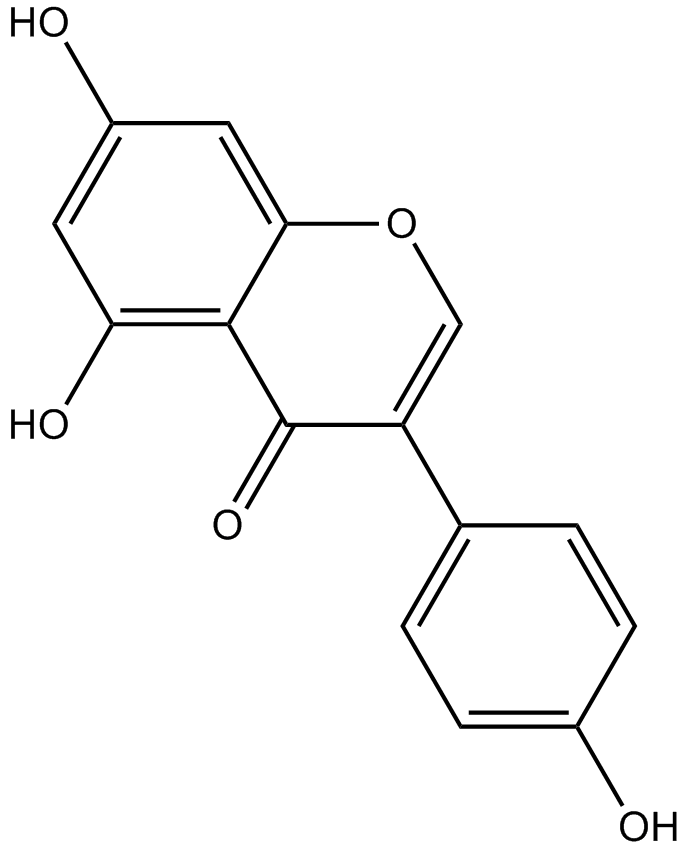
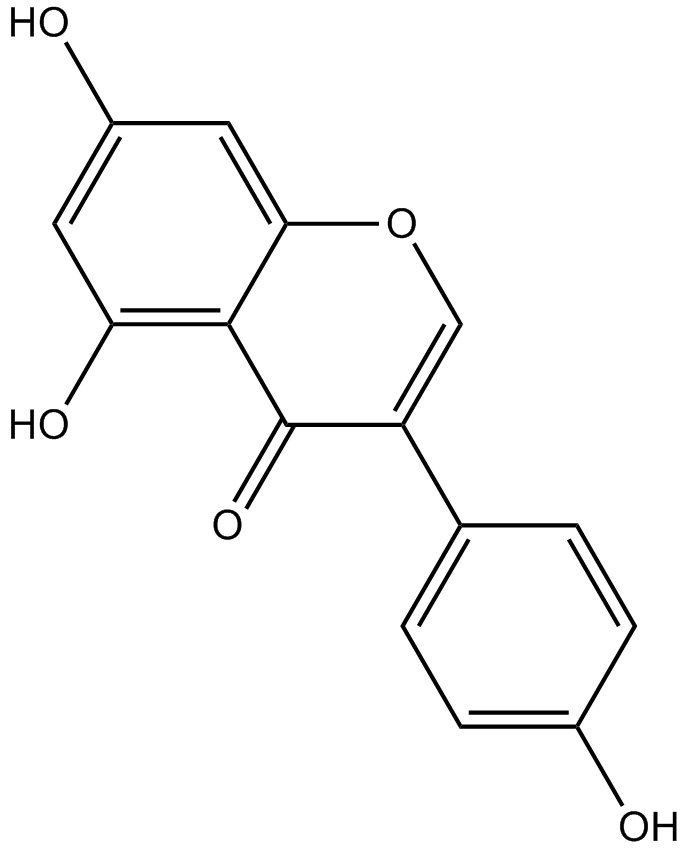
Genistein
Genistein is a highly specific inhibitor of tyrosine kinase with IC50 value of ~ 8 μM [1].
Tyrosine protein kinase activities are known to be associated with oncogene products of the retroviral “src” gene family and with various cellular growth factor receptors including epidermal growth factor (EGF), platelet derived growth factor (PDGF), insulin and insulin-like growth factors, which play a key role in tumorogenesis. Thus, inhibitors of protein tyrosine kinase activity might represent a new class of antitumor agents [2].
In NIH-3T3 cells, Genistein blocked the mitogenic effect mediated by EGF with IC50 value of 12 μM or by insulin with IC50 value of 19 μM. In NIH-3T3 cells treated with EGF, Genistein partially prevented the increase of S6 kinase activity at 6 μM, with the maximal effect observed at 15 μM [2].
In a chemically (N-methylnitrosourea) induced prostate cancer rat model, Genistein (0, 25 or 250 mg/kg, p.o.) in the diet dose-dependently inhibited the development of invasive adenocarcinomas. In a transgenic mouse model that spontaneously developed prostate cancer, Genistein (0, 100, 250 or 500 mg/kg, p.o.) in the diet lowered the incidence of poorly differentiated prostatic adenocarcinomas in a dose-dependent manner [3].
[1]. Baltuch G H, Yong V W. Signal transduction for proliferation of glioma cells in vitro occurs predominantly through a protein kinase C-mediated pathway. Brain Research, 1996, 710(1-2): 143-149.
[2]. Linassier C, Pierre M, Le Pecq J B, et al. Mechanisms of action in NIH-3T3 cells of genistein, an inhibitor of EGF receptor tyrosine kinase activity. Biochemical Pharmacology, 1990, 39(1): 187-193.
[3]. Lamartiniere C A, Cotroneo M S, Fritz W A, et al. Genistein chemoprevention: timing and mechanisms of action in murine mammary and prostate. Journal of Nutrition, 2002, 132(3): 552S-558S.
- 1. Yuxin Zhang, Jie Zhou, et al. "Modulating tumor-stromal crosstalk via a redox-responsive nanomedicine for combination tumor therapy." J Control Release. 2023 Apr:356:525-541. PMID: 36918084
- 2. Zhou J, Ma S, et al. "Virus-Inspired Mimics: Dual-pH-Responsive Modular Nanoplatforms for Programmable Gene Delivery without DNA Damage with the Assistance of Light." ACS Appl Mater Interfaces. 2020;12(20):22519-22533. PMID:32329598
| Storage | Store at -20°C |
| M.Wt | 270.24 |
| Cas No. | 446-72-0 |
| Formula | C15H10O5 |
| Solubility | ≥13.5 mg/mL in DMSO; insoluble in H2O; ≥2.59 mg/mL in EtOH with gentle warming |
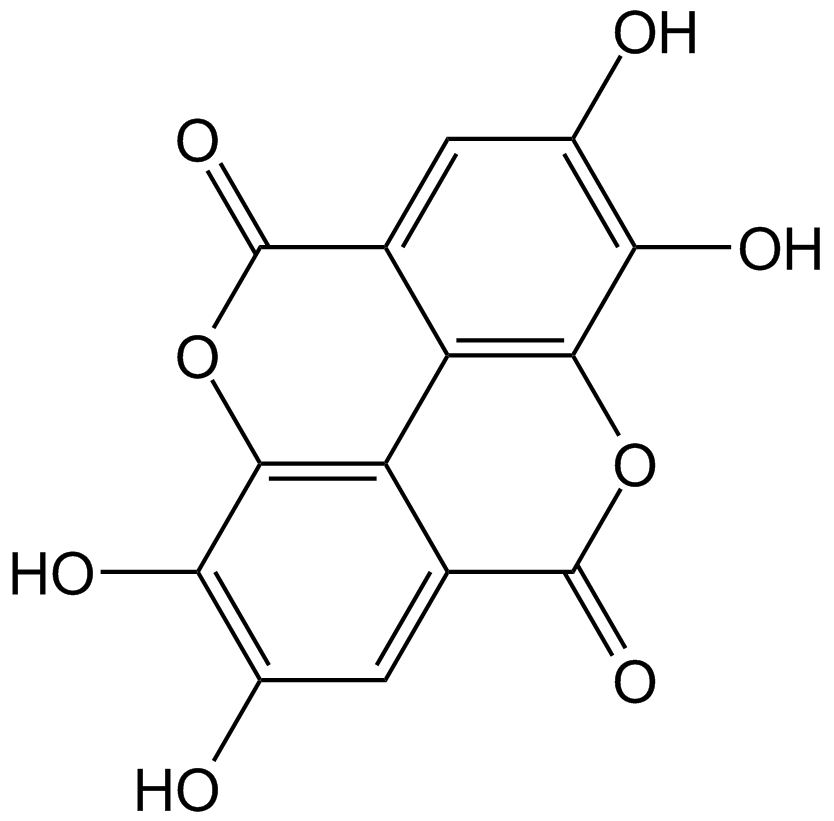
| Chemical Name | 5,7-dihydroxy-3-(4-hydroxyphenyl)chromen-4-one |
| SDF | Download SDF |
| Canonical SMILES | C1=CC(=CC=C1C2=COC3=CC(=CC(=C3C2=O)O)O)O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1]: | |
|
Cell lines |
NIH-3T3 cells |
|
Preparation method |
The solubility of this compound in DMSO is > 55.6 mg/mL. General tips for obtaining a higher concentration: Please warm the tube at 37 °C for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below - 20 °C for several months. |
|
Reacting condition |
0 ~ 1000 μM |
|
Applications |
NIH-3T3 cell growth was inhibited after Genistein treatment at concentrations of 4 ~ 75 μM for 72 hrs. At concentrations below 40 μM, Genistein-treated cells recovered growth after drug withdrawal. In contrast, at 75 μM or above, Genistein-treated cells were unable to recover growth. Cytotoxicity assessed by cloning efficiency measured after 3 hrs of Genistein treatment showed an ED50 value of 35 μM. |
| Animal experiment [2]: | |
|
Animal models |
Female SD rats |
|
Dosage form |
0, 25 and 250 mg Genistein/kg AIN-76A; p.o. |
|
Applications |
In female SD rats, Genistein dose-dependently suppressed dimethylbenz[a]anthracene (DMBA)-induced mammary tumor development. Rats exposed to 25 and 250 mg Genistein/kg AIN-76A showed 7.1 and 4.3 mammary tumors, respectively. Dietary Genistein protected against mammary tumors through regulating specific sex steroid receptors as well as growth factor signaling pathways. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Linassier C, Pierre M, Le Pecq JB, Pierre J. Mechanisms of action in NIH-3T3 cells of genistein, an inhibitor of EGF receptor tyrosine kinase activity. Biochem Pharmacol. 1990 Jan 1;39(1):187-93. [2]. Lamartiniere CA, Cotroneo MS, Fritz WA, Wang J, Mentor-Marcel R, Elgavish A. Genistein chemoprevention: timing and mechanisms of action in murine mammary and prostate. J Nutr. 2002 Mar;132(3):552S-558S. |
|
Quality Control & MSDS
- View current batch:
Chemical structure