GTP-Binding Protein Fragment, G alpha
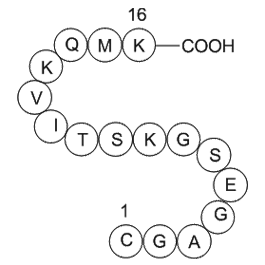
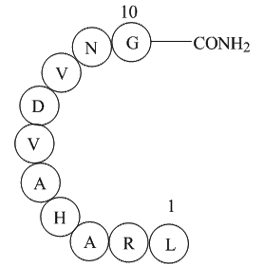
Sequence: Cys-Gly-Ala-Gly-Glu-Ser-Gly-Lys-Ser-Thr-Ile-Val-Lys-Gln-Met-Lys
Using specific antisera raised against synthetic peptides, we find that three distinct GTP-binding protein alpha subunits remain bound to the plasma membrane even after activation with nonhydrolyzable GTP analog. Trypsin cleaves each alpha subunit at a site near the amino-terminus and quantitatively releases the large fragment (composed of all but an amino-terminal 2kDa piece) from the membrane. Previous results indicated that alpha subunits are essentially cytoplasmic proteins tethered to the inner surface of the membrane via an amino terminal stalk.
Ref:
1. Brock Eide, Peter Gierschik, Graeme Milligan, Ian Mullaney, Cecilia Unson, Paul Goldsmith, Allen Spiegel, Biochemical and Biophysical Research Communications, Volume 148, Issue 3, 13 November 1987, Pages 1398–1405
2. J. Falloon, H. Malech, G. Milligan, C. Unson, R. Kahn, P. Goldsmith, A. Spiegel, FEBS Letters, 209 (1986), pp. 352–356
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 1623.89 |
| Formula | C66H118N20O23S2 |
| Synonyms | H2N-Cys-Gly-Ala-Gly-Glu-Ser-Gly-Lys-Ser-Thr-Ile-Val-Lys-Gln-Met-Lys-OH |
| Solubility | insoluble in EtOH; ≥162.3 mg/mL in DMSO; ≥51.5 mg/mL in H2O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure