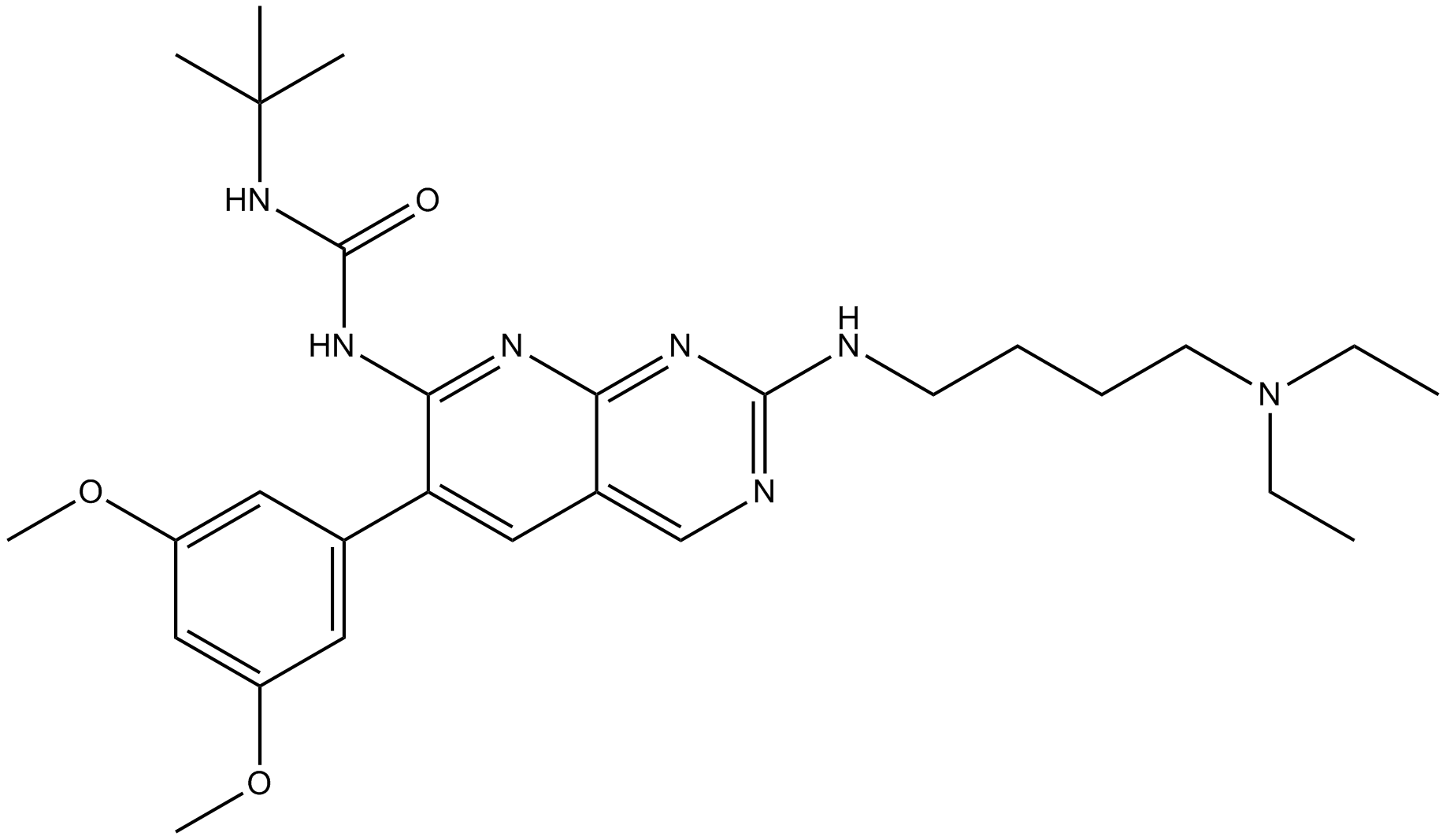
Pazopanib (GW-786034)
Pazopanib is a potent and selective second generation multi-targeted receptor tyrosine kinase inhibitor of vascular endothelial growth factor receptor (VEGFR), platelet-derived growth factor receptor (PDGFR) and c-kit tyrosine kinases. It is involved in inhibiting signaling pathways, angiogenesis, and cell proliferation. Pazopanib exhibited excellent anti-angiogenic and anti-tumor activity and synergism was observed in combination with chemotherapeutic drugs in several mouse models of a variety of tumors, accompanied by desirable pharmacokinetics and oral bioavailability. It inhibits the intracellular tyrosine kinase portion of all the VEGFR subtypes, and exhibits distinct pharmacokinetic and toxicity profiles compared with other agents among VEGF signaling pathway inhibitors. Recent studies elucidated its importance of signaling cascades related to angiogenesis in the management of RCC.
Reference
Sonpavde G, Hutson T E. Pazopanib: a novel multitargeted tyrosine kinase inhibitor. Current oncology reports. 2007; 9(2): 115 - 119.
Podar K, Tonon G, Sattler M, et al. The small-molecule VEGF receptor inhibitor pazopanib (GW786034B) targets both tumor and endothelial cells in multiple myeloma. Proceedings of the National Academy of Sciences. 2006; 103(51): 19478 - 19483.
| Storage | Desiccate at -20°C |
| M.Wt | 437.52 |
| Cas No. | 444731-52-6 |
| Formula | C21H23N7O2S |
| Synonyms | Pazopanib, Votrient, GW786034B, GW 786034, GW-786034 |
| Solubility | insoluble in EtOH; insoluble in H2O; ≥10.95 mg/mL in DMSO |
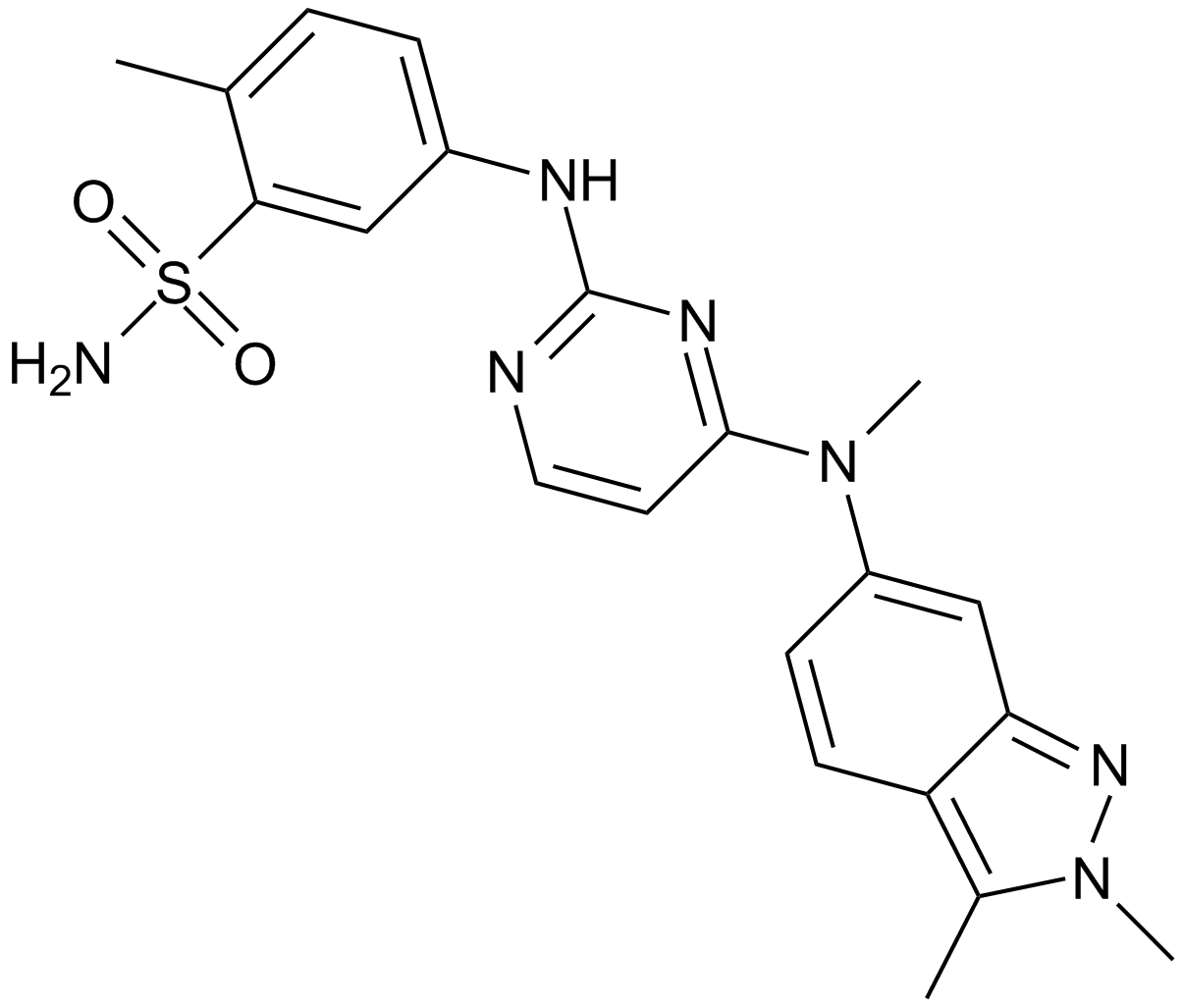
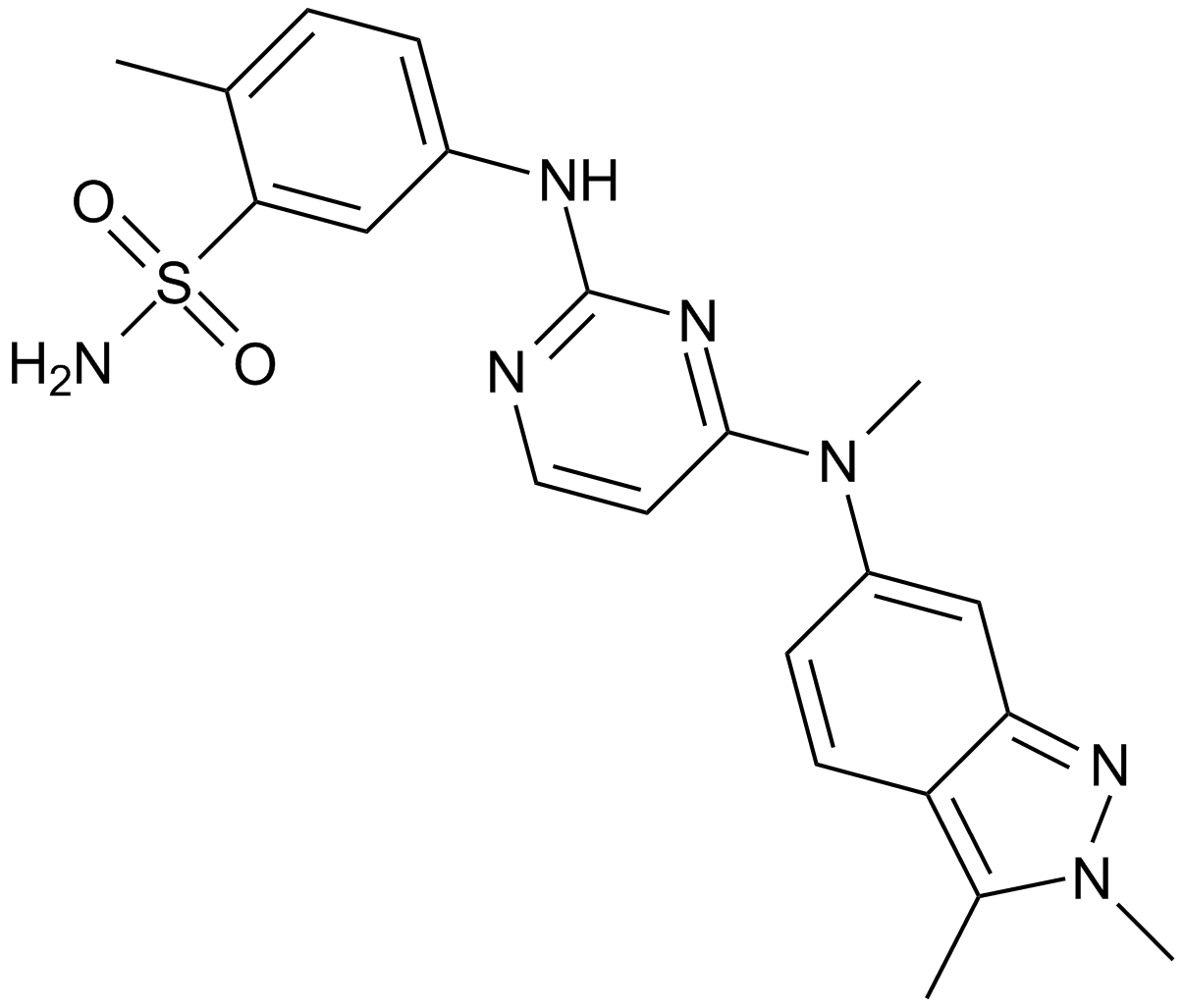
| Chemical Name | 5-[[4-[(2,3-dimethylindazol-6-yl)-methylamino]pyrimidin-2-yl]amino]-2-methylbenzenesulfonamide;hydrochloride |
| SDF | Download SDF |
| Canonical SMILES | O=S(=O)(N)C1=CC(=CC=C1C)NC2=NC=CC(=N2)N(C=3C=CC=4C(=NN(C4C)C)C3)C |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment: [1] | |
|
Cell lines |
Primary human brain microvascular endothelial cells (HBMEC) |
|
Preparation method |
The solubility of this compound in DMSO is >10 mM. General tips for obtaining a higher concentration: Please warm the tube at 37 °C for 10 minutes and/or shake it in the ultrasonic bath for a while.Stock solution can be stored below -20°C for several months. |
|
Reaction Conditions |
IC50: 2 μM, 48 hours |
|
Applications |
The IC50 for pazopanib for anchorage-dependent growth was 2 μM and 1 μM after 48 h and 72 h, respectively. Pazopanib abrogated the phosphorylation of VEGFR2 with disruption of downstream PLCγ1. It also disrupted the Ras-Raf-ERK pathway through decreased phosphorylated MEK1/2 and ERK1/2 and affected the phosphorylation of 70S6K. Our findings confirmed that pazopanib targeted endothelial cells, affecting cell growth, VEGFR-induced signaling, and tube formation. |
| Animal experiment: [2] | |
|
Animal models |
Immune-deficient beige-nude-xid (BNX) mice injected with MM.1S cells |
|
Dosage form |
Oral administration, 30 mg/kg and 100 mg/kg, daily for five weeks |
|
Applications |
Tumor growth in treated mice was significantly delayed (30 mg/kg) or almost totally inhibited (100 mg/kg) compared with the control group. However, tumors rapidly regrew after cessation of treatment at day 30. Using Kaplan–Meier and log-rank analysis, the mean overall survival (OS) was 20 days in the control cohort versus 41 days and 51 days in groups treated with 30 mg/kg and 100 mg/kg pazopanib, respectively. Statistically significant prolongation in mean OS compared with control mice was observed in animals treated with 30 mg/kg and 100 mg/kg. Importantly, treatment with either the vehicle alone or pazopanib did not affect body weight. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1] Gril B, Palmieri D, Qian Y, et al. Pazopanib reveals a role for tumor cell B-Raf in the prevention of HER2+ breast cancer brain metastasis. Clinical Cancer Research, 2011, 17(1): 142-153. [2] Podar K, Tonon G, Sattler M, et al. The small-molecule VEGF receptor inhibitor pazopanib (GW786034B) targets both tumor and endothelial cells in multiple myeloma. Proceedings of the National Academy of Sciences, 2006, 103(51): 19478-19483. | |
| Description | Pazopanib is a novel multi-target inhibitor of VEGFR1, VEGFR2, VEGFR3, PDGFR, FGFR, c-Kit and c-Fms with IC50 of 10 nM, 30 nM, 47 nM, 84 nM, 74 nM, 140 nM and 146 nM, respectively. | |||||
| Targets | VEGFR1 | VEGFR2 | VEGFR3 | PDGFR | FGFR | |
| IC50 | 10 nM | 30 nM | 47 nM | 84 nM | 74 nM | |
Quality Control & MSDS
- View current batch:
Chemical structure
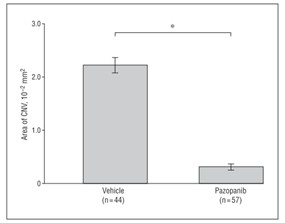
Related Biological Data
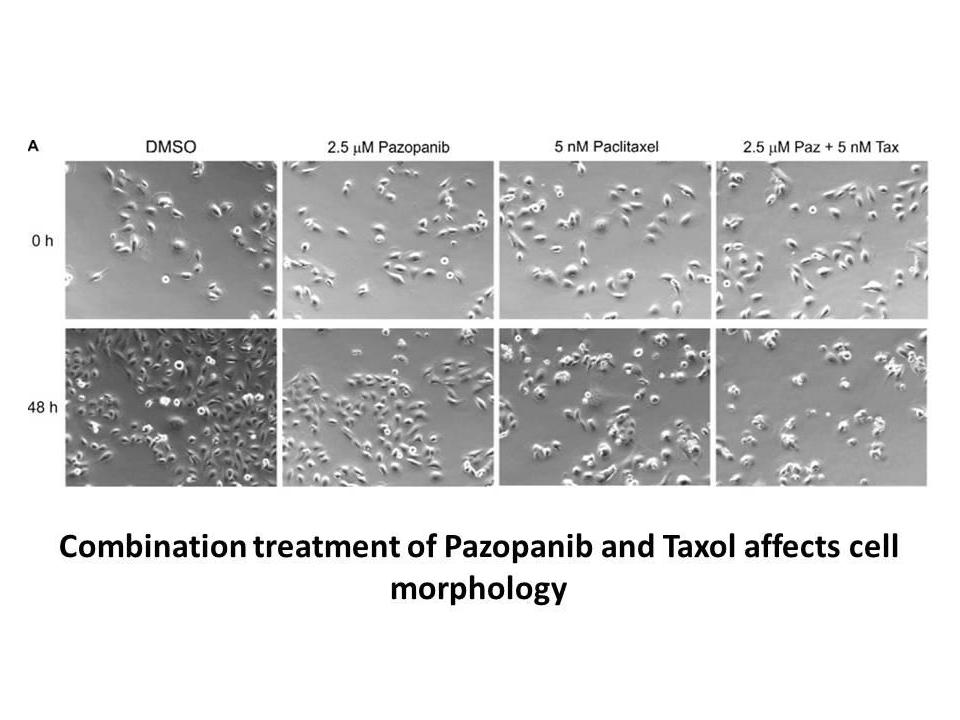
Related Biological Data
Related Biological Data