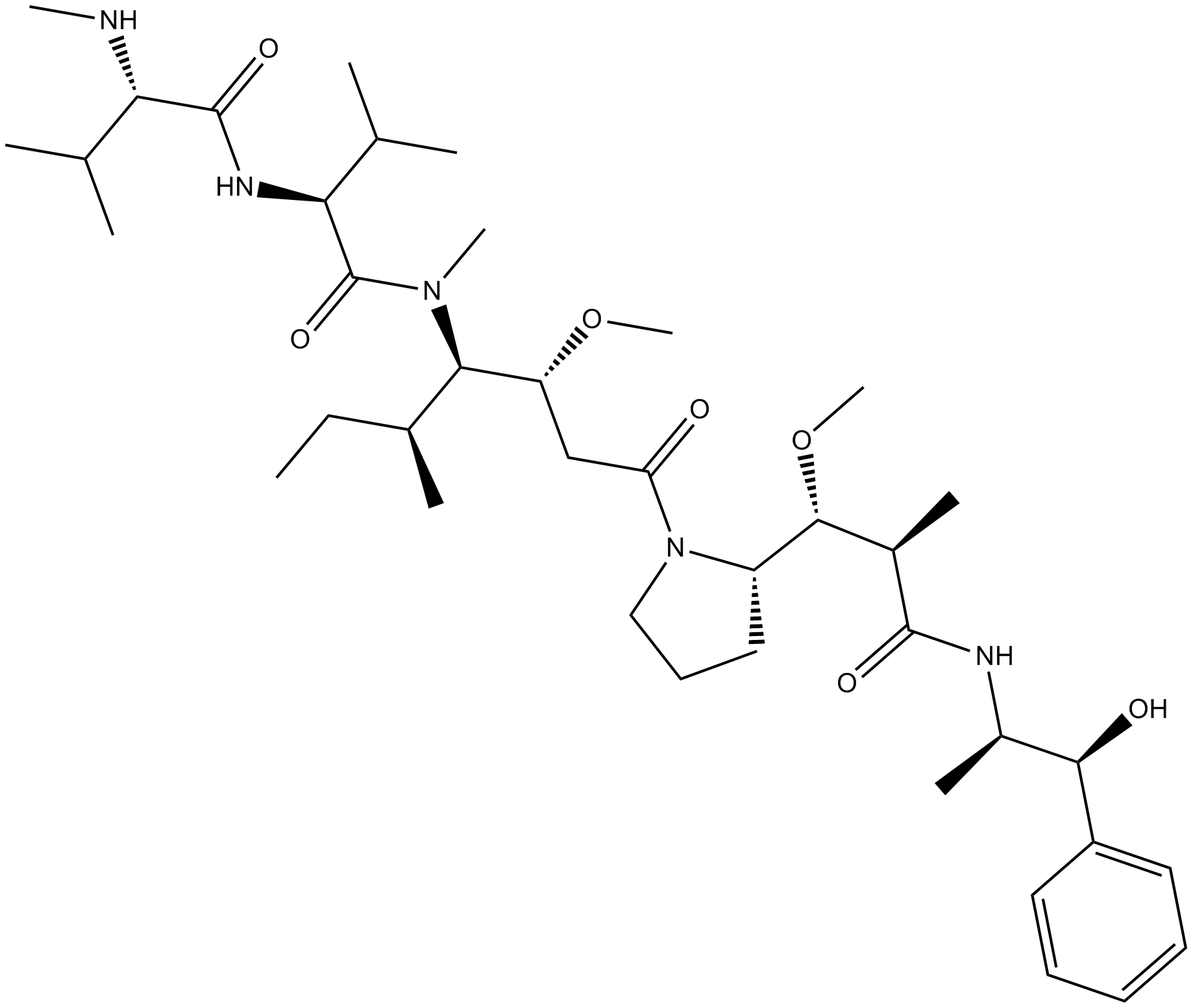
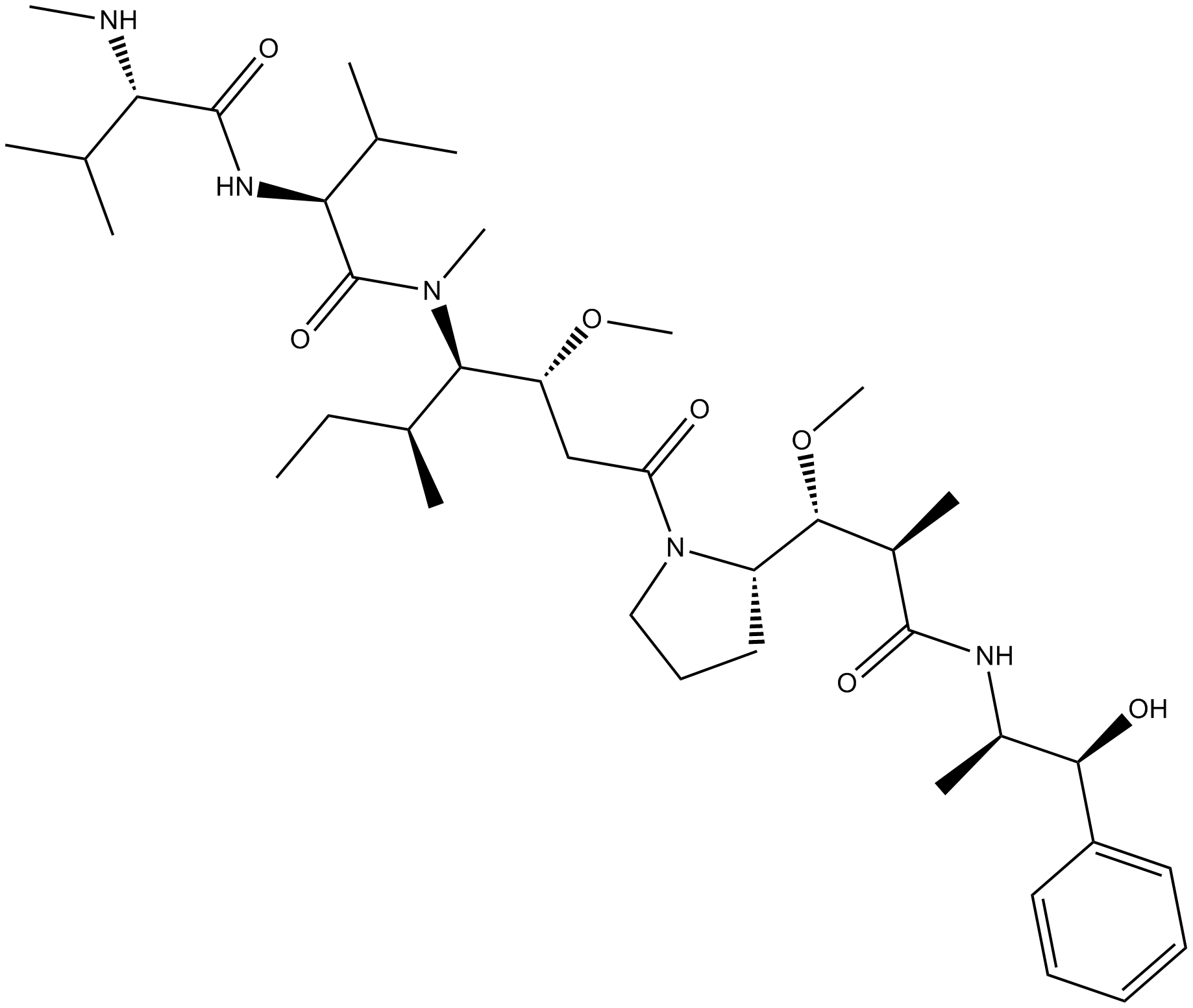
Monomethyl auristatin E
IC50: less than 1 nM for various cancer cell lines
Monomethyl auristatin E(MMAE) is a potent antimitotic agent by blocking the polymerisation of tubulin.
Microtubules play essential role in the function of the cell. Microtubules are also reported to be involved in migration, transport and reorganization and have numerous dynamic roles including movement through motor proteins such as dynein and kinesin and the separation and segregation ofchromosomes during cell division.
In vitro: The cytotoxic effects of the MMAE conjugates on H3396 cells were determined using both pulsed and long-term drug exposure assays. It was found that under both exposure conditions, high degrees of immunological specificity were obtained with the Val-Cit conjugates. cBR96-Val-Cit-MMAE was highly active at <1/100th of the concentration required for antigen saturation [1].
In vivo: In vivo therapy tests were undertaken in athymic mice with subcutaneous L2987 human lung adenocarcinoma xenografts. MMAE conjugates were administered at 3 mg mAb component/kg/dose. All of the tested MMAE conjugates were highly efficacious, leading to long-term regressions of established tumors, whereas the nonbinding control conjugates had no effect on tumor growth. In addition, there were no apparent toxicities associated with conjugate treatment [1].
Clinical trial: In a ongoing Phase I study with platinum-resistant ovarian cancer patients, nonlinear PK of MMAE conjugate had been observed in the dose range of 0.3 to 3.2 mg/kg. Circulating CA125 results suggested no impact on the PK at 2.4 mg/kg, which was the potential clinically relevant dose. Systemic free MMAE concentration was low and consistent with other MMAE containing ADCs [2].
References:
[1] Doronina SO,Toki BE,Torgov MY,Mendelsohn BA,Cerveny CG,Chace DF,DeBlanc RL,Gearing RP,Bovee TD,Siegall CB,Francisco JA,Wahl AF,Meyer DL,Senter PD. Development of potent monoclonal antibody auristatin conjugates for cancer therapy. Nat Biotechnol.2003 Jul;21(7):778-84.
[2] Jian Xu*, Priya Agarwal, Ola Saad, et al. Clinical Pharmacokinetics (PK) of Anti-MUC16 Antibody-Drug Conjugates (ADCs), DMUC5754A, in Patients with Platinum-Resistant Ovarian Cancer: Results from Phase I study. This poster was presented at World ADC Summit 2014.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 717.98 |
| Cas No. | 474645-27-7 |
| Formula | C39H67N5O7 |
| Synonyms | Vedotin; MMAE |
| Solubility | ≥35.9 mg/mL in DMSO; insoluble in H2O; ≥48.5 mg/mL in EtOH with gentle warming and ultrasonic |
| Chemical Name | (2S)-N-[(2S)-1-[[(3R,4S,5S)-1-[(2S)-2-[(1R,2R)-3-[[(1S,2R)-1-hydroxy-1-phenylpropan-2-yl]amino]-1-methoxy-2-methyl-3-oxopropyl]pyrrolidin-1-yl]-3-methoxy-5-methyl-1-oxoheptan-4-yl]-methylamino]-3-methyl-1-oxobutan-2-yl]-3-methyl-2-(methylamino)butanamide |
| Canonical SMILES | CCC(C)C(C(CC(=O)N1CCCC1C(C(C)C(=O)NC(C)C(C2=CC=CC=C2)O)OC)OC)N(C)C(=O)C(C(C)C)NC(=O)C(C(C)C)NC |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1]: | |
|
Cell lines |
Karpas 299, H3396 and RCA cell lines |
|
Preparation method |
The solubility of this compound in DMSO is >35.9 mg/mL. General tips for obtaining a higher concentration: Please warm the tube at 37℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reacting condition |
0-10000 ng/ml for 92 h |
|
Applications |
The cytotoxic activities was obtained on RCA colorectal carcinoma cells that were treated with the mAb-Val-Cit-MMAE conjugates for 96 h. Result showed that there was as much as a 104-fold reduction in cell viability in cBR96-Val-Cit-MMAE-treated cells. It was also found that, in all cases, the conjugates were potent, and the effects were due to specific drug delivery, because unconjugated, non-cross-linked mAbs had little to no cytotoxic activities. |
| Animal experiment [1]: | |
|
Animal models |
CB17 SCID mouse xenograft model with Karpas 299 and L2987 solid tumors |
|
Dosage form |
3 mg mAb component/kg/injection, i.v. for L2987 human lung adenocarcinoma tumors; 1 mg mAb component/kg/injection, i.v. for Karpas 299 human ALCL tumors |
|
Application |
An experiment was undertaken in SCID mice with subcutaneous Karpas 299 ALCL tumors (cBR96 Ag–, cAC10Ag+), in which the cAC10-Val-Cit-MMAE was now the binding conjugate, whereas cBR96-Val-Cit-MMAE was the nonbinding control. The therapeutic effects of cAC10-Val-Cit-MMAE were pronounced. Cures of relatively large tumors (>200 mm3) were obtained at 1 mg mAb component/kg/injection (0.035 mg MMAE component/kg/injection), corresponding to 1/30th of the MTD. Equivalent doses of the nonbinding control conjugate, cBR96-Val-Cit-MMAE, were ineffective. Treatment with cAC10-Val-Cit-MMAE at 1 mg mAb component/kg/injection and at 0.5 mg/kg/injection resulted in 100% and 80% tumor cures, respectively. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1] Doronina SO,Toki BE,Torgov MY,μMendelsohn BA,Cerveny CG,Chace DF,DeBlanc RL,Gearing RP,Bovee TD,Siegall CB,Francisco JA,Wahl AF,μMeyer DL,Senter PD. Development of potent monoclonal antibody auristatin conjugates for cancer therapy. Nat Biotechnol.2003 Jul;21(7):778-84. | |
Quality Control & MSDS
- View current batch:
Chemical structure