IOX2(Glycine)
IOX2 is a potent and selective inhibitor of HIF-1α prolyl hydroxylase-2 (PHD2) with an IC50 of 21 nM for PHD2/ELGN-1 in a cell-free assay, IOX2 has shown >100-fold selectivity over JMJD2A, JMJD2C, JMJD2E, JMJD3, or the 2OG oxygenase FIH [1].
PHDs consist three isoforms, named PHD1, PHD2 and PHD3. PHDs are dioxygenases that utilize oxygen as co-substrate providing the molecular basis for the oxygen-sensing function of these enzymes. It has been reported that PHDs are strikingly sensitive to graded levels of hypoxia, mirroring the progressive increases in HIF-1α protein and DNA binding activity that are observed when cells are exposed to gradual hypoxia [2]. In RCC4 cells, IOX2 inhibited HIF-1α hydroxylation at 50 μM [1].
References:
[1] Murray J K, Balan C, Allgeier A M, et al. Dipeptidyl-quinolone derivatives inhibit hypoxia inducible factor-1α prolyl hydroxylases-1,-2, and-3 with altered selectivity[J]. Journal of combinatorial chemistry, 2010, 12(5): 676-686.
[2] Berra E, Benizri E, Ginouvès A, et al. HIF prolyl‐hydroxylase 2 is the key oxygen sensor setting low steady‐state levels of HIF‐1α in normoxia[J]. The EMBO journal, 2003, 22(16): 4082-4090.
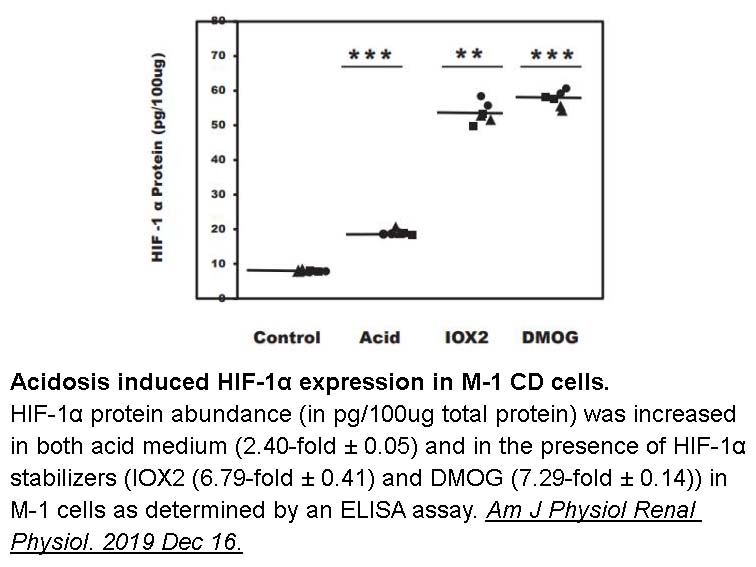
- 1. Peng H, Purkerson JM, et al. "Acidosis induces antimicrobial peptide expression and resistance to uropathogenic E coli infection in kidney collecting duct cells via HIF-1α." Am J Physiol Renal Physiol. 2019 Dec 16. PMID:31841391
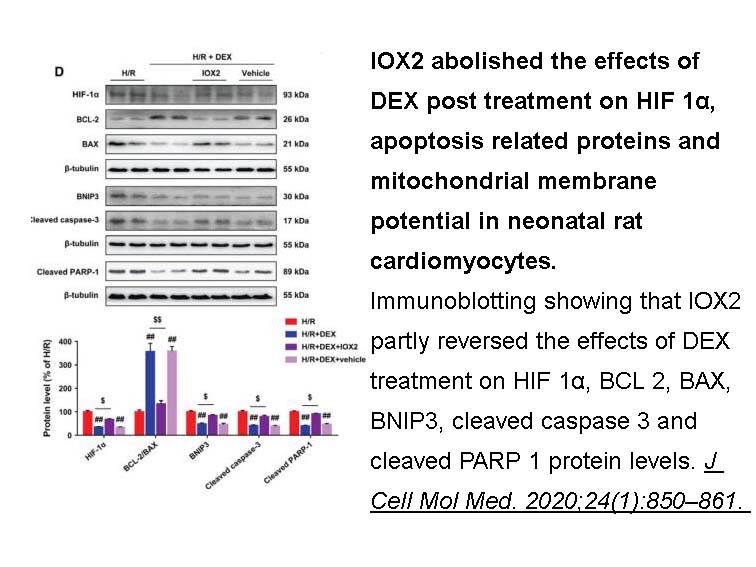
- 2. Peng K, Chen WR, et al. "Dexmedetomidine post-treatment attenuates cardiac ischaemia/reperfusion injury by inhibiting apoptosis through HIF-1α signalling." J Cell Mol Med. 2020;24(1):850–861. PMID:31680420
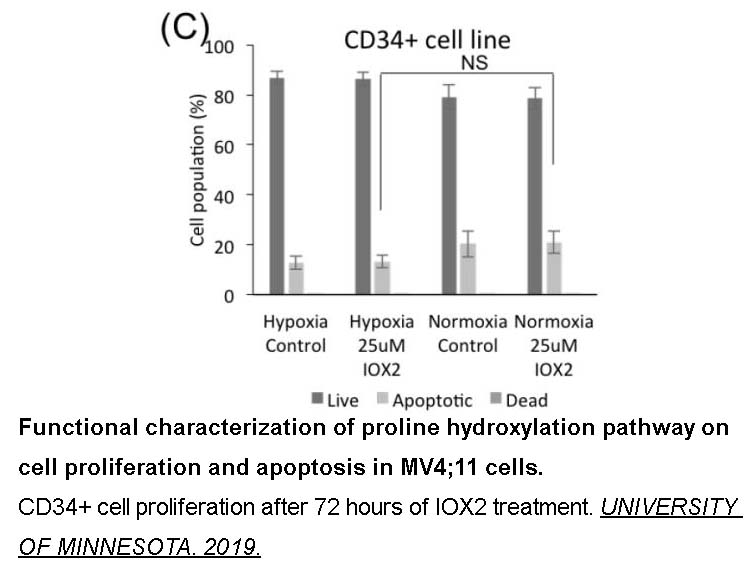
- 3. LUKE ERBER. "Functional Proteomics Analysis To Discover And Characterize Oxygen-Dependent Cellular Pathways." UNIVERSITY OF MINNESOTA. 2019.
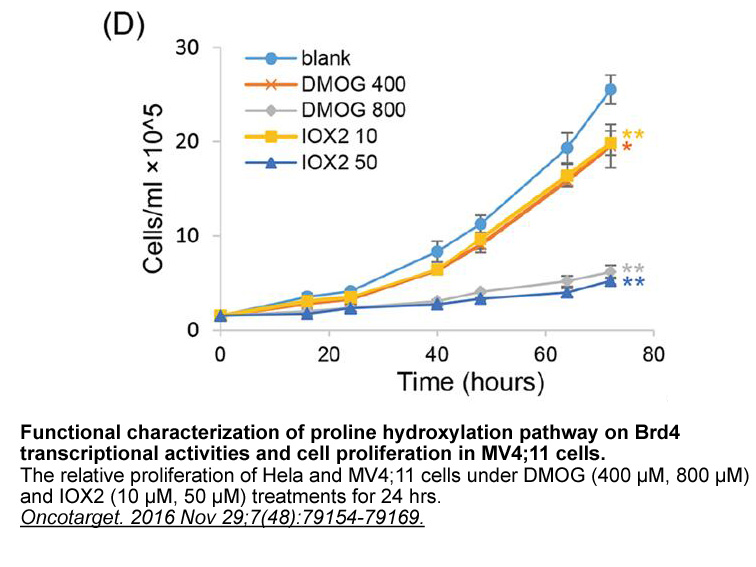
- 4. Zhou T, Erber L, et al. "Proteomic analysis reveals diverse proline hydroxylation-mediated oxygen-sensing cellular pathways in cancer cells." Oncotarget. 2016 Nov 29;7(48):79154-79169. PMID:27764789
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 352.34 |
| Cas No. | 931398-72-0 |
| Formula | C19H16N2O5 |
| Solubility | insoluble in EtOH; insoluble in H2O; ≥17.6 mg/mL in DMSO |
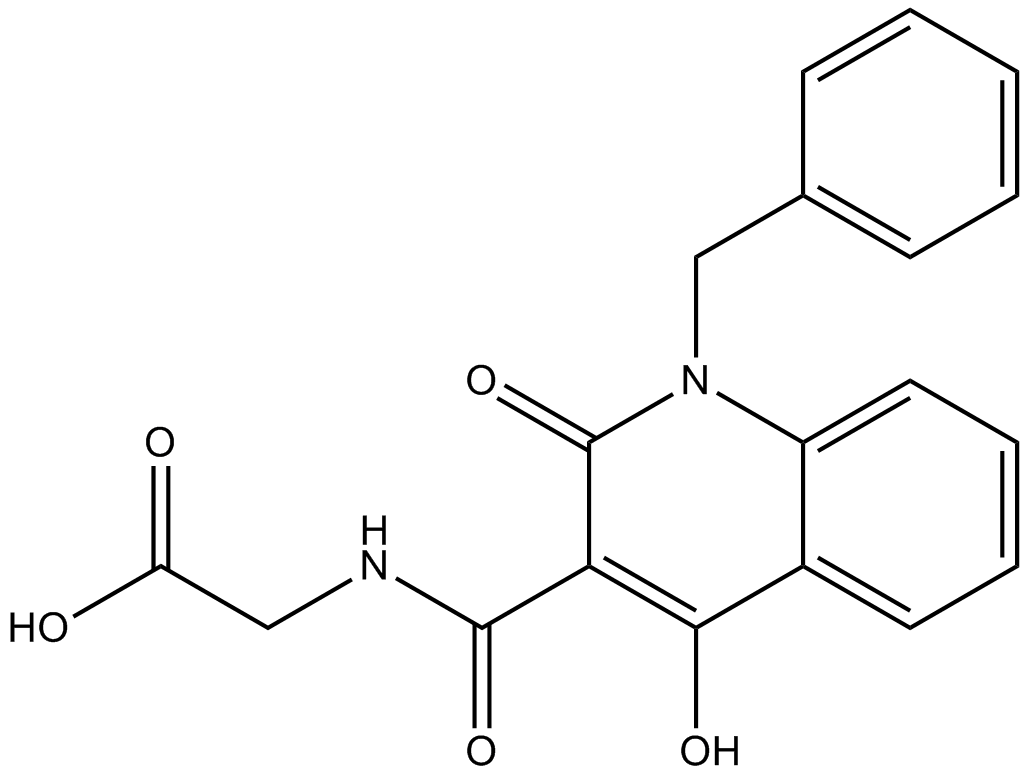
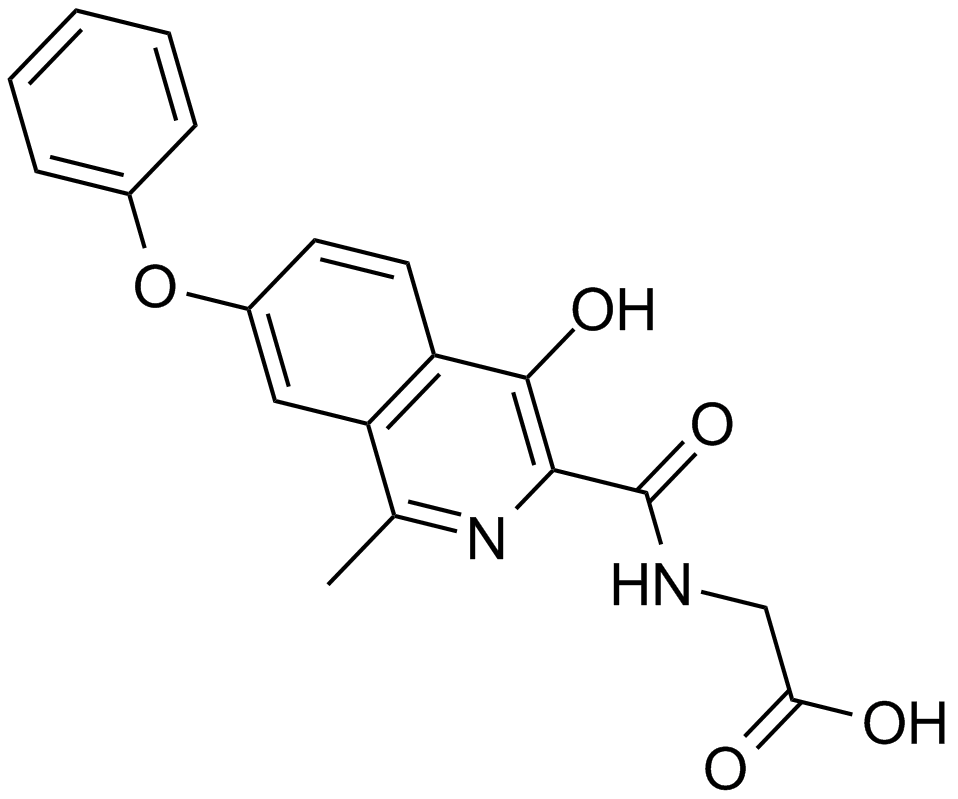
| Chemical Name | 2-[(1-benzyl-4-hydroxy-2-oxoquinoline-3-carbonyl)amino]acetic acid |
| SDF | Download SDF |
| Canonical SMILES | C1=CC=C(C=C1)CN2C3=CC=CC=C3C(=C(C2=O)C(=O)NCC(=O)O)O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Kinase experiment [1]: | |
|
Alpha screen assay |
All reagents were diluted in 50 mM HEPES, 0.1 % BSA, pH 7.5 supplemented with 0.01 % Tween20 and allowed to equilibrate to room temperature prior to addition to plates. Catalytic turnover assays were run in 10 μM volumes in low-volume 384-well plates at RT. The reaction consisted of enzyme (0.5 ~ 25 nM), biotinylated substrate peptide (30 ~ 1000 nM), Fe(II) (1 ~ 10 μM), Ascorbate (100 μM), 2OG (5 ~ 40 μM) and run at RT. EDTA was used to quench the reaction (5 μM) and AlphaScreen donor (Streptavidin-conjugated) and acceptor (ProteinA-conjugated) beads preincubated with peptide product antibodies were added (5 μM). Plates were foil-sealed to protect from light, incubated at room temperature for 60 mins and read on a PHERAstar FS plate reader using an AlphaScreen 680 excitation/570 emission filter set. The final bead concentration in 20 μM reaction was 20 μM/mL. IC50 values were calculated in Prism 5. |
| Cell experiment [1]: | |
|
Cell lines |
RCC4 cells |
|
Preparation method |
The solubility of this compound in DMSO is > 10 mM. General tips for obtaining a higher concentration: Please warm the tube at 37 °C for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20 °C for several months. |
|
Reaction Conditions |
50 μM |
|
Applications |
In RCC4 cells, IOX2 inhibited HIF-1α hydroxylation at 50 μM. |
|
References: [1]. Murray JK1, Balan C, Allgeier AM et al. Dipeptidyl-quinolone derivatives inhibit hypoxia inducible factor-1α prolyl hydroxylases-1, -2, and -3 with altered selectivity. J Comb Chem. 2010 Sep 13;12(5):676-86. |
|
| Description | IOX2 is a potent inhibitor of HIF-1α prolyl hydroxylase-2 (PHD2) with IC50 of 21 nM. | |||||
| Targets | PHD2 | |||||
| IC50 | 21 nM | |||||
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data
Related Biological Data
Related Biological Data
Related Biological Data