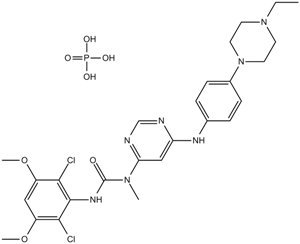
PD 173074
PD 173074 (CAS No. 219580-11-7) is a highly selective tyrosine kinase inhibitor. Its core biological function is to inhibit fibroblast growth factor receptor (FGFR) and vascular endothelial growth factor receptor 2 (VEGFR2) activity, blocking angiogenesis, tumor cell proliferation, and metastasis mediated by related signaling pathways. It can also reverse ABCB1/ABCC10-mediated multidrug resistance, and in schizophrenia-related studies it has shown potential for high-affinity binding to FGFR1. Its targets and mode of action are inhibition of the tyrosine kinase activity of FGFR1 (primary target), FGFR3, and VEGFR2 (ATP-competitive binding to the ATP-binding pocket of FGFR1), antagonizing FGF-2-mediated biological effects, with low selectivity toward other kinases such as PDGFR, c-Src, EGFR, and the insulin receptor.
Key activity data: FGFR1 binding IC₅₀ 21.5±0.8 nM, functional cAMP IC₅₀ 15.7±5.4 nM; mouse FGFR1 IC₅₀ 122 nM, rat 727±350 nM, rhesus monkey 55.5 nM; VEGFR2 autophosphorylation inhibition IC₅₀ 100-200 nM; PDGFR IC₅₀ 17.6±1.9 μM, c-Src IC₅₀ 19.8±2.3 μM; EGFR and insulin receptor IC₅₀ both > 50 μM.
Animal modeling is commonly used for model treatment; common models include the mouse corneal neovascularization model (to evaluate inhibition of FGF/VEGF-induced angiogenesis), the human GCGR transgenic mouse acute glucagon challenge model, colorectal cancer xenograft models, head and neck squamous cell carcinoma (HNSCC)-related models, and schizophrenia-related models.
Common applications and concentrations: In cell culture, CHO-hGCGR cells use nM-level concentrations for receptor binding and functional assays; HNSCC cell lines (e.g., A253, CAL-27, etc.) are used for drug sensitivity testing; ABCB1-overexpressing cell lines (e.g., HEK293, human epidermoid carcinoma drug-resistant sublines) use 100 μM-level concentrations for multidrug resistance reversal experiments. In animal studies, intraperitoneal injection in mice at 1-2 mg/kg/d is used to inhibit corneal neovascularization, and oral dosing at 3-30 mg/kg is used for treatment in tumor-related models. Effective treatment concentrations: in animal studies, 1 mg/kg/d can inhibit 86% of FGF-induced angiogenesis and 2 mg/kg/d can inhibit 81% of VEGF-induced angiogenesis; at the cellular level, nM-level concentrations can significantly inhibit FGFR1/VEGFR2 activity, and 100 μM-level concentrations can reverse ABCB1-mediated paclitaxel and vinblastine resistance. In HNSCC cells, the methylation status of specific CpG sites is associated with drug sensitivity, and hypomethylated cells are more sensitive to it.
References:
[1] Mohammadi M, Froum S, Hamby JM, Schroeder MC, Panek RL, Lu GH, Eliseenkova AV, Green D, Schlessinger J, Hubbard SR. Crystal structure of an angiogenesis inhibitor bound to the FGF receptor tyrosine kinase domain. EMBO J. 1998 Oct 15;17(20):5896-904. doi: 10.1093/emboj/17.20.5896. PMID: 9774334; PMCID: PMC1170917.
[2] Skaper SD, Kee WJ, Facci L, Macdonald G, Doherty P, Walsh FS. The FGFR1 inhibitor PD 173074 selectively and potently antagonizes FGF-2 neurotrophic and neurotropic effects. J Neurochem. 2000 Oct;75(4):1520-7. doi: 10.1046/j.1471-4159.2000.0751520.x. PMID: 10987832.
[3] Widberg CH, Newell FS, Bachmann AW, Ramnoruth SN, Spelta MC, Whitehead JP, Hutley LJ, Prins JB. Fibroblast growth factor receptor 1 is a key regulator of early adipogenic events in human preadipocytes. Am J Physiol Endocrinol Metab. 2009 Jan;296(1):E121-31. doi: 10.1152/ajpendo.90602.2008. Epub 2008 Oct 21. PMID: 18940940.
[4] Rodriguez-Otero P, Román-Gómez J, Vilas-Zornoza A, José-Eneriz ES, Martín-Palanco V, Rifón J, Torres A, Calasanz MJ, Agirre X, Prosper F. Deregulation of FGFR1 and CDK6 oncogenic pathways in acute lymphoblastic leukaemia harbouring epigenetic modifications of the MIR9 family. Br J Haematol. 2011 Oct;155(1):73-83. doi: 10.1111/j.1365-2141.2011.08812.x. Epub 2011 Aug 2. PMID: 21810092.
[5] Saito S, Morishima K, Ui T, Hoshino H, Matsubara D, Ishikawa S, Aburatani H, Fukayama M, Hosoya Y, Sata N, Lefor AK, Yasuda Y, Niki T. The role of HGF/MET and FGF/FGFR in fibroblast-derived growth stimulation and lapatinib-resistance of esophageal squamous cell carcinoma. BMC Cancer. 2015 Feb 25;15:82. doi: 10.1186/s12885-015-1065-8. PMID: 25884729; PMCID: PMC4345039.
[6] Bao Y, Gabrielpillai J, Dietrich J, Zarbl R, Strieth S, Schröck F, Dietrich D. Fibroblast growth factor (FGF), FGF receptor (FGFR), and cyclin D1 (CCND1) DNA methylation in head and neck squamous cell carcinomas is associated with transcriptional activity, gene amplification, human papillomavirus (HPV) status, and sensitivity to tyrosine kinase inhibitors. Clin Epigenetics. 2021 Dec 21;13(1):228. doi: 10.1186/s13148-021-01212-4. PMID: 34933671; PMCID: PMC8693503.
[7] Boichuk S, Dunaev P, Mustafin I, Mani S, Syuzov K, Valeeva E, Bikinieva F, Galembikova A. Infigratinib (BGJ 398), a Pan-FGFR Inhibitor, Targets P-Glycoprotein and Increases Chemotherapeutic-Induced Mortality of Multidrug-Resistant Tumor Cells. Biomedicines. 2022 Mar 3;10(3):601. doi: 10.3390/biomedicines10030601. PMID: 35327403; PMCID: PMC8945560.
[8] Yan C, Niu Y, Li F, Zhao W, Ma L. System analysis based on the pyroptosis-related genes identifies GSDMC as a novel therapy target for pancreatic adenocarcinoma. J Transl Med. 2022 Oct 5;20(1):455. doi: 10.1186/s12967-022-03632-z. Erratum in: J Transl Med. 2025 Oct 20;23(1):1130. doi: 10.1186/s12967-025-07200-z. PMID: 36199146; PMCID: PMC9533512.
[9] Shi T, Hu Z, Tian L, Yang Y. Advances in lung adenocarcinoma: A novel perspective on prognoses and immune responses of CENPO as an oncogenic superenhancer. Transl Oncol. 2023 Aug;34:101691. doi: 10.1016/j.tranon.2023.101691. Epub 2023 May 17. PMID: 37207381; PMCID: PMC10209335.
[10] Lian K, Yang W, Yang R, Xu X, Guan J. Novel Insights into Schizophrenia Treatment: Comprehensive Analysis Unveiling FGFR1 as a Promising Druggable Gene. Mol Neurobiol. 2025 Dec;62(12):15550-15563. doi: 10.1007/s12035-025-05221-9. Epub 2025 Jul 21. PMID: 40685484; PMCID: PMC12559107.
- 1. Soher Nagi Jayash, Thomas Duff, et al. "Osteoblasts sense extracellular levels of phosphate to control the local expression of phosphatases for matrix mineralisation." Volume 26, September 2025, 101863
- 2. Cheukyau Luk, Katherine I. Bridge, et al. "Paracrine role of endothelial IGF-1 receptor in depot-specific adipose tissue adaptation in male mice." Nat Commun. 2025 Jan 2;16(1):170. PMID: 39747815
- 3. Abeer Alghamdi, Hussah Alobaid, et al."Inhibitory effect of PD173074 drug on DMBA-induced mammary carcinoma in female Swiss albino mice." January 2025 Tropical Journal of Pharmaceutical Research 23(12):1983-1989
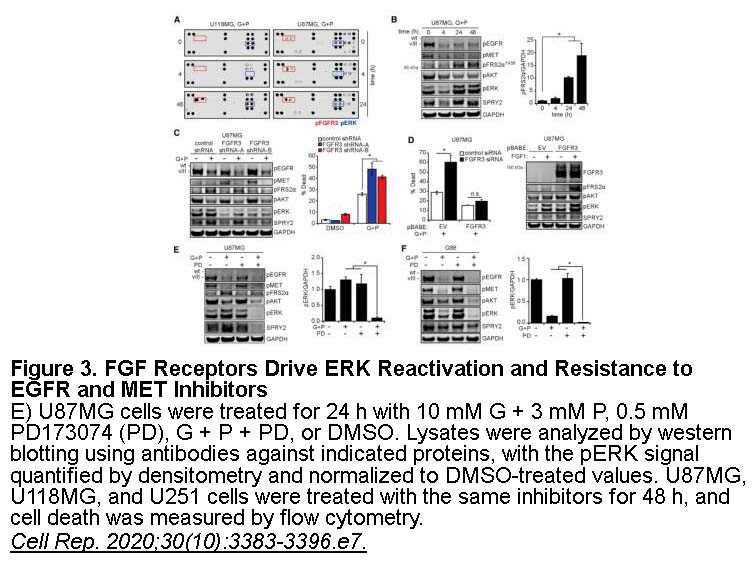
- 4. Day EK, Sosale NG, et al. "Glioblastoma Cell Resistance to EGFR and MET Inhibition Can Be Overcome via Blockade of FGFR-SPRY2 Bypass Signaling." Cell Rep. 2020;30(10):3383-3396.e7. PMID:32160544
- 5. Chen P, Zhang H, et al. "Basic Fibroblast Growth Factor Reduces Permeability and Apoptosis of Human Brain Microvascular Endothelial Cells in Response to Oxygen and Glucose Deprivation Followed by Reoxygenation via the Fibroblast Growth Factor Receptor 1 (FGFR1)/ERK Pathway." Med Sci Monit. 2019 Sep 25;25:7191-7201. PMID:31551405
| Physical Appearance | A solid |
| Storage | Store at 4°C |
| M.Wt | 523.67 |
| Cas No. | 219580-11-7 |
| Formula | C28H41N7O3 |
| Synonyms | PD 173074,PD-173074 |
| Solubility | ≥26.18 mg/mL in DMSO; insoluble in H2O; ≥108.4 mg/mL in EtOH with ultrasonic |
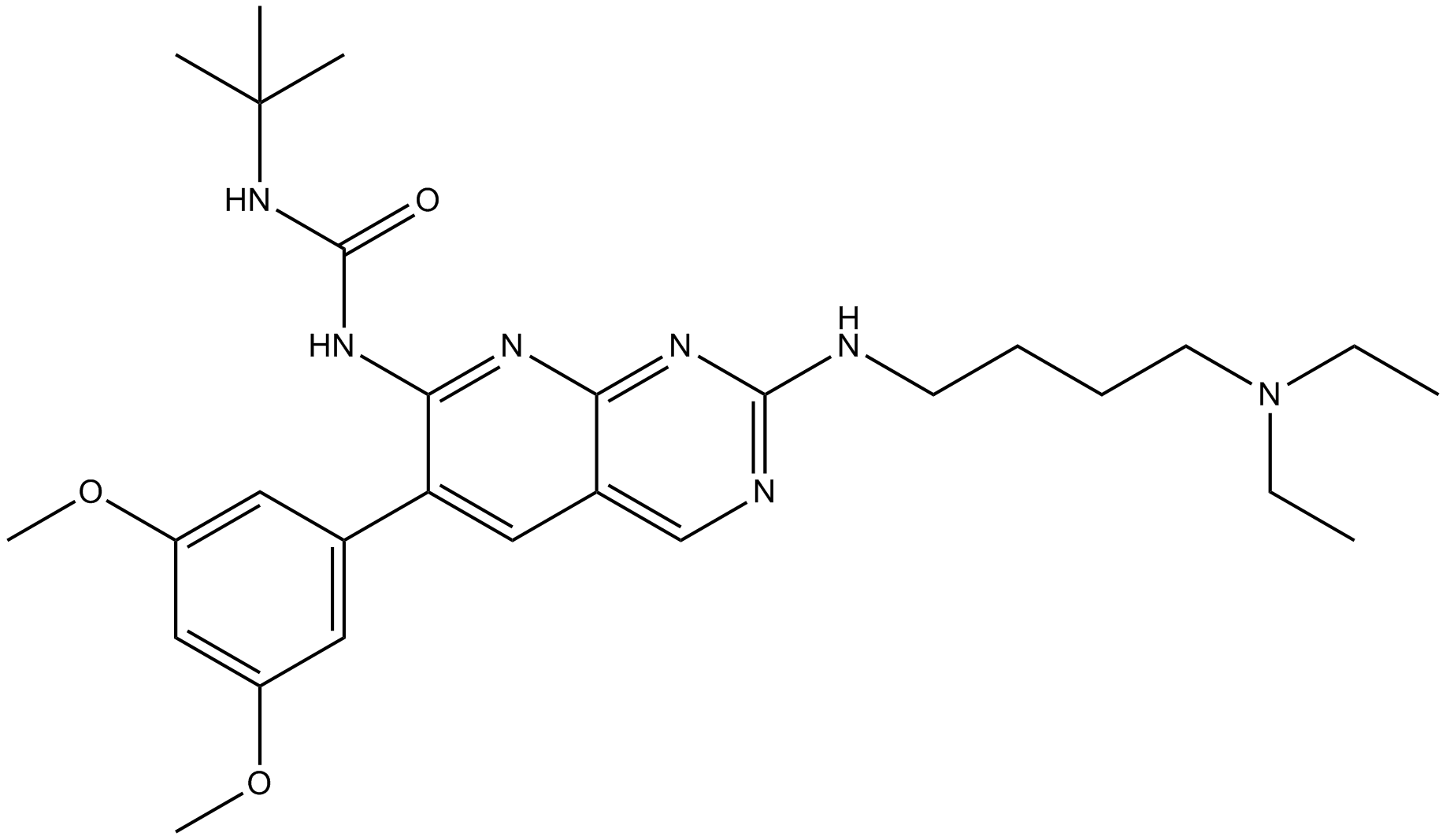
| Chemical Name | 1-tert-butyl-3-[2-[4-(diethylamino)butylamino]-6-(3,5-dimethoxyphenyl)pyrido[2,3-d]pyrimidin-7-yl]urea |
| Canonical SMILES | CCN(CC)CCCCNC1=NC2=NC(=C(C=C2C=N1)C3=CC(=CC(=C3)OC)OC)NC(=O)NC(C)(C)C |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Kinase experiment [1]: | |
|
In vitro kinase inhibition assays |
Assays using the full-length FGFR-1 kinase were performed in a total volume of 100 μL containing 25 mM HEPES buffer (pH 7.4), 150 mM NaCl, 10 mM MnCl2, 0.2 mM sodium orthovanadate, 750 μg/mL concentration of a random copolymer of glutamic acid and tyrosine (4:1), various concentrations of PD173074 and 60 to 75 ng of enzyme. The reaction was initiated by the addition of [γ-32P]ATP (5 μM ATP containing 0.4 μCi of [γ-32P]ATP per incubation), and samples were incubated at 25 °C for 10 mins. The reaction was terminated by the addition of 30% trichloroacetic acid and the precipitation of material onto glass-fiber filter mats. Filters were washed three times with 15% trichloroacetic acid, and the incorporation of [32P] into the glutamate tyrosine polymer substrate was determined by counting the radioactivity retained on the filters in a Wallac 1250 betaplate reader. |
| Cell experiment [1]: | |
|
Cell lines |
NIH 3T3 cells |
|
Preparation method |
The solubility of this compound in DMSO is >10 mM. General tips for obtaining a higher concentration: Please warm the tube at 37 °C for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20 °C for several months. |
|
Reaction Conditions |
0 ~ 1000 nM; 5 mins |
|
Applications |
PD 173074 dose-dependently inhibited autophosphorylation of FGFR1, with an IC50 value in the range of 1 ~ 5 nM. In addition, PD 173074 inhibited autophosphorylation of VEGFR2 with an IC50 value of 100 ~ 200 nM. |
| Animal experiment [1]: | |
|
Animal models |
Swiss Webster mice with induced corneal angiogenesis |
|
Dosage form |
1 or 2 mg/kg/day; i.p. |
|
Applications |
At the dose of 1 or 2 mg/kg, PD 173074 significantly inhibited angiogenesis induced by either FGF or VEGF in a dose-dependent manner. Besides, it showed no apparent toxicity. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Mohammadi M, Froum S, Hamby JM, Schroeder MC, Panek RL, Lu GH, Eliseenkova AV, Green D, Schlessinger J, Hubbard SR. Crystal structure of an angiogenesis inhibitor bound to the FGF receptor tyrosine kinase domain. EMBO J. 1998 Oct 15;17(20):5896-904. |
|
| Description | PD173074 is a potent inhibitor of FGFR1 with IC50 of ~25 nM and also inhibits VEGFR2 with IC50 of 100-200 nM, ~1000-fold selective for FGFR1 than PDGFR and c-Src. | |||||
| Targets | FGFR1 | VEGFR2 | ||||
| IC50 | ~25 nM | 100-200 nM | ||||
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data
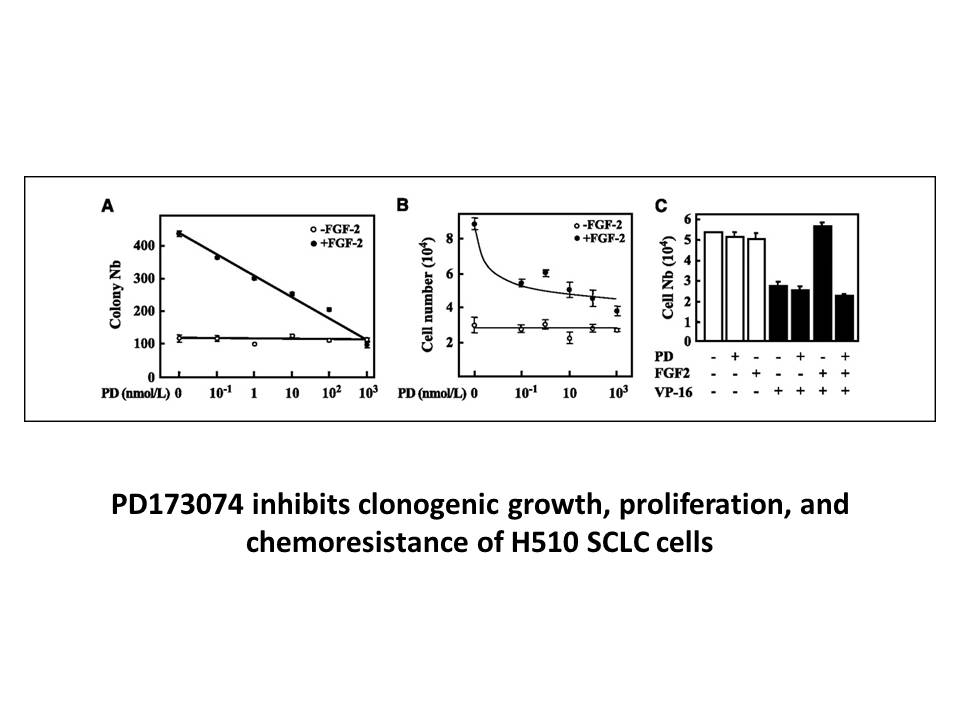
Related Biological Data
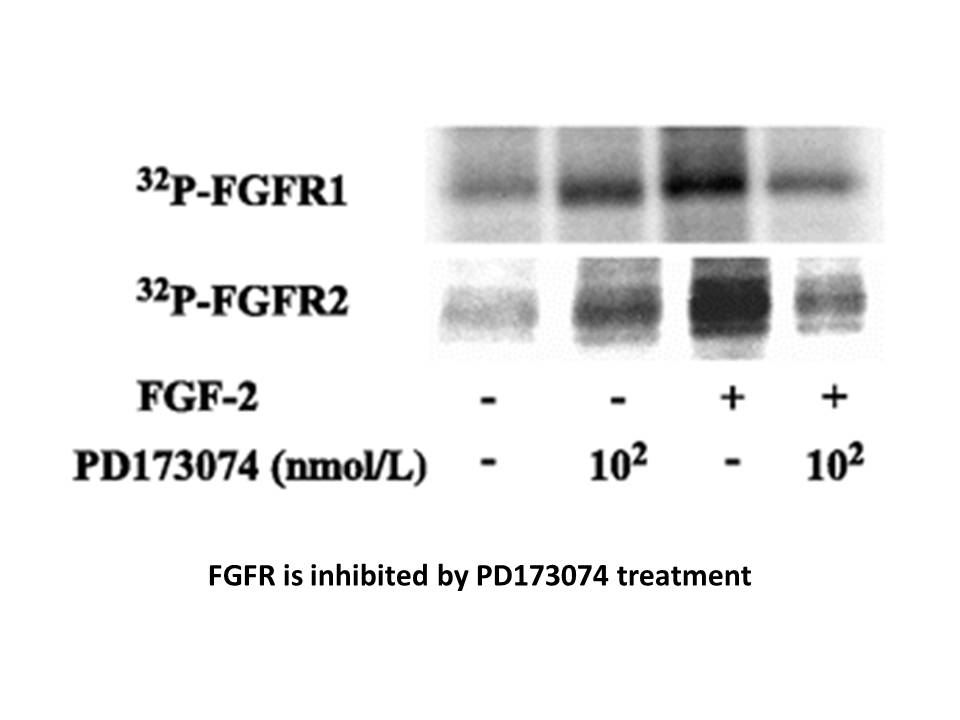
Related Biological Data