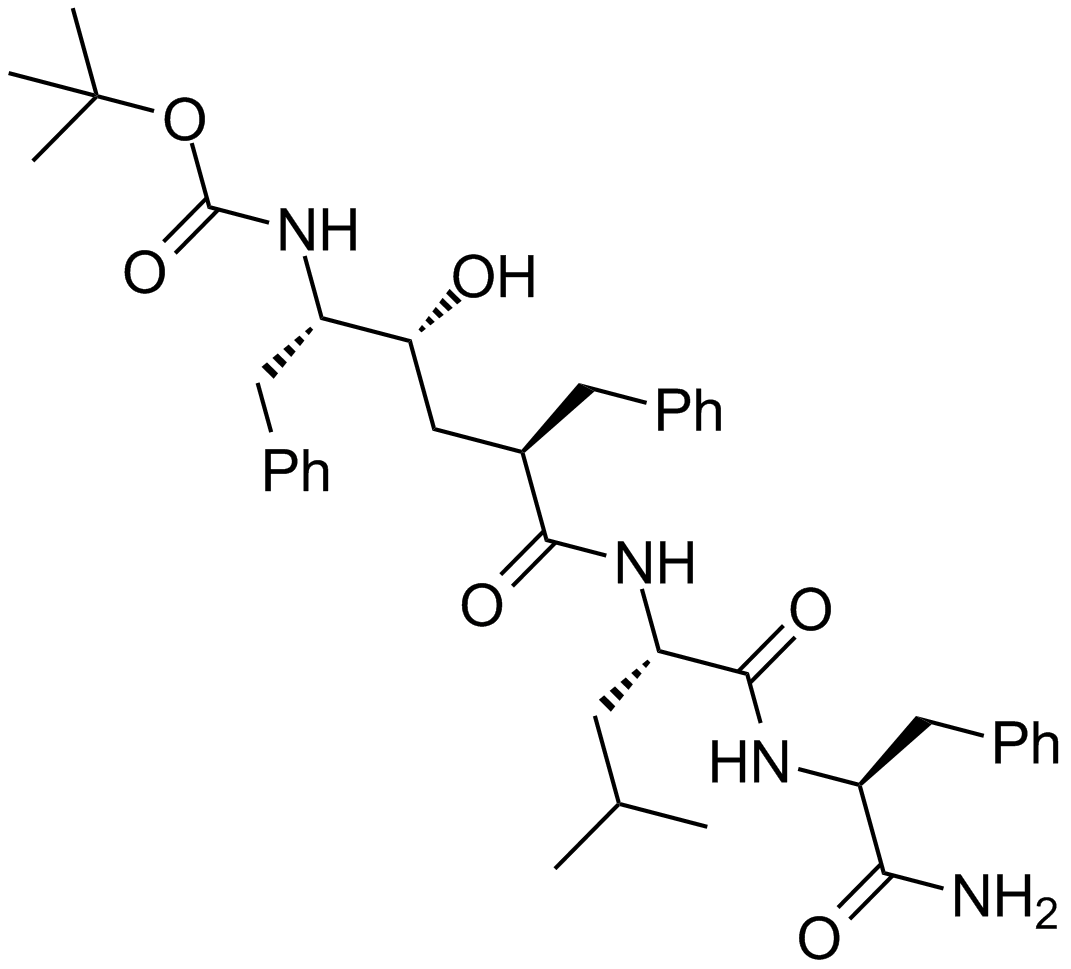
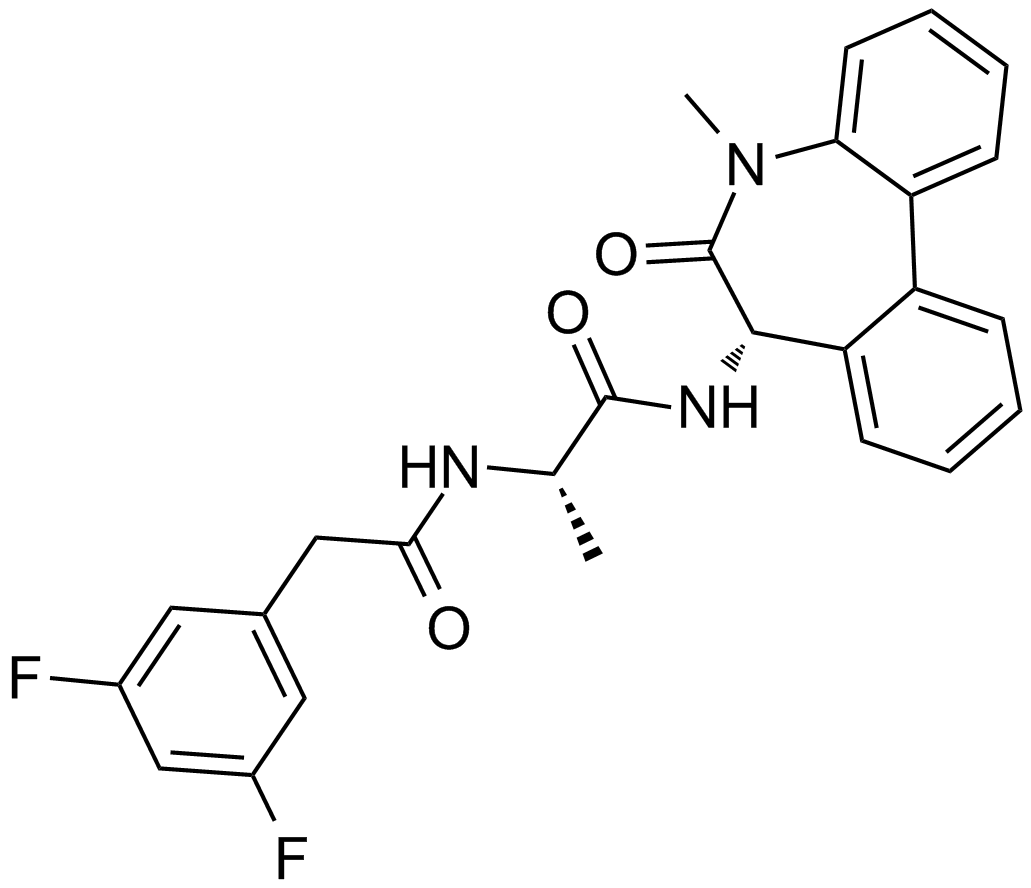
L-685,458
L-685,458 is a selective and Potent inhibitor of γ-secretase with IC50 value of 17 nM[1].
γ-Secretase is one of intramenbrane-cleaving aspartyl protease which cleaves many type-I membrane proteins and many of them have important biological functions. γ-Secretase is a multi-subunit protease and it contains presenilin, nicastrin, APH-1(Anterior Pharynx- defective 1) and PEN-2. Presenilin contains Asp258 and Asp385 embedded in the sixth and seventh transmembrane domain and forms the active site. The feature of being a membrane integrated protease complex makes it difficult to be purified as well as studying its mechanism. γ-secretase is responsible for the generation Aβfrom the amyloid precusor protein. γ-Secretase has been considered as an important drug target for Alzheimer's disease. γ-Secertase also is responsible for Notch processing which is related to cancer such as leukemia.
L-685,458 is a transition state analogue inhibitor.
It is a pan-inhibitor of γ-secretase which decrease the products of Aβ(42) and Aβ(40) peptides. It displays a selectivity of 50-fold or greater inhibition than a range of aspartyl, serine, and cysteine protease in vitro membrane assay. L-685,458 reduced the product both total Aβand Aβ42 in human primary neuronal cells with IC50 value of 115 nM for Aβand Aβ42 for 200 nM[2]. L-685,458 also inhibitedγ-secretase activity by characterizing the production of total secreted Aβby ELISA with IC50 value of about 5 nM in HEK 293 cells overexpressing human APP751.
L-685,458 reduced the cortical total Aβwith a 50% reduction occurring at 100 mg/kg in a dose-dependent manner in PDAPP, and similar reductions in cortical Aβ42 were observed. L-685,458 decreased brain levels of Aβwhen given orally to Tg2576 mice that are transgenic for human APPV717F mutation. [3] L-685,458 also inhibit the Notch signaling, then affects embryonic development in zebrafish embryos[4].
References:
1.Shearman MS, Beher D, Clarke EE, Lewis HD, Harrison T, Hunt P, Nadin A, Smith AL, Stevenson G, Castro JL: L-685,458, an aspartyl protease transition state mimic, is a potent inhibitor of amyloid beta-protein precursor gamma-secretase activity. Biochemistry 2000, 39(30):8698-8704.
2.Dovey HF, John V, Anderson JP, Chen LZ, de Saint Andrieu P, Fang LY, Freedman SB, Folmer B, Goldbach E, Holsztynska EJ et al: Functional gamma-secretase inhibitors reduce beta-amyloid peptide levels in brain. J Neurochem 2001, 76(1):173-181.
3.Lanz TA, Himes CS, Pallante G, Adams L, Yamazaki S, Amore B, Merchant KM: The gamma-secretase inhibitor N-[N-(3,5-difluorophenacetyl)-L-alanyl]-S-phenylglycine t-butyl ester reduces A beta levels in vivo in plasma and cerebrospinal fluid in young (plaque-free) and aged (plaque-bearing) Tg2576 mice. J Pharmacol Exp Ther 2003, 305(3):864-871.
4.Geling A, Steiner H, Willem M, Bally-Cuif L, Haass C: A gamma-secretase inhibitor blocks Notch signaling in vivo and causes a severe neurogenic phenotype in zebrafish. EMBO Rep 2002, 3(7):688-694.
| Physical Appearance | White solid |
| Storage | Store at -20°C |
| M.Wt | 672.85 |
| Cas No. | 292632-98-5 |
| Formula | C39H52N4O6 |
| Solubility | ≥33.65 mg/mL in DMSO; insoluble in H2O; insoluble in EtOH |
| Chemical Name | tert-butyl N-[(2S,3R,5R)-6-[[(2S)-1-[[(2S)-1-amino-1-oxo-3-phenylpropan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]amino]-5-benzyl-3-hydroxy-6-oxo-1-phenylhexan-2-yl]carbamate |
| Canonical SMILES | CC(C)CC(C(=O)NC(CC1=CC=CC=C1)C(=O)N)NC(=O)C(CC2=CC=CC=C2)CC(C(CC3=CC=CC=C3)NC(=O)OC(C)(C)C)O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1-4]: | |
|
Cell lines |
HeLa and SiHa cells, SGHPL-5 cells |
|
Preparation method |
The solubility of this compound in DMSO is > 10 mM. General tips for obtaining a higher concentration: Please warm the tube at 37 ℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reacting condition |
48 hours |
|
Applications |
In HeLa and SiHa cells, L-685,458 (8 μM) blocked Notch activation. In L-685,458-treated cells, Hes1 nuclear translocation was largely blocked and about 6.63% of HeLa and 9.03% of SiHa cells were subjected to apoptosis. L-685,458 (10 μM, 3 days) inhibited Notch pathways and caused accumulation of oocytes at the pachytene stages and decreased the number of oocytes that are able to reach diplotene. In SGHPL-5 cells, L-685,458 suppressed canonical notch activity. L-685,458 significantly increased the amount of BrdU-labeled primary CTB cells. L-685,458 increased migration of SGHPL-5 cells. L-685,458 (10 μM, 48 hours) strongly increased motility of the nonproliferating EVTs. L-685,458 (0.5 μM) attenuated the isoflurane-induced increase in Tau-PS262 levels in WT mice and AD Tg mice primary neurons. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Zhang P, Li H, Yang B, et al. Biological significance and therapeutic implication of resveratrol-inhibited Wnt, Notch and STAT3 signaling in cervical cancer cells[J]. Genes & cancer, 2014, 5(5-6): 154. [2]. Feng Y M, Liang G J, Pan B, et al. Notch pathway regulates female germ cell meiosis progression and early oogenesis events in fetal mouse[J]. Cell Cycle, 2014, 13(5): 782-791. [3]. Haider S, Meinhardt G, Velicky P, et al. Notch signaling plays a critical role in motility and differentiation of human first-trimester cytotrophoblasts[J]. Endocrinology, 2014, 155(1): 263-274. [4]. Dong Y, Wu X, Xu Z, et al. Anesthetic isoflurane increases phosphorylated tau levels mediated by caspase activation and Aβ generation[J]. PloS one, 2012, 7(6): e39386. | |
Quality Control & MSDS
- View current batch:
Chemical structure