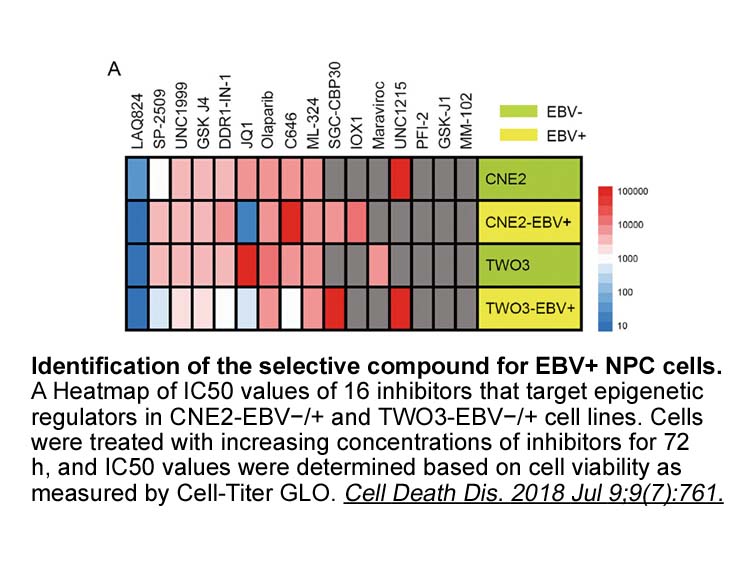
UNC1215
UNC1215 is a selective inhibitor of L3MBTL3 with IC50 value of 40 nM [1].
L3MBTL3 is a member of the MBT (malignant brain tumor) family of methyl-lysine reader proteins and is reported to play an important role in haematopoiesis and cancer biology. And it is reported that inhibition of L3MBTL3 can be regarded as a promising target used in clinic [2].
UNC1215 is a potent L3MBTL3 inhibitor and has a more potent inhibitory ability than other MBT family members. When tested with HEK293 cells transfected with a GFP fusion protein of the 3 MBT domains of L3MBTL3, UNC1215 treatment decreased the recovery time in a dose responsive manner via binding and co-localizing L3MBTL3 [1]. Using AlphaScreen○R methylated histone peptide competition assay, UNC1215 showed high antagonism ability to L3MBTL3 with IC50 value of 24±7.6 nM [3]. As the first potent and selective inhibitor for methyl-lysine reader protein-L3MBTL3-UNC1215 showed highly selective inhibitor ability via antagonizing the mono- and dimethyl-lysine reading function of L3MBTL3 [2].
References:
1. James, L.I., et al., Discovery of a chemical probe for the L3MBTL3 methyllysine reader domain. Nat Chem Biol, 2013. 9(3): p. 184-91.
2. James, L.I., et al., Small-molecule ligands of methyl-lysine binding proteins: optimization of selectivity for L3MBTL3. J Med Chem, 2013. 56(18): p. 7358-71.
3. Camerino, M.A., et al., The structure-activity relationships of L3MBTL3 inhibitors: flexibility of the dimer interface. Medchemcomm, 2013. 4(11): p. 1501-1507.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 529.72 |
| Cas No. | 1415800-43-9 |
| Formula | C32H43N5O2 |
| Synonyms | UNC 1215;UNC-1215 |
| Solubility | ≥26.05 mg/mL in DMSO; insoluble in H2O; ≥4.94 mg/mL in EtOH with gentle warming and ultrasonic |
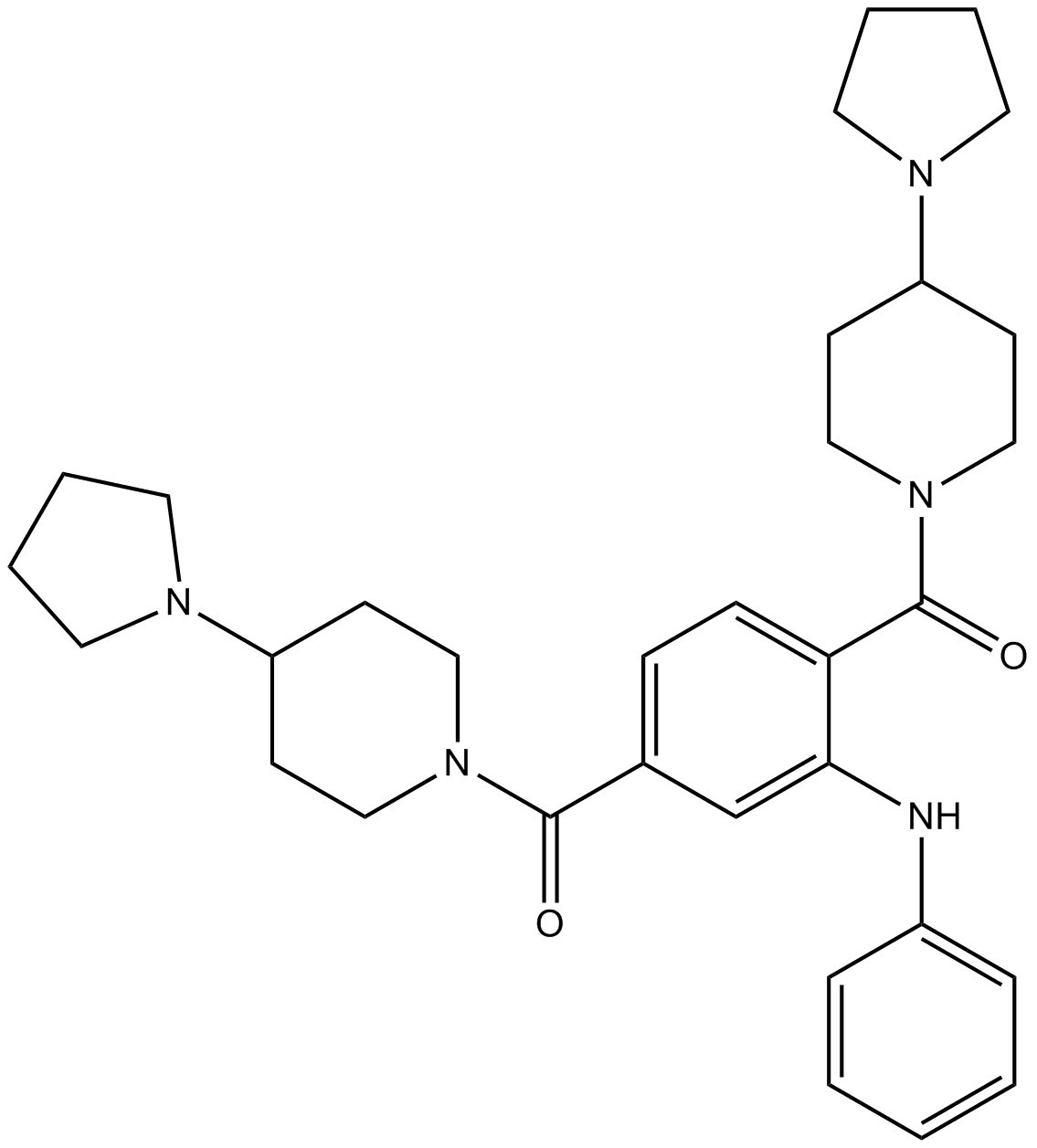
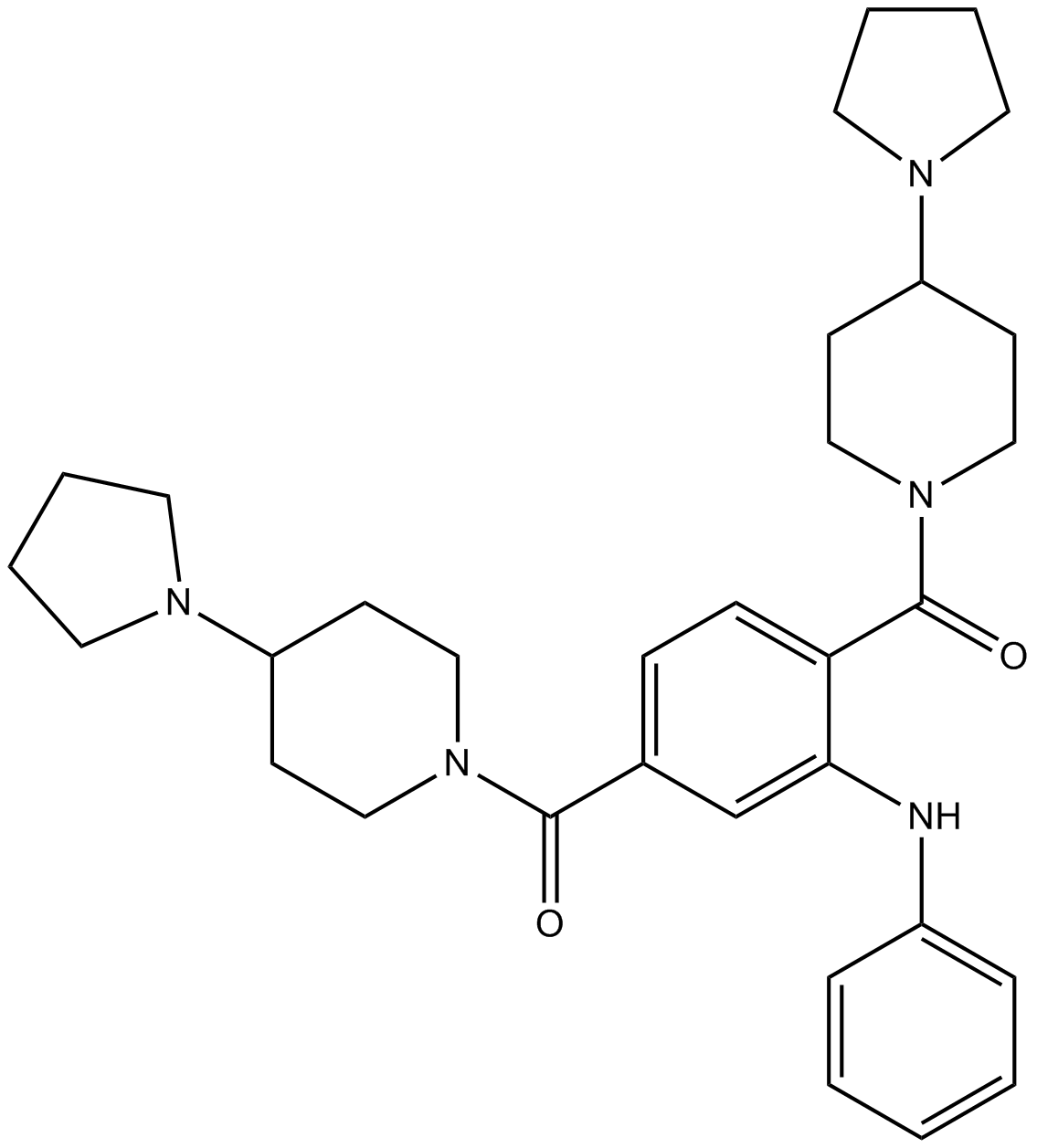
| Chemical Name | [3-anilino-4-(4-pyrrolidin-1-ylpiperidine-1-carbonyl)phenyl]-(4-pyrrolidin-1-ylpiperidin-1-yl)methanone |
| SDF | Download SDF |
| Canonical SMILES | C1CCN(C1)C2CCN(CC2)C(=O)C3=CC(=C(C=C3)C(=O)N4CCC(CC4)N5CCCC5)NC6=CC=CC=C6 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1]: | |
|
Cell lines |
Human embryonic kidney 293 cells transfected with a GFP fusion protein of the three MBT domains of L3MBTL3 (GFP-3MBT) |
|
Preparation method |
The solubility of this compound in DMSO is >26.05 mg/mL. General tips for obtaining a higher concentration: Please warm the tube at 37 ℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reacting condition |
EC50: 50–100 nM |
|
Applications |
Treatment with UNC1215 decreased the recovery time fluorescence intensity following photobleaching in a dose-responsive manner, with the EC50 of 50–100 nM, indicating that UNC1215 promoted diffusibility of GFP-3MBT within the nucleus. UNC1215 competed with cellular factors for binding of the MBT domains at concentrations well below 1 μM. UNC1215 showed potent effects on the subnuclear localization of GFP-3MBT, with an IC50 of approximately 500 nM for disruption of foci formation. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. James L I, Barsyte-Lovejoy D, Zhong N, et al. Discovery of a chemical probe for the L3MBTL3 methyllysine reader domain[J]. Nature chemical biology, 2013, 9(3): 184-191. |
|
| Description | Ko 143 is a potent inhibitor of L3MBTL3 methyllysine (Kme) reader domain with an IC50 value of 40 nM and Kd value of 120 nM. | |||||
| Targets | L3MBTL3 | L3MBTL3- D274A | ||||
| IC50 | 40 nM (Kd=120 nM) | 3.5 μM | ||||
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data