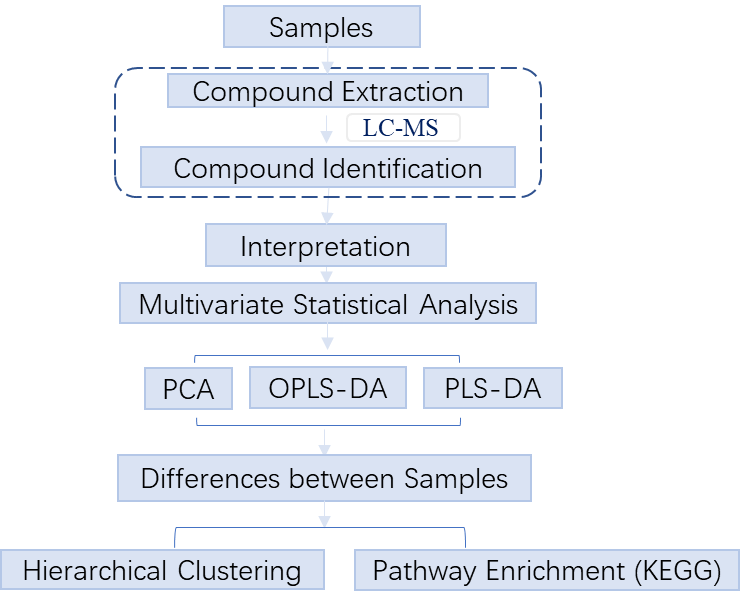
2. Workflow
Untargeted metabolomics
Targeted metabolomics
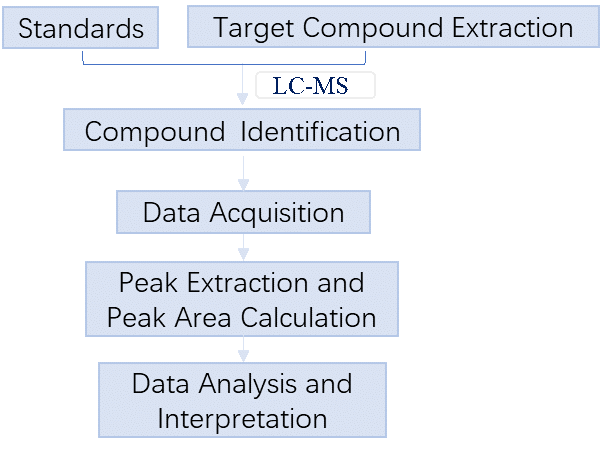
3. Applications
3.1 Identification of key taste components in loquat using widely targeted metabolomics
Abstract
Loquats can be divided into white- and yellow-fleshed cultivars. Generally, white-fleshed cultivars taste better than yellow-fleshed cultivars. Currently, metabolic causes of differences in taste are unknown, due to the lack of a large-scale and comprehensive investigation of metabolites in loquat fruit. Here, we performed a LC-MS/MSbased widely targeted metabolome analysis on two cultivars, ‘Baiyu’ (white-fleshed) and ‘ZaozhongNo. 6’ (yellow-fleshed). A total of 536 metabolites were identified, 193 of which (including 7 carbohydrates, 12 organic acids and 8 amino acids) were different between the cultivars. Pathway enrichment analysis also identified significant differences in phenolic pathways between the cultivars. Our results suggest that taste differences between the cultivars can be explained by variations in composition and abundance of carbohydrates, organic acids, amino acids, and phenolics. This study provides new insights into the underlying metabolic causes of taste variation in loquat.
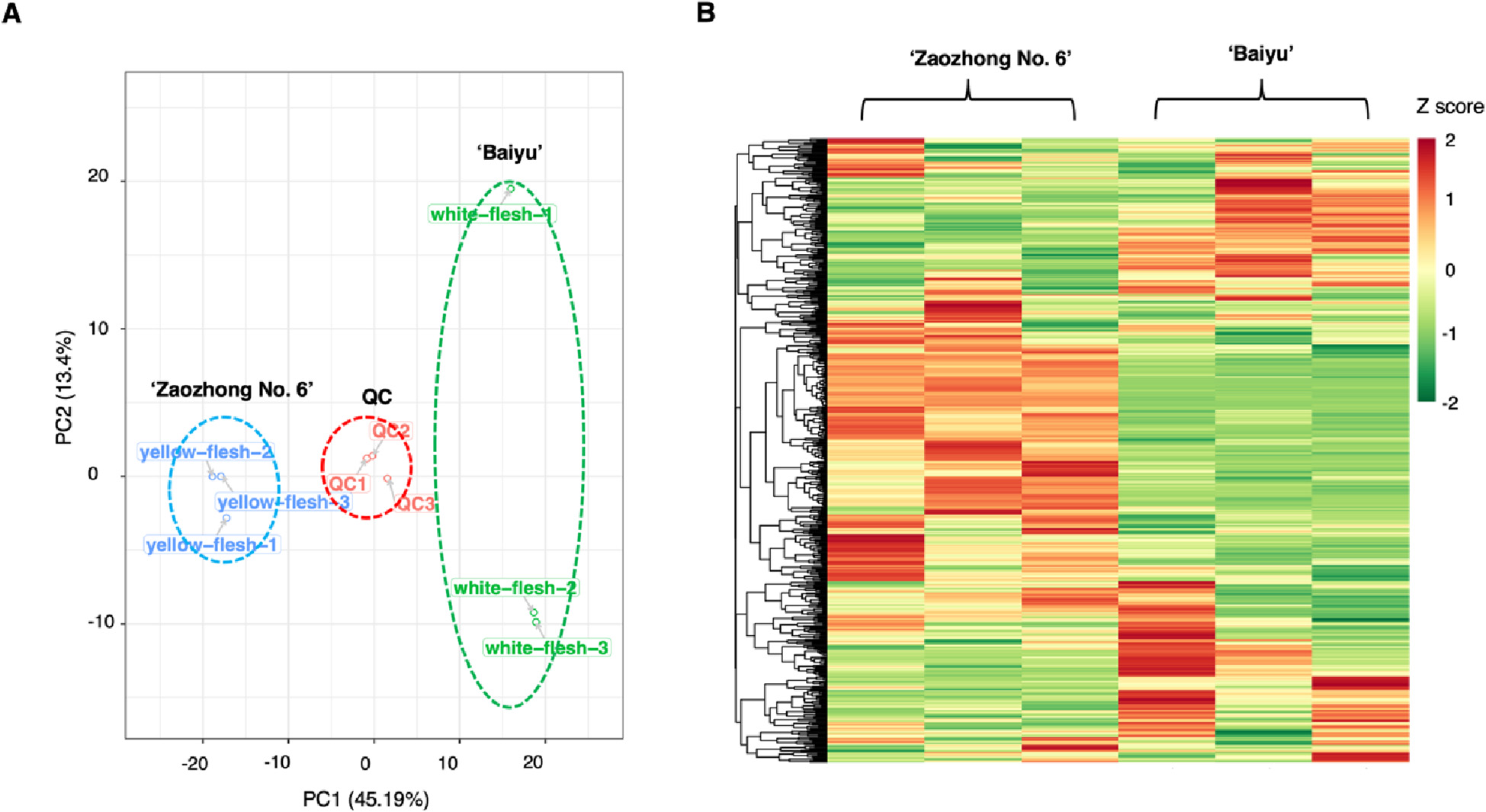
Figure 3.1.1 Differential fruit chemotype between ‘Baiyu’ and ‘Zaozhong No. 6’. (A) PCA analysis of metabolites identified from ‘Baiyu’ and ‘Zaozhong No. 6’. Equal volumes of ‘Baiyu’ and ‘Zaozhong No. 6’ fruit samples were mixed for use as a quality control (QC). (B) Cluster analysis of metabolites from samples of ‘Baiyu’ and ‘Zaozhong No. 6’. The colour indicates the level of accumulation of each metabolite, from low (green) to high (red). The Z-score represents the deviation from the mean by standard deviation units
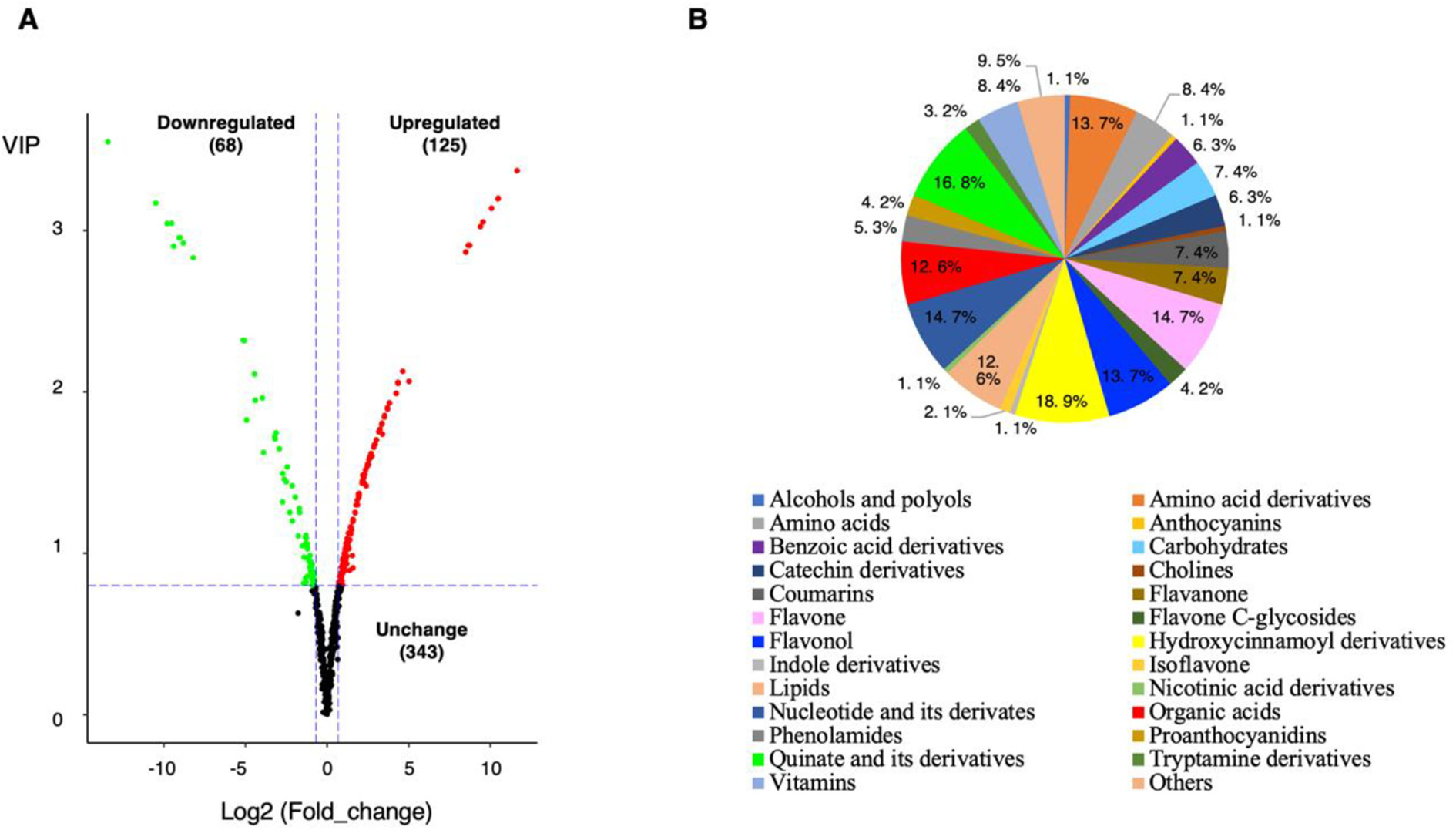
Differentially accumulating metabolites between ‘Baiyu’ and ‘Zaozhong No. 6’. (A)Volcano plot of the 536 metabolites identified. Differential metabolites were defined as metabolites with fold change ≥ 1.6 or ≤ 0.625 in ‘Zaozhong No. 6’ compared to ‘Baiyu’. A threshold of VIP ≥ 0.8 was used to separate differential metabolites from unchanged metabolites. (B) Pie chart depicting the biochemical categories of the differential metabolites identified between ‘Baiyu’ and ‘Zaozhong No. 6’.
Conclusion
In this study, we have performed successfully LC-MS/MS-based metabolic analysis to systematically compare taste differences between loquat cultivars. This work provides comprehensive information on both metabolite compositions and abundances in loquat, an important commercial fruit. The results infer that these compositional differences and concentrations of carbohydrates, organic acids, amino acids, and phenolic compounds might be the underlying causes of the differences in taste between white- and yellow-fleshed loquat.
3.2 High-throughput non-targeted metabolomics study of the effects of perfluorooctane sulfonate (PFOS) on the metabolic characteristics of A. thaliana leaves
Abstract
The ecotoxicity of perfluorooctane sulfonate (PFOS) is complex and has been reported in animals (including fish and mice), but the effects of PFOS in plants, especially the toxic mechanisms, have rarely been studied. Highthroughput nontargeted metabolomics methods for comprehensive assessment were selected to study changes in metabolic characteristics in Arabidopsis thaliana leaves by exposure to different concentrations of PFOS throughout the growth period (30 days). All the metabolites were analyzed by PCA and OPLS-DA methods, by the cutoff of VIP and p-value, 53 biomarkers were found and significantly regulated, all amino acids except glutamate were inhibited and probably associated with binding to protein, auxin and cytokinin of phytohormones were significantly down-regulated. In response mechanism to oxidative stress from PFOS, the phenylpropanoid pathway were fully activated to form several polyphenols and further enhanced into several flavonoids against the reactive oxygen species (ROS) as the primary defend pathway, in addition, ascorbate, trehalose and nicotinamide also were activated and help decrease the damage from oxidative stress. These results provide insights into the mechanism underlying the phytotoxicity of PFOS.
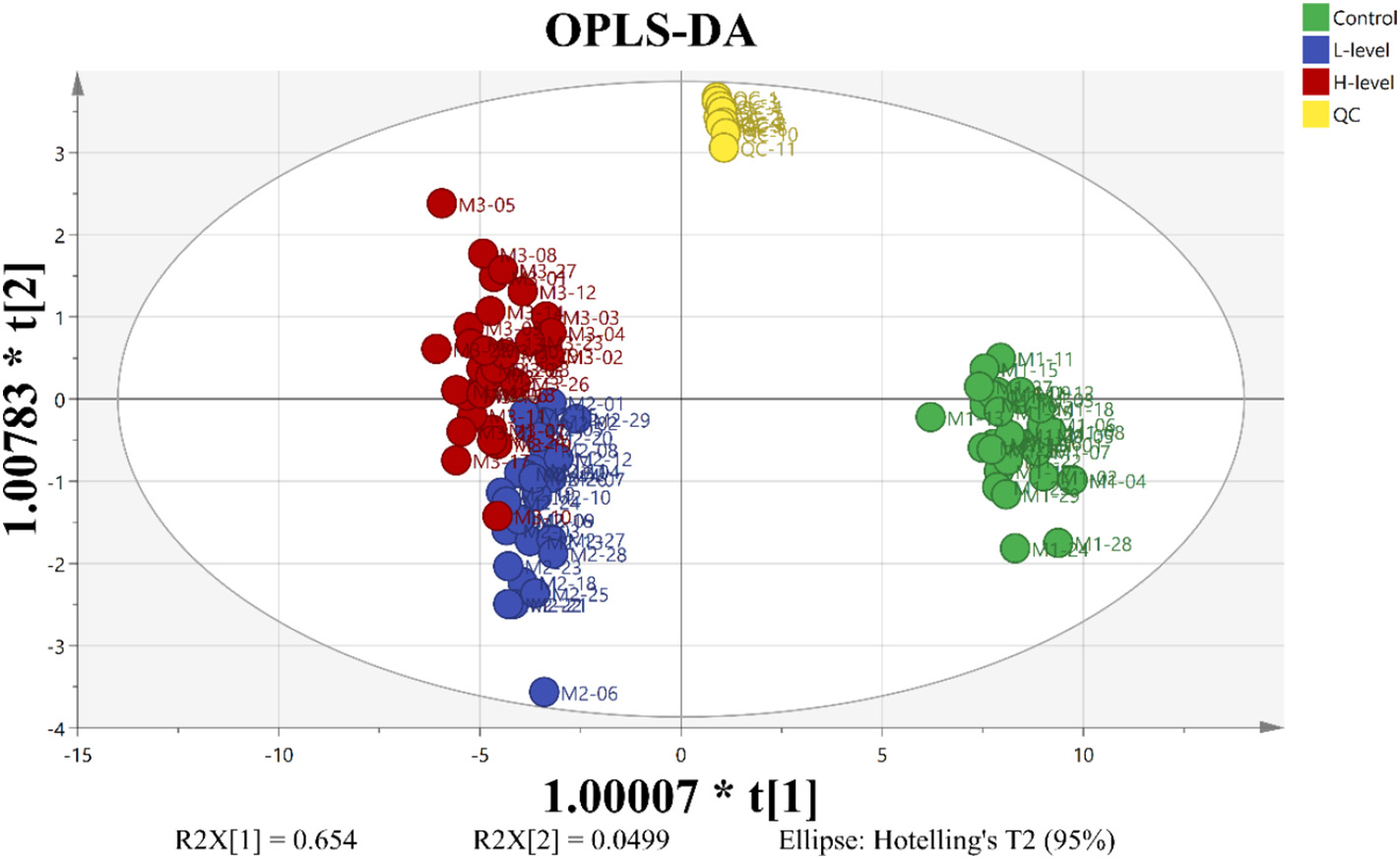
Figure 3.2.1 OPLS-DA score map based on metabolic characteristics of mature A. thaliana leaves exposed to 0.00, 0.01 and 1.00 mg/L PFOS. R2X(cum) = 0.861, R2(cum) = 0.851, Q2(cum) = 0.814 (red for 1.00 mg/L PFOS exposure group, blue for 0.01 mg/L PFOS exposure group, green for the control group of PFOS, yellow for the QC).
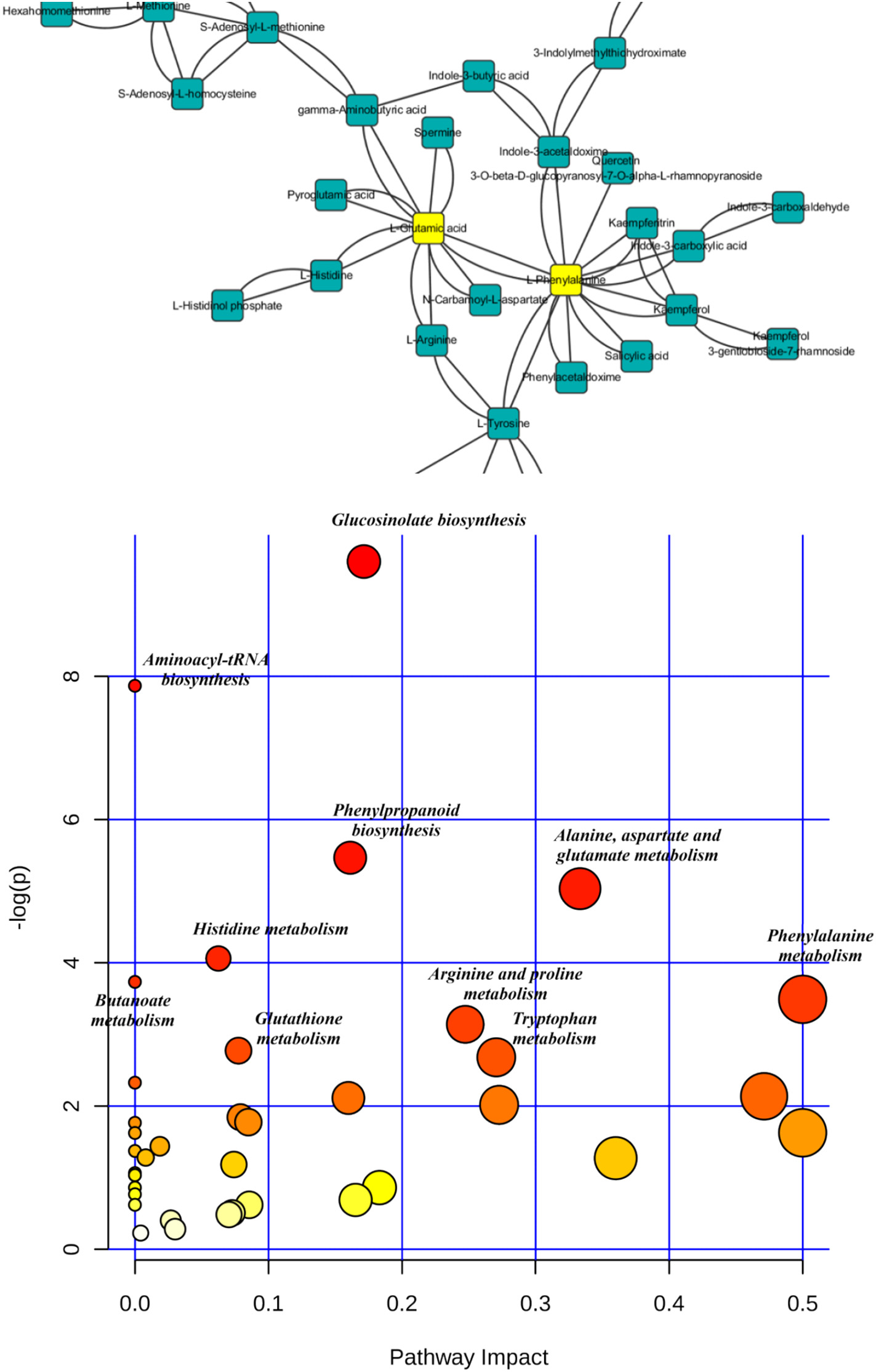
Figure 3.2.2 The most relevant pathways influenced by PFOS based on the pathway analysis (labeled metabolic pathway's p b 0.05). Metabolic pathway disruption caused by PFOS intervention was further investigated using MetaboAnalyst (https://www.metaboanalyst.ca/) based on significantly different metabolites. Pathway analysis was performed to reveal the most relevant pathways. There were 47 metabolites with identified KEGG (www.kegg.jp/) pathways that showed significant changes after PFOS intervention, and these pathways include glucosinolate biosynthesis, phenylpropanoid biosynthesis, and amino acid metabolism.
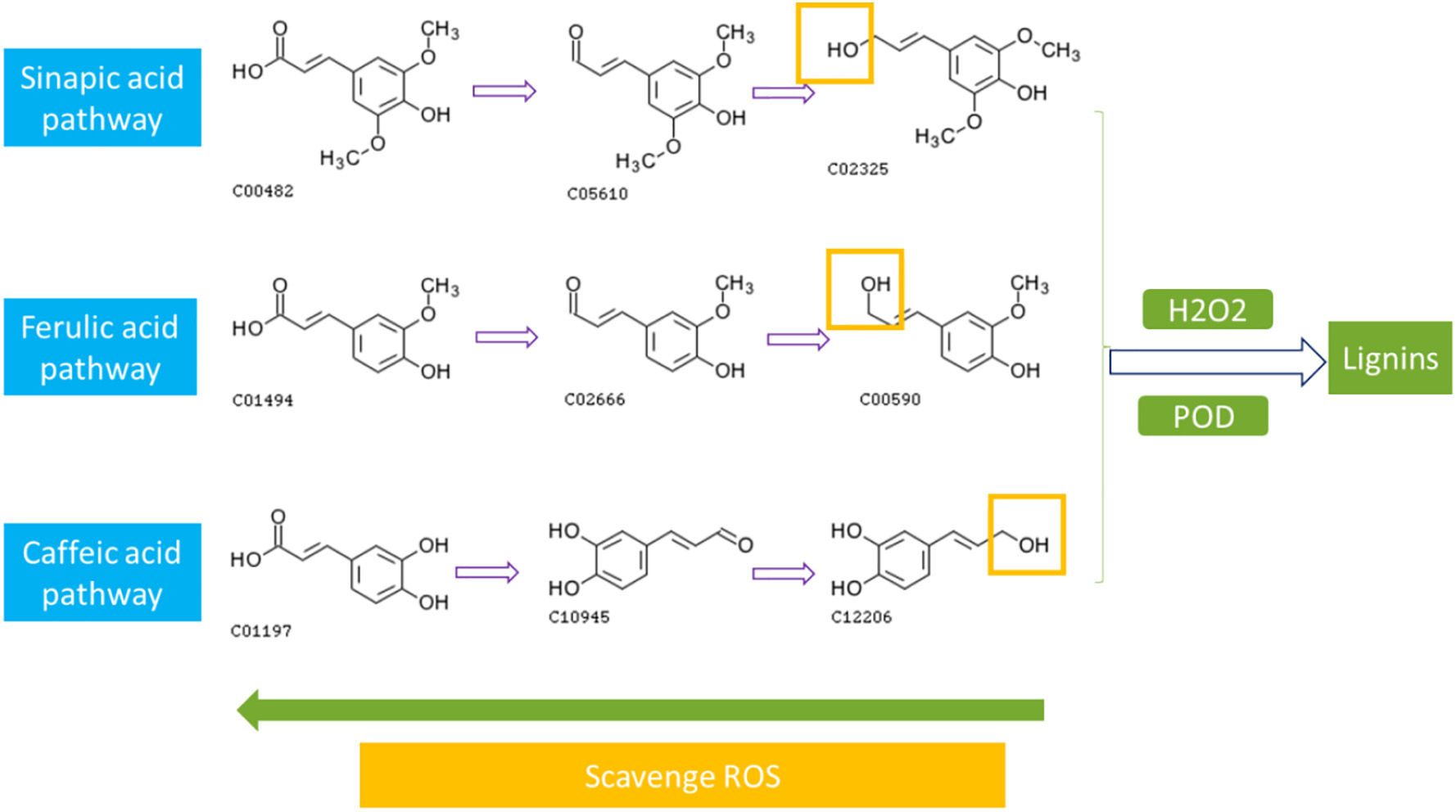
Figure 3.2.3 Activation of phenylpropanoid pathway by PFOS exposure
Conclusion
High-throughput nontargeted metabolomic methods were used to identify differential metabolites in A. thaliana leaves exposed to PFOS, and the absorption and toxicity of PFOS at different exposure concentrations in A. thaliana were studied. Fifty-three metabolites were identified as potential biomarkers of the presence of PFOS in plant leaves. The metabolic pathways of the primary and secondary metabolites are affected. The initiation of antioxidation and other defense systems can alleviate the damage triggered by oxidation, thereby stimulating the biosynthesis of defensive compounds, achieving self-protection in plants. However, the study of the oxidative stress and antioxidative mechanism were not far enough for phytotoxicity of PFOS, the interference of phytohormones and glucobrassicin were found indicating that it is necessary to further study on deep phytotoxicity mechanism of PFOS. The results of this study are based on only LC-Q-TOF and a limited range of metabolites. The use of alternative analytical techniques (GC-QTOF and NMR) may supplement comprehensive metabolite information. It is difficult to elucidate the mechanism underlying the toxicity of PFOS in A. thaliana leaves based solely on metabolite information. Multi-omics integration will be helpful to elucidate the physiological mechanisms of plants under PFOS stress.
4. References
[1] Kai Zhang, Yunsheng Han, Qingyu Zhao, Tengfei Zhan, Ying Li, Wenjuan Sun, Shuang Li, Dandan Sun, Xueyang Si, Xiaonan Yu, Yuchang Qin, Chaohua Tang, Junmin Zhang, Targeted Metabolomics Analysis Reveals that Dietary Supranutritional Selenium Regulates Sugar and Acylcarnitine Metabolism Homeostasis in Pig Liver, The Journal of Nutrition, Volume 150, Issue 4, April 2020, Pages 704–711, https://doi.org/10.1093/jn/nxz317
[2] Qiqi Guo, Zeying He, Xiaowei Liu, Bingjie Liu, Yanwei Zhang, High-throughput non-targeted metabolomics study of the effects of perfluorooctane sulfonate (PFOS) on the metabolic characteristics of A. thaliana leaves, Science of The Total Environment, Volume 710, 2020, 135542, ISSN 0048-9697, https://doi.org/10.1016/j.scitotenv.2019.135542.


