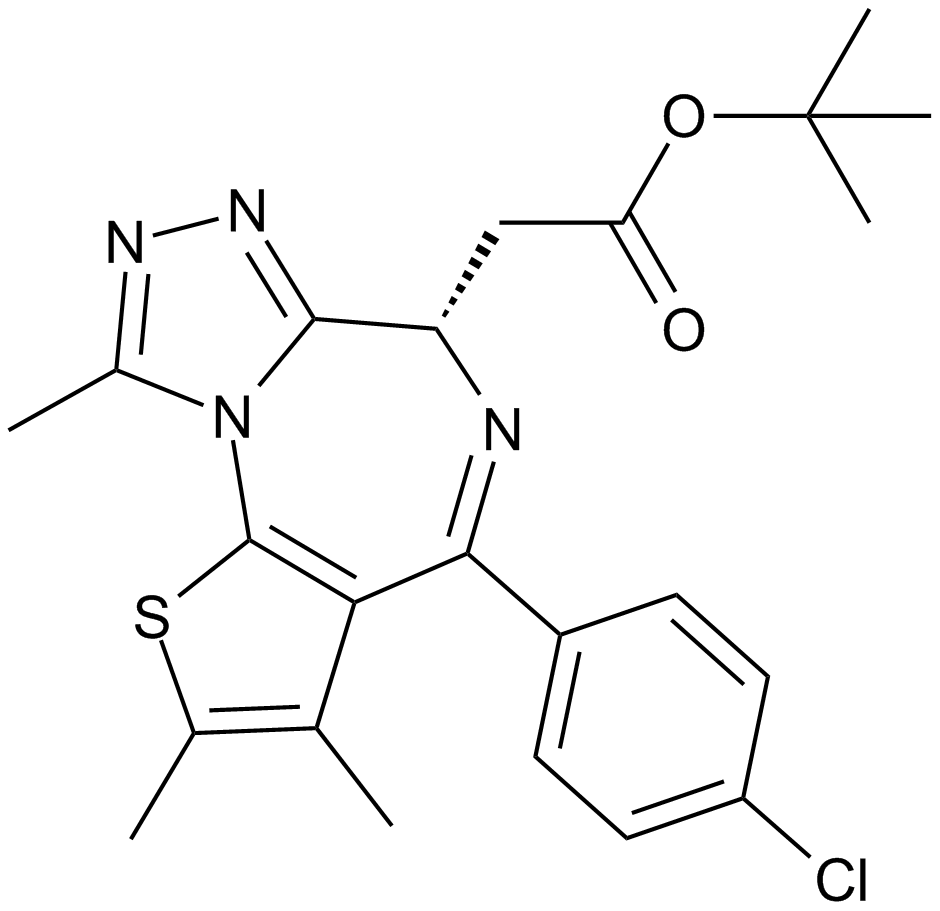
(-)-JQ1
(-)-JQ1 is the stereoisomer of JQ1, a cell-permeable small-molecule inhibitor of BET bromodomain that competitively binds to acetyl-lysine recognition motifs. JQ1 is a novel thieno-triazolo-1,4-diazepine with an appended and bulky t-butyl ester functional group at C6 position in its chemical structure, which allows for additional pendant group diversity and mitigates binding to the central benzodiazepine receptor. JQ1 competitively binds to the bromodomain displacing the BRD4 fusion oncoprotein from chromatin, which induces squamous differentiation and specific anti-proliferative effect in BRD4-dependent cell lines and patient-derived xenograft models. However, study results have shown that (-)-JQ1 fails to significantly interact with any bromodomain tested and exhibits inhibition against BRD4(1) with 50% inhibition concentration IC50 value of 10,000 nM.
Reference
Filippakopoulos P, Qi J, Picaud S, Shen Y, Smith WB, Fedorov O, Morse EM, Keates T, Hickman TT, Felletar I, Philpott M, Munro S, McKeown MR, Wang Y, Christie AL, West N, Cameron MJ, Schwartz B, Heightman TD, La Thangue N, French CA, Wiest O, Kung AL, Knapp S, Bradner JE. Selective inhibition of BET bromodomains. Nature. 2010 Dec 23;468(7327):1067-73. doi: 10.1038/nature09504. Epub 2010 Sep 24.
- 1. Kangjie Qin, Jie Zheng, et al. "Inhibition of BRD4 activates the AKT-SIRT3 signaling pathway to suppress apoptosis and attenuate hyperoxia-induced lung injury." Front Bioeng Biotechnol. 2025 Nov 7;13:1674916. PMID: 41280641
- 2. Jian Jiang, Tianci Shen, et al. "FOXM1, a super enhancer-associated gene, is related to poorer prognosis and gemcitabine resistance in pancreatic cancer." Cell Biochem Biophys. 2025 Jun;83(2):2441-2452. PMID: 39899193
- 3. Aakarsha Rao, Milan S Stosic, et al. "Targeted inhibition of BET proteins in HPV16-positive head and neck squamous cell carcinoma reveals heterogeneous transcriptional responses." Front Oncol. 2024 Sep 5:14:1440836. PMID: 39301555
- 4. Aakarsha Rao, Zijian Ni, et al. "Targeted inhibition of BET proteins in HPV-16 associated head and neck squamous cell carcinoma reveals heterogeneous transcription response." bioRxiv. 2023 Oct 4:2023.10.02.560587. PMID: 37873389
- 5. Peeters JGC, Boltjes A, et al. "Epigenetic changes in inflammatory arthritis monocytes contribute to disease and can be targeted by JAK inhibition." Rheumatology (Oxford) 2023 Jan 10. PMID: 36625523
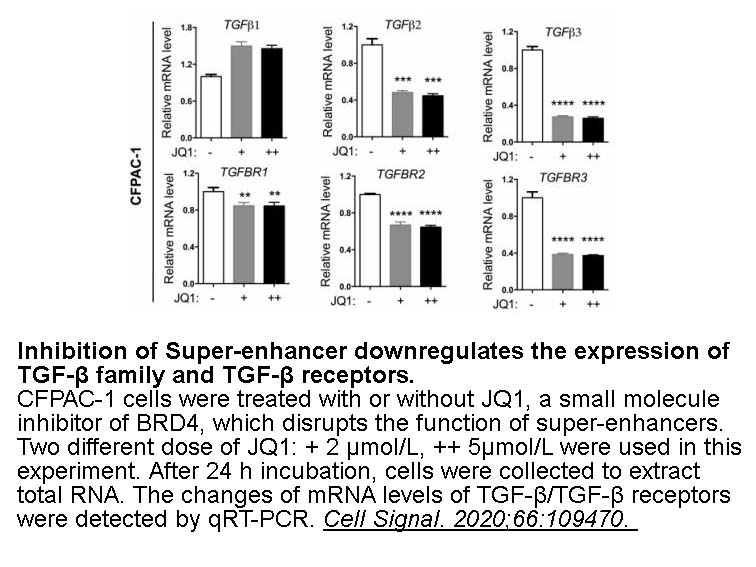
- 6. Te Zhang, Wenjie Xia, et al. "Super-enhancer hijacking LINC01977 promotes malignancy of early-stage lung adenocarcinoma addicted to the canonical TGF-β/SMAD3 pathway." J Hematol Oncol. 2022 Aug 18;15(1):114. PMID: 35982471
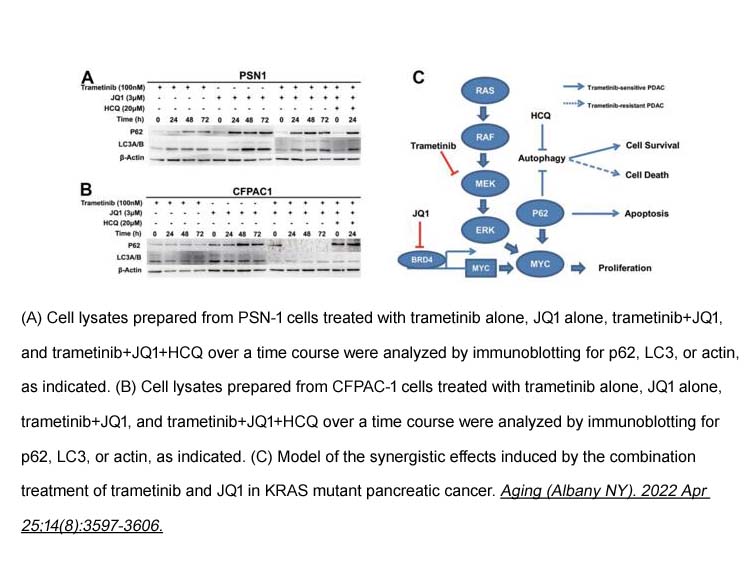
- 7. Xiaofei Zhang, Tiebo Mao, et al. "Synergistic blocking of RAS downstream signaling and epigenetic pathway in KRAS mutant pancreatic cancer." Aging (Albany NY). 2022 Apr 25;14(8):3597-3606. PMID: 35468095
- 8. Nora-Guadalupe P Ramirez, Jeon Lee, et al. "ADAP1 promotes latent HIV-1 reactivation by selectively tuning KRAS–ERK–AP-1 T cell signaling-transcriptional axis." Nat Commun. 2022 Mar 1;13(1):1109. PMID: 35232997
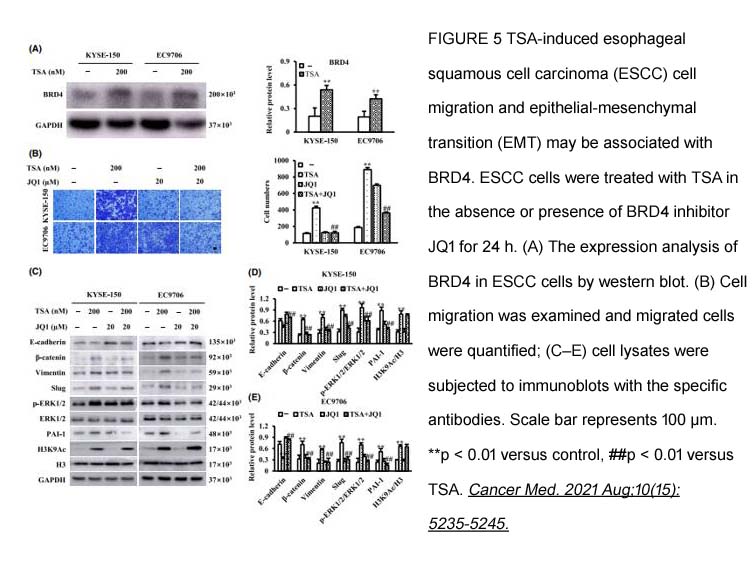
- 9. Danhui Liu, Yuzhen Liu, et al. "Trichostatin A promotes esophageal squamous cell carcinoma cell migration and EMT through BRD4/ERK1/2-dependent pathway." Cancer Med. 2021 Aug;10(15):5235-5245 PMID: 34160902
- 10. Seung-Kyoon Kim, Xihui Liu, et al. "Functional coordination of BET family proteins underlies altered transcription associated with memory impairment in fragile X syndrome." Sci Adv. 2021 May 19;7(21):eabf7346. PMID: 34138732
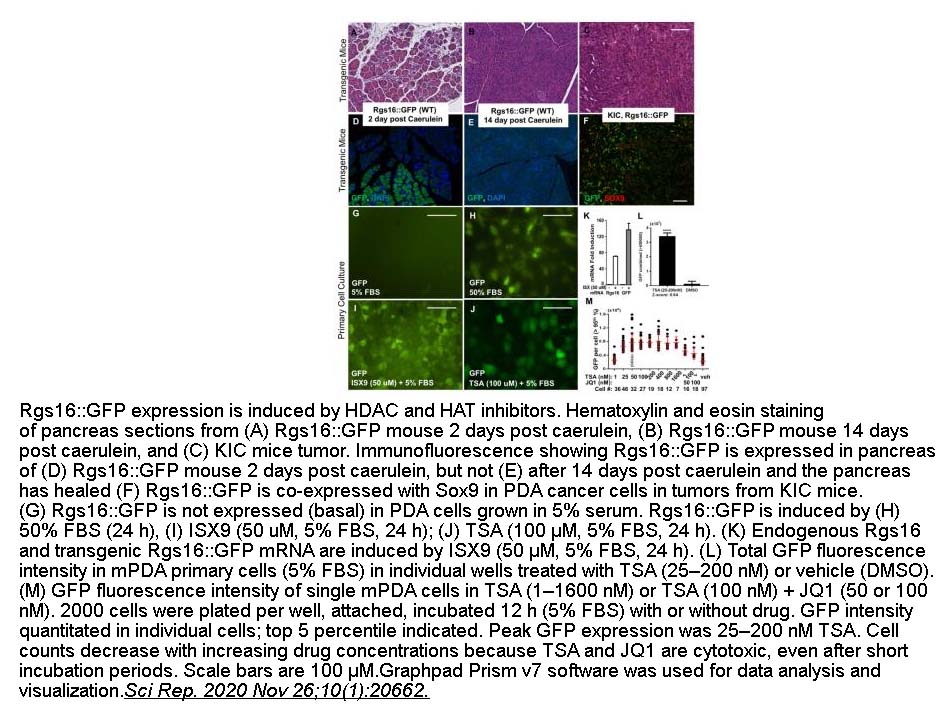
- 11. Somayeh Layeghi-Ghalehsoukhteh, Shreoshi Pal Choudhuri, et al. "Concerted cell and in vivo screen for pancreatic ductal adenocarcinoma (PDA) chemotherapeutics." Sci Rep. 2020 Nov 26;10(1):20662 PMID: 33244070
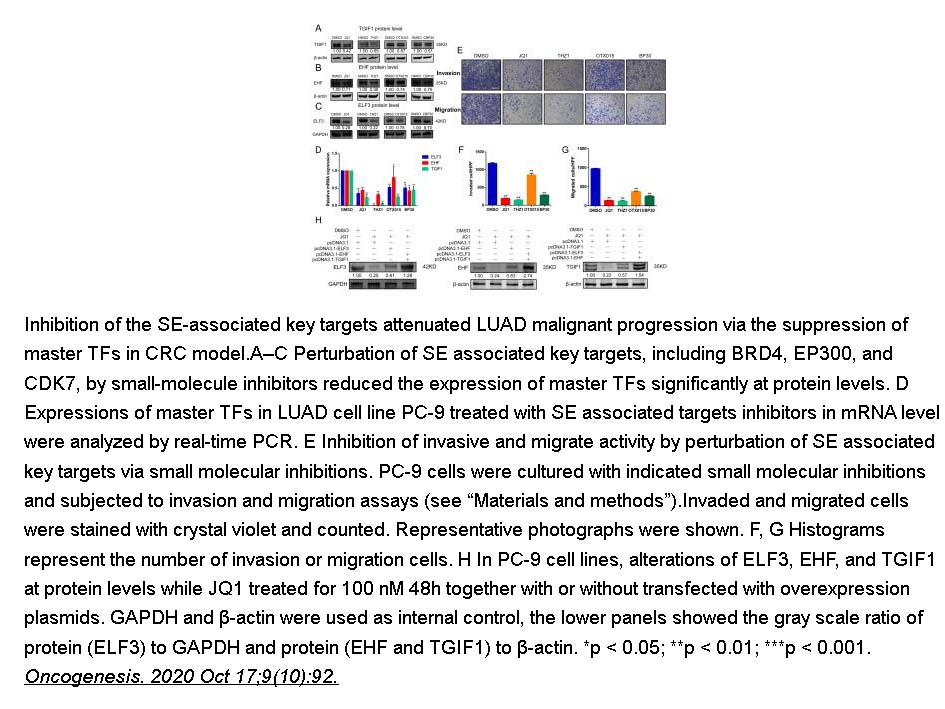
- 12. Te Zhang, Xuming Song, et al. "Aberrant super-enhancer landscape reveals core transcriptional regulatory circuitry in lung adenocarcinoma." Oncogenesis. 2020 Oct 17;9(10):92 PMID: 33070167
- 13. Wu SY, Lee CF, et al. "Opposing Functions of BRD4 Isoforms in Breast Cancer." Mol Cell. 2020;78(6):1114-1132.e10 PMID: 32446320
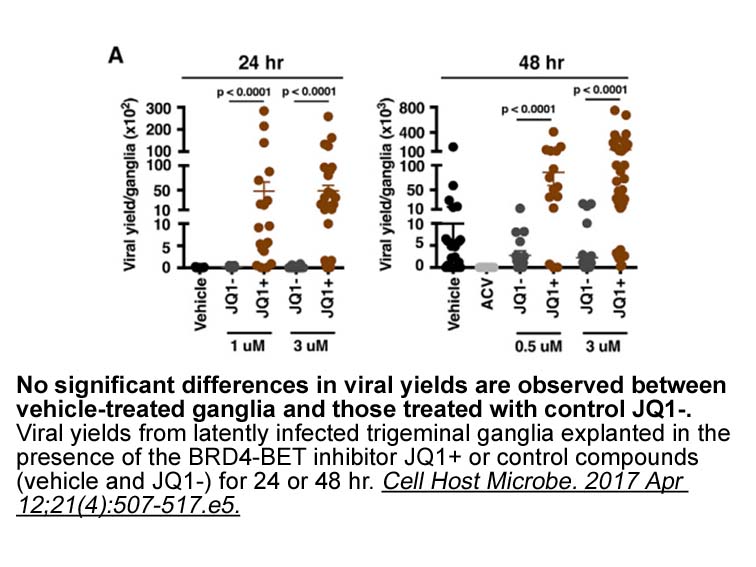
- 14. Alfonso-Dunn R, Turner AW, et al. "Transcriptional Elongation of HSV Immediate Early Genes by the Super Elongation Complex Drives Lytic Infection and Reactivation from Latency." Cell Host Microbe. 2017 Apr 12;21(4):507-517.e5 PMID: 28407486
- 15. Alonso, Victoria Lucia, et al. "Overexpression of bromodomain factor 3 in Trypanosoma cruzi (TcBDF3) affects differentiation of the parasite and protects it against bromodomain inhibitors." FEBS Journal (2016) PMID: 27007774
- 16. Peeters, Janneke GC, et al. "Inhibition of Super-Enhancer Activity in Autoinflammatory Site-Derived T Cells Reduces Disease-Associated Gene Expression." Cell reports (2015) PMID: 26387944
- 17. Sengupta, Surojeet, et al. "Inhibition of BET proteins impairs estrogen-mediated growth and transcription in breast cancers by pausing RNA polymerase advancement." Breast cancer research and treatment (2015): 1-14 PMID: 25721606
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 456.99 |
| Cas No. | 1268524-71-5 |
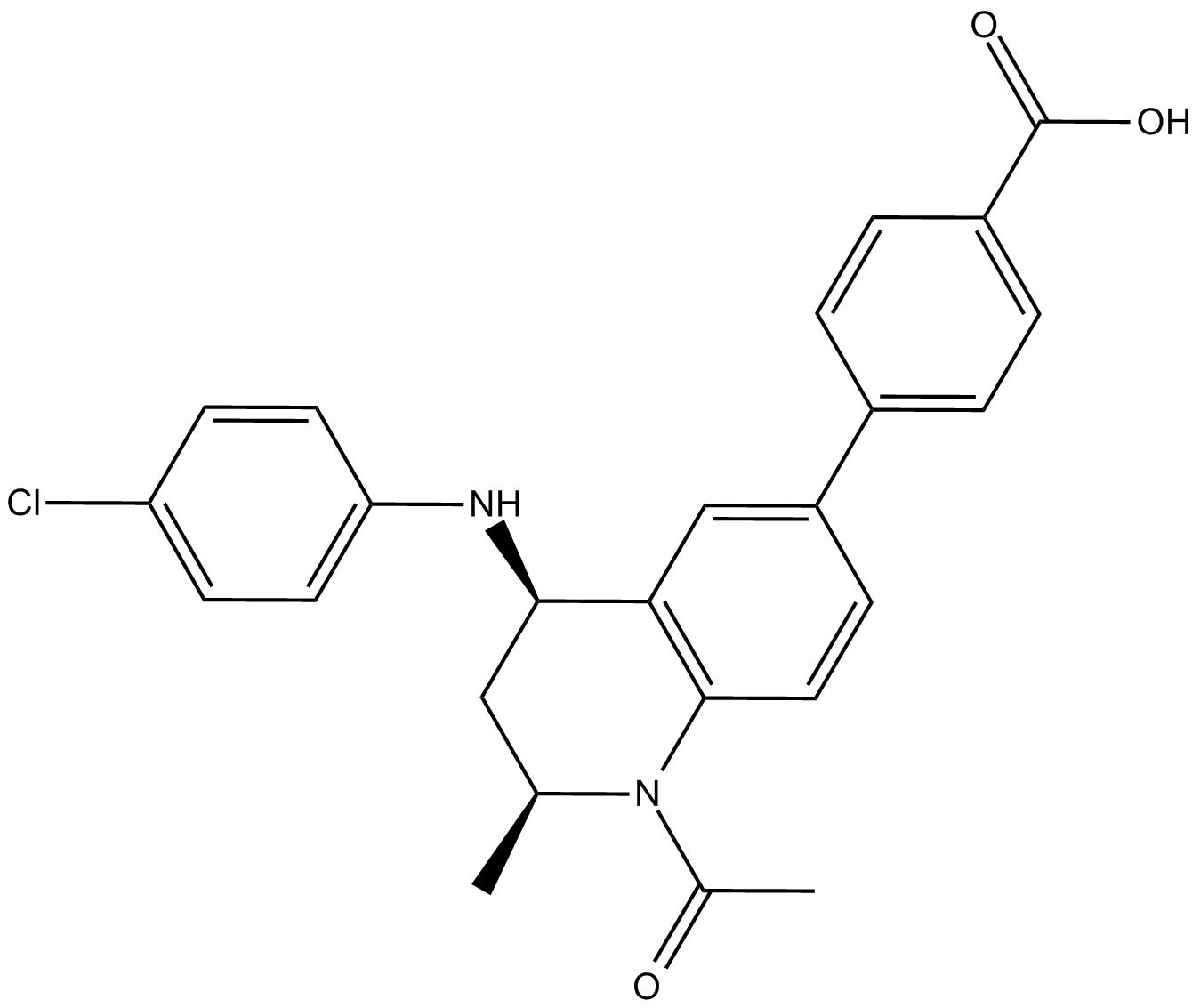
| Formula | C23H25ClN4O2S |
| Synonyms | (-)-JQ1 |
| Solubility | ≥22.85 mg/mL in DMSO; insoluble in H2O; ≥46.9 mg/mL in EtOH with ultrasonic |
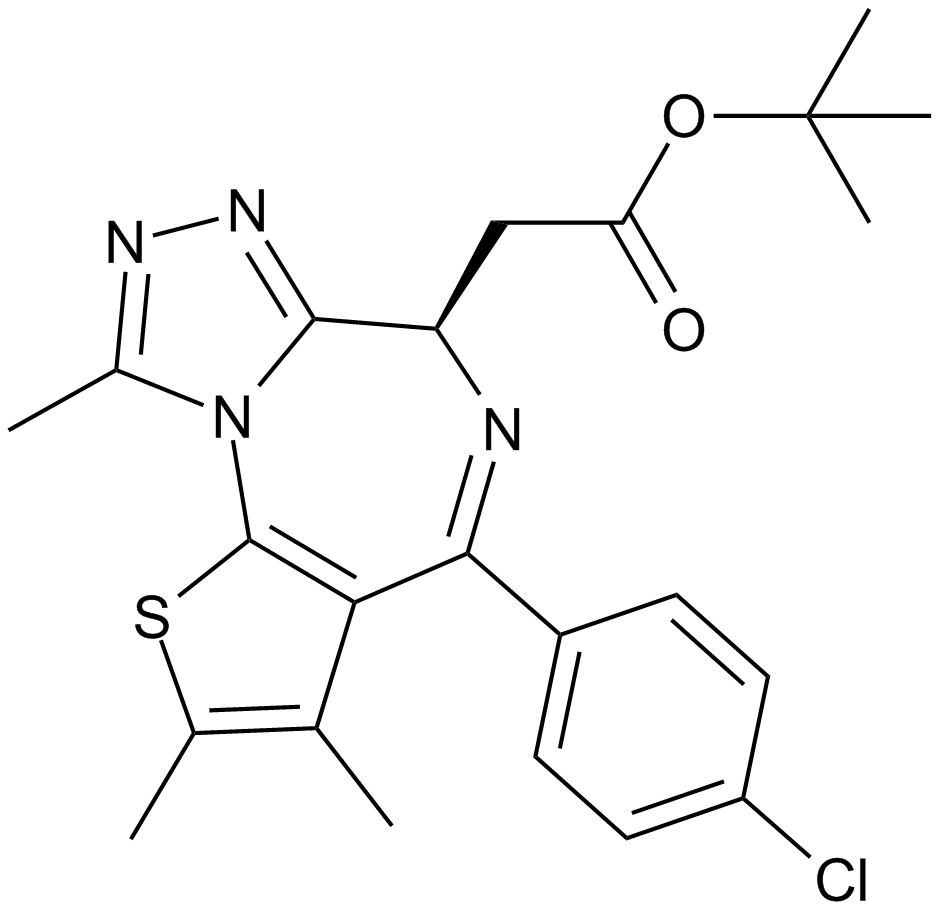
| Chemical Name | (R)-tert-butyl 2-(4-(4-chlorophenyl)-2,3,9-trimethyl-6H-thieno[3,2-f][1,2,4]triazolo[4,3-a][1,4]diazepin-6-yl)acetate |
| Canonical SMILES | CC(C)(C)OC(C[C@H](c1nnc(C)[n]1-c1c2c(C)c(C)[s]1)N=C2c(cc1)ccc1Cl)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment: [1] | |
|
Cell lines |
BRD4-dependent NMC cells |
|
Preparation method |
The solubility of this compound in DMSO is >10 mM. General tips for obtaining a higher concentration: Please warm the tube at 37 °C for 10 minutes and/or shake it in the ultrasonic bath for a while.Stock solution can be stored below -20°C for several months. |
|
Reaction Conditions |
250 nM, 72 hours for proliferation inhibition 250 nM, 48 hours for cell-cycle arrest induction |
|
Applications |
In BRD4-dependent NMC cells, differentiation is expectedly accompanied by growth arrest, as demonstrated by reduced Ki67 staining, sustained inhibition of proliferation and G1 cell-cycle arrest. The quantitative RT–PCR for RAD21 and RAN was performed. (-)-JQ1 enantiomer was an inactive control of (+)-JQ1. (+)-JQ1 potently decreased expression of both BRD4 target genes, whereas (-)-JQ1 had no effect. |
| Animal experiment: [1] | |
|
Animal models |
Female NCr nude mice bearing NMC 797 xenografts |
|
Dosage form |
Intraperitoneal injection, 50 mg/kg, daily |
|
Applications |
After 4 days of therapy, mice were evaluated by FDG-PET imaging. A marked reduction in FDG uptake was observed with JQ1 ((+)/-) treatment, whereas vehicle-treated mice demonstrated progressive disease. Tumour-volume measurements confirmed a reduction in tumour growth with JQ1 treatment. JQ1 was well tolerated at this dose and schedule without overt signs oftoxicity or weight loss. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1] Filippakopoulos P, Qi J, Picaud S, et al. Selective inhibition of BET bromodomains. Nature, 2010, 468(7327): 1067-1073. |
|
| Description | (-)-JQ-1, the stereoisomer (+)-JQ1, showed no significant interaction with any bromodomain. | |||||
| Targets | BET bromodomain | |||||
| IC50 | ~ 50 and 90 nM | |||||
Quality Control & MSDS
- View current batch:
Chemical structure
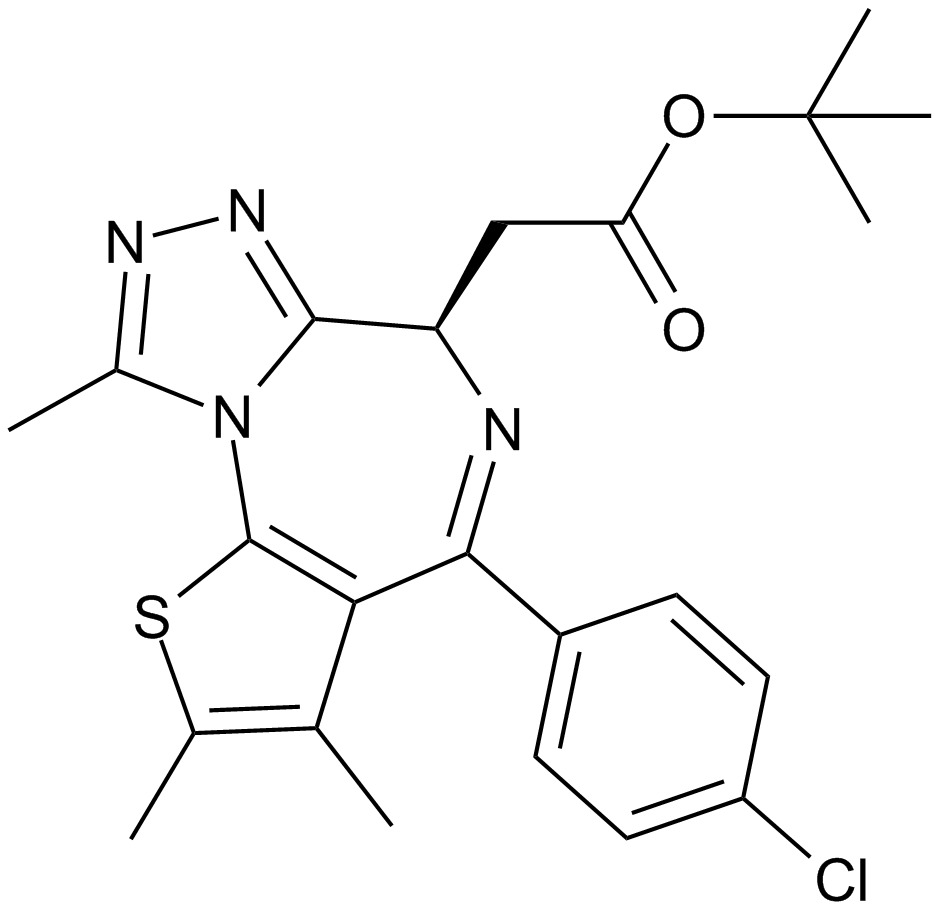
Related Biological Data
Related Biological Data
Related Biological Data
Related Biological Data
Related Biological Data
Related Biological Data
Related Biological Data