Colchicine
Colchicine is a tubulin inhibitor that blocks microtubule polymerization by binding to tubulin (IC50 = 3.2 μM).
Tubulin is a member of a small family of globular proteins. The tubulin superfamily includes five distinct families. α- and β-tubulin dimers bind GTP and, in the GTP-bound state, assemble at the (+) end of microtubules, thereby forming microtubules [1]. The β-tubulin subunit is exposed at the plus end of the microtubule, and the α-tubulin subunit is exposed at the minus end. After the dimer is incorporated into the microtubule, the GTP molecule bound to the β-tubulin subunit is ultimately hydrolyzed to GDP through inter-dimer contacts along the microtubule protofilament [2]. This is the GTP cycle that is essential for the dynamic instability of microtubules.
Colchicine inhibits microtubule polymerization by binding to tubulin, one of the main components of microtubules. Tubulin function is critical for mitosis; therefore, colchicine acts as a “mitotic inhibitor” or spindle poison [3]. Mitosis is blocked at prometaphase of the cell cycle (especially metaphase) before completion. In addition to inhibiting mitosis, colchicine also inhibits neutrophil movement and activity; in a mouse model of gouty arthritis, colchicine at a dose of 5 μmol/kg shows net anti-inflammatory activity; colchicine can also inhibit urate deposition, a key aspect of gout treatment [4]. The main side effects are gastrointestinal discomfort and neutropenia. High doses can also damage the bone marrow, leading to anemia, and can cause hair loss.
References:
1.? Heald R, Nogales E. "Microtubule dynamics". J. Cell. Sci. 2002,115 (Pt 1): 3–4.
2.? Howard J, Hyman A. "Dynamics and mechanics of the microtubule plus end". Nature 2003,422 (6933): 753–8.
3.? "Information for Healthcare Professionals: New Safety Information for Colchicine (marketed as Colcrys)". U.S. Food and Drug Administration.
4.? Chen LX, Schumacher HR. "Gout: an evidence-based review". J Clin Rheumatol 2008, 14: S55–62.
5.? Colchicine. National Institute for Occupational Safety and Health. Emergency Response Safety and Health Database, August 22, 2008. Retrieved December 23, 2008.
- 1. Yi Liu, Chao Bian, et al. "Reference genome provide insights into sex determination of silver aworana (Osteoglossum bicirrhosum)." BMC Biol. 2025 Jan 29;23(1):29. PMID: 39875888
- 2. Jundong Lin, Xiaoxia Cai, et al. "Fluorescent Nanoparticles Achieve Efficient Photothermal Conversion and Enhanced Antitumor Efficacy Through Intermolecular Aggregation-Caused Quenching" Aggregate. 19 December 2024
- 3. Yi Wang, Jiakun Wang, et al. "M1 macrophage-membrane-cloaked paclitaxel/β-elemene nanoparticles targeting cervical cancer for enhanced therapy" International Journal of Pharmaceutics: X Volume 8, December 2024, 100276
- 4. Falong Lu, et al. "Reprogramming mechanism dissection and trophoblast replacement application in monkey somatic cell nuclear transfer." Nat Commun. 2024 Jan 16;15(1):5. PMID: 38228612
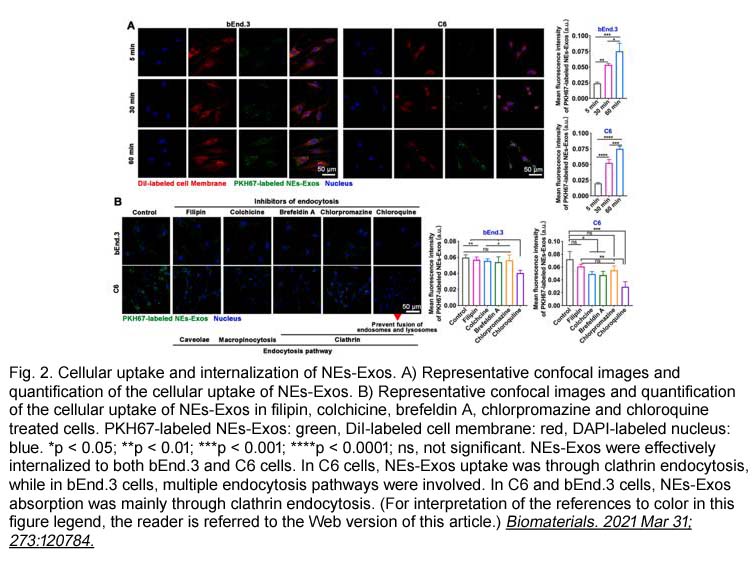
- 5. Jun Wang, Wei Tang, et al. "Inflammatory tumor microenvironment responsive neutrophil exosomes-based drug delivery system for targeted glioma therapy." Biomaterials. 2021 Jun;273:120784. PMID: 33848731
| Physical Appearance | A solid |
| Storage | -20°C |
| M.Wt | 399.44 |
| Cas No. | 64-86-8 |
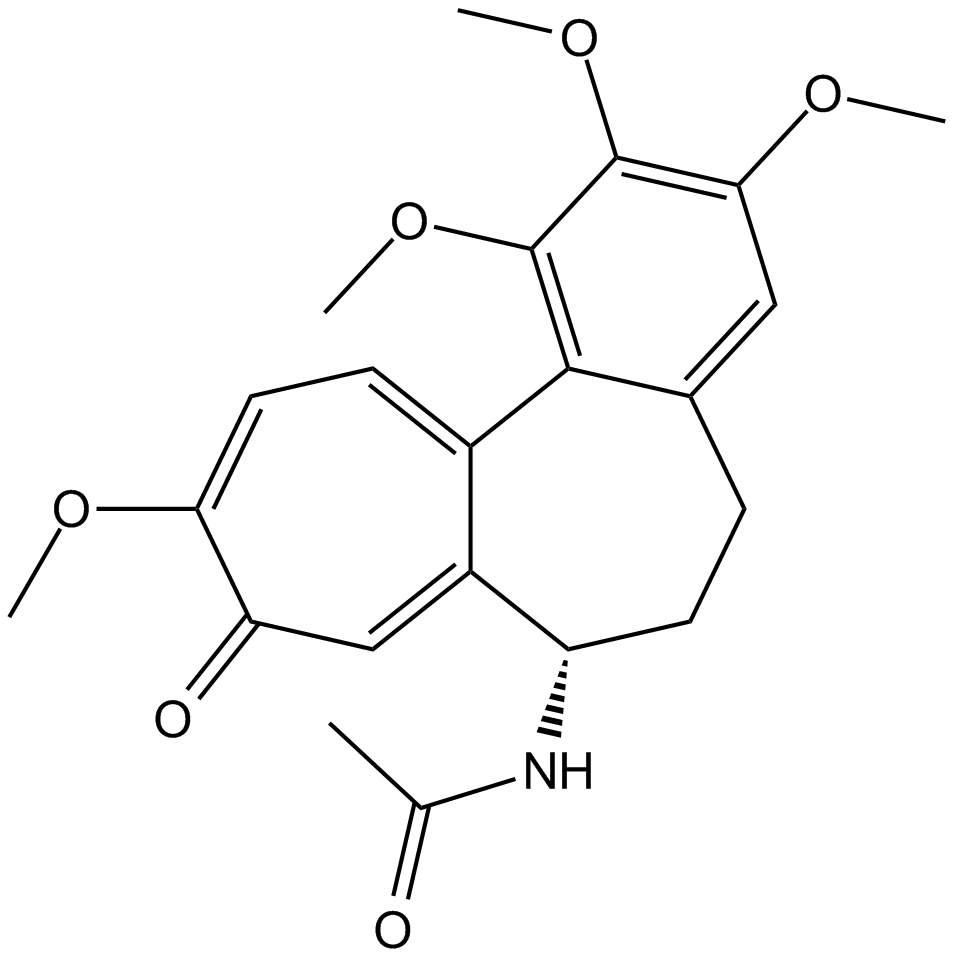
| Formula | C22H25NO6 |
| Solubility | ≥19.97 mg/mL in DMSO; ≥45.5 mg/mL in H2O with gentle warming; ≥50.8 mg/mL in EtOH with gentle warming |
| Chemical Name | N-[(7S)-1,2,3,10-tetramethoxy-9-oxo-6,7-dihydro-5H-benzo[a]heptalen-7-yl]acetamide |
| Canonical SMILES | O(C)C1=C2C=3C([C@@H](NC(C)=O)CCC2=CC(OC)=C1OC)=CC(=O)C(OC)=CC3 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment:[1] | |
|
Cell lines |
Rat cerebellar granule cells (CGCs) |
|
Reaction Conditions |
1 μM colchicine for 12 ~ 24 h incubation |
|
Applications |
Exposure to 1 μM colchicine, a microtubule disrupting agent, triggered apoptosis in rat CGCs. Apoptotic nuclei began to appear after 12 h followed by oligonucleosomal DNA laddering, whereas inhibition of the mitochondrial 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide metabolism became significant between 18 and 24 h, when most cells already had apoptotic nuclei. |
| Animal experiment:[2] | |
|
Animal models |
Male C57BL/J6 mice, aged 8 ~ 12 weeks |
|
Dosage form |
5 μmol/kg Administered orally |
|
Applications |
Oral colchicine inhibited monosodium urate (MSU)-induced superoxide production by neutrophils in vivo at doses 100 times lower than those required to inhibit neutrophil infiltration and without acute liver or renal toxicity. Neutrophils treated with colchicine in vivo still produced superoxide in response to another stimulus, 4-β-phorbol-12-myristate-13-acetate. These results show a beneficial effect of colchicine for the treatment of MSU-induced superoxide production in vivo at sub-toxic doses without compromising superoxide production by other physiological processes. |
|
Note |
The technical data provided above is for reference only. |
|
References: 1. Bonfoco E, Ceccatelli S, Manzo L, et al. Colchicine induces apoptosis in cerebellar granule cells. Experimental Cell Research, 1995, 218(1): 189-200. 2. Chia EW, Grainger R, Harper JL. Colchicine suppresses neutrophil superoxide production in a murine model of gouty arthritis: a rationale for use of low-dose colchicine. British Journal of Pharmacology, 2008, 153(6): 1288-1295. |
|
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data