Ganetespib (STA-9090)
Ganetespib (STA-9090; CAS 888216-25-9) is a small-molecule inhibitor targeting heat shock protein 90 (Hsp90). It binds competitively to the ATP-binding pocket located at the N-terminal region of Hsp90, disrupting its chaperone activity and subsequently causing degradation of client proteins involved in tumor growth and survival. Structurally distinct from geldanamycin-derived inhibitors, Ganetespib features a triazolone moiety. Preclinical studies have demonstrated antitumor activity across various cancer cell types, including lung, prostate, colon, breast cancers, melanoma, and leukemia, highlighting its therapeutic potential for cancer research.
- 1. Jaewon Song, Li Zhang, et al. "Norovirus co-opts NINJ1 for selective protein secretion." Sci Adv. 2025 Feb 28;11(9):eadu7985. PMID: 40020060
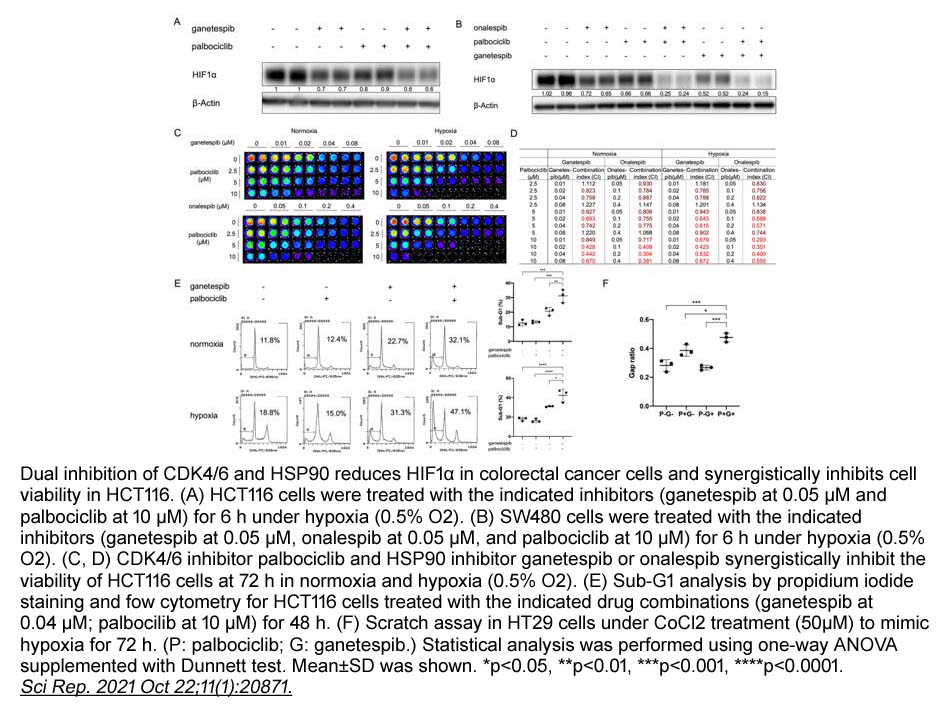
- 2. Shuai Zhao, Lanlan Zhou, et al. "Anti-cancer efficacy including Rb-deficient tumors and VHL-independent HIF1α proteasomal destabilization by dual targeting of CDK1 or CDK4/6 and HSP90." Sci Rep. 2021 Oct 22;11(1):20871. PMID: 34686682
- 3. Diwei Zheng, Weihai Liu, et al. "AHA1 upregulates IDH1 and metabolic activity to promote growth and metastasis and predicts prognosis in osteosarcoma." Signal Transduct Target Ther. 2021 Jan 20;6(1):25. PMID: 33468990
- 4. White SM, Avantaggiati ML, et al. "YAP/TAZ Inhibition Induces Metabolic and Signaling Rewiring Resulting in Targetable Vulnerabilities in NF2-Deficient Tumor Cells." Dev Cell. 2019 May 6;49(3):425-443.e9. PMID: 31063758
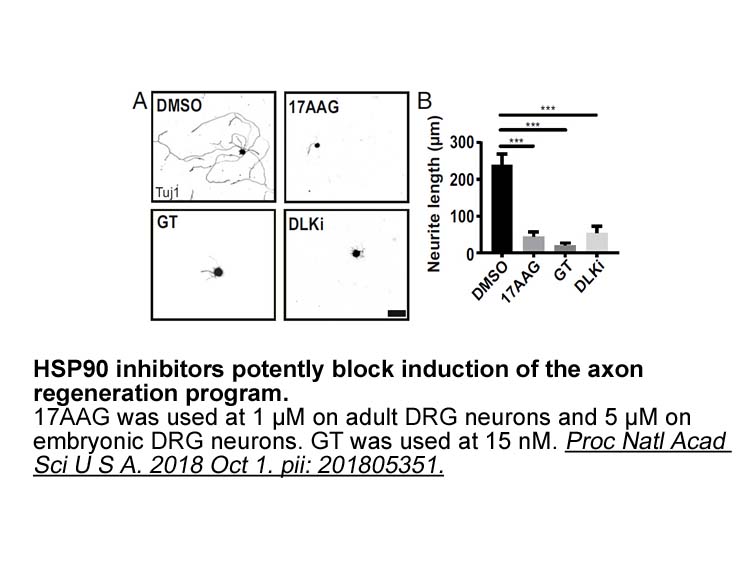
- 5. Karney-Grobe S, Russo A, et al. "HSP90 is a chaperone for DLK and is required for axon injury signaling." Proc Natl Acad Sci U S A. 2018 Oct 16;115(42):E9899-E9908. PMID: 30275300
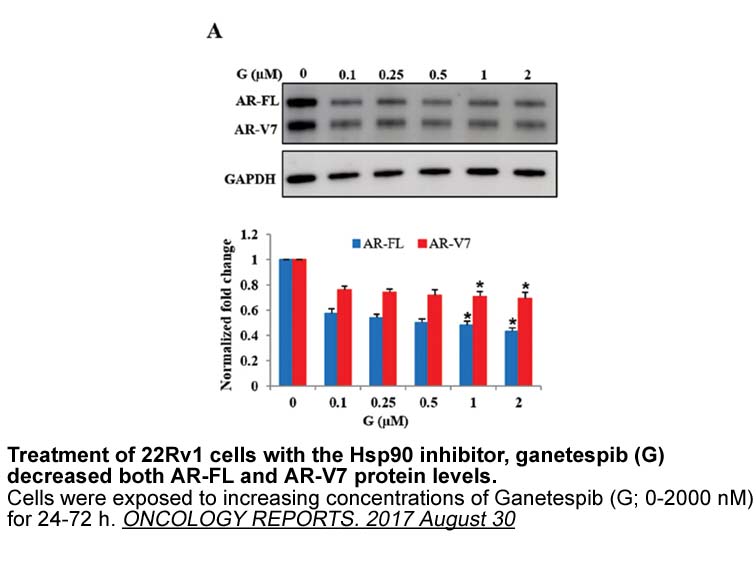
- 6. Khurana N, Kim H, et al. "Multimodal actions of the phytochemical sulforaphane suppress both AR and AR-V7 in 22Rv1 cells: Advocating a potent pharmaceutical combination against castration-resistant prostate cancer." Oncol Rep. 2017 Aug 30. PMID: 28901514
- 7. Li QQ, Hao JJ,et al. "Proteomic analysis of proteome and histone post-translational modifications in heat shock protein 90 inhibition-mediated bladder cancer therapeutics." Sci Rep. 2017 Mar 15;7(1):201. PMID: 28298630
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 364.4 |
| Cas No. | 888216-25-9 |
| Formula | C20H20N4O3 |
| Synonyms | STA9090, STA 9090 |
| Solubility | insoluble in H2O; ≥18.22 mg/mL in DMSO; ≥6.4 mg/mL in EtOH with gentle warming and ultrasonic |
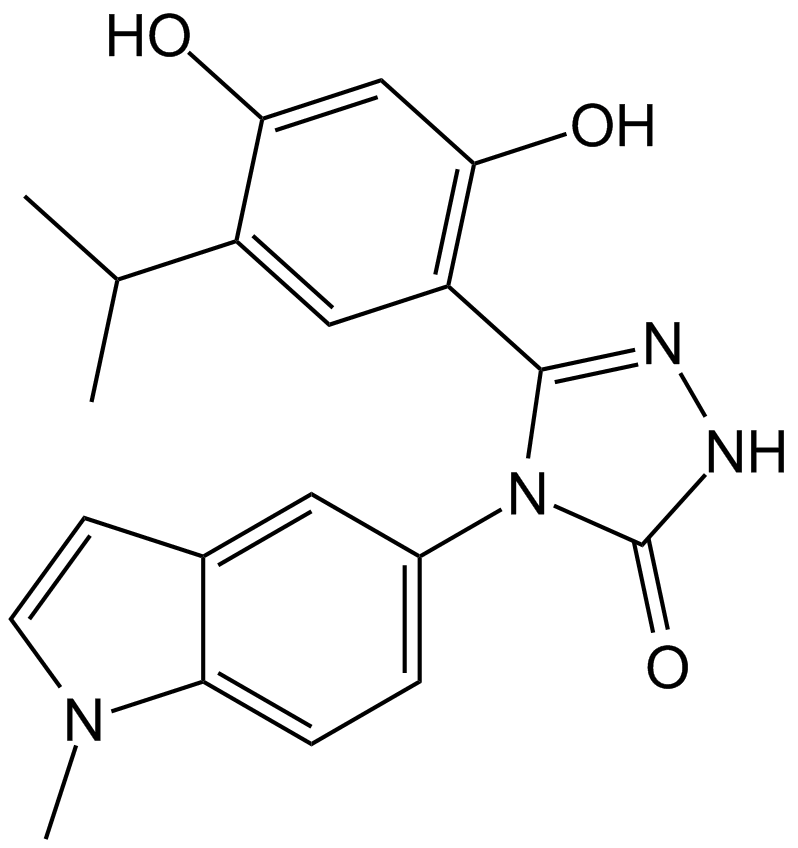
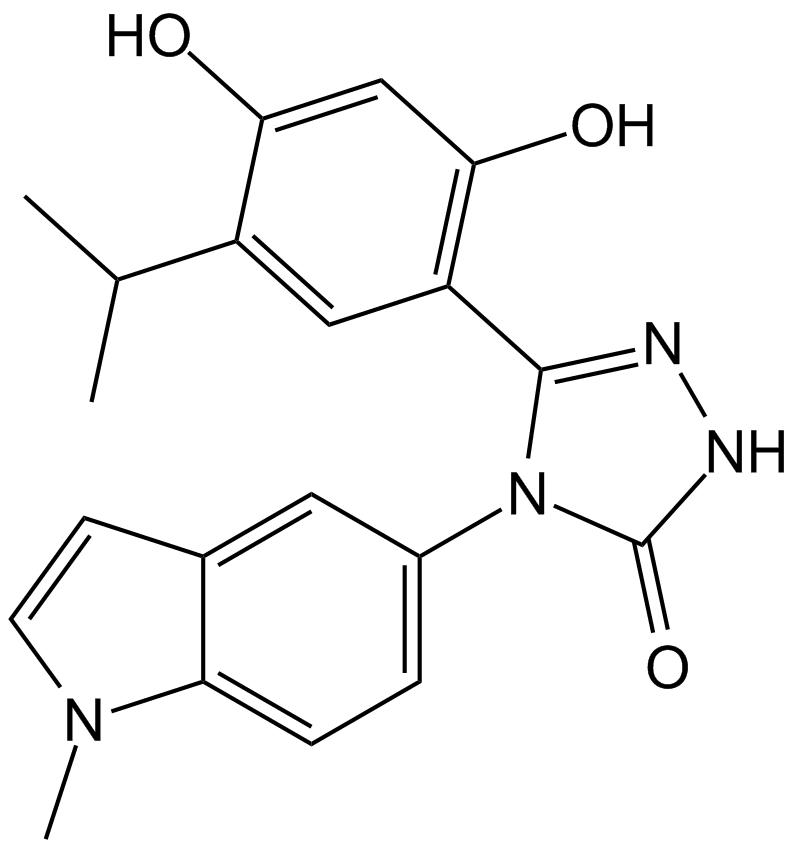
| Chemical Name | (5Z)-5-(4-hydroxy-6-oxo-3-propan-2-ylcyclohexa-2,4-dien-1-ylidene)-4-(1-methylindol-5-yl)-1,2,4-triazolidin-3-one |
| SDF | Download SDF |
| Canonical SMILES | CC(C)C(C(O)=CC1=O)=CC1=C(NNC1=O)N1c(cc1)cc2c1[n](C)cc2 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1]: | |
|
Cell lines |
NCI-H1975 and HCC827 cells |
|
Preparation method |
The solubility of this compound in DMSO is > 10 mM. General tips for obtaining a higher concentration: Please warm the tube at 37 °C for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20 °C for several months. |
|
Reaction Conditions |
0.001 ~ 1 μM; 5, 15 or 60 mins or 24 hrs |
|
Applications |
In NCI-H1975 and HCC827 cells, exposure to Ganetespib for only 60 mins resulted in cytotoxicity with IC50 values of 510 and 800 nM, respectively. In NCI-H1975 cells, a 5-minute exposure to Ganetespib still resulted in an IC50 value < 1 μM. |
| Animal experiment [1]: | |
|
Animal models |
SCID mice bearing NCI-H1395 NSCLC xenografts |
|
Dosage form |
150 mg/kg; i.v.; once weekly |
|
Applications |
Ganetespib induced NCI-H1395 tumor regression, with a T/C value of -49%. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Weiwen Ying, Zhenjian Du, Lijun Sun, Kevin P. Foley, David A. Proia, Ronald K. Blackman, Dan Zhou, Takayo Inoue, Noriaki Tatsuta, Jim Sang, Shuxia Ye, Jamie Acquaviva, Luisa Shin Ogawa, Yumiko Wada, James Barsoum, and Keizo Koya. Ganetespib, a unqiue triazolone-containing Hsp90 inhibitor, exhibits potent antitumor activity and a superior safety profile for cancer therapy. Mol Cancer Ther 2012; 11: 475-484. |
|
| Description | Ganetespib (STA-9090) is an inhibitor of HSP90 with IC50 of 4 nM in OSA 8 cells. | |||||
| Targets | HSP90 | |||||
| IC50 | 4 nM | |||||
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data
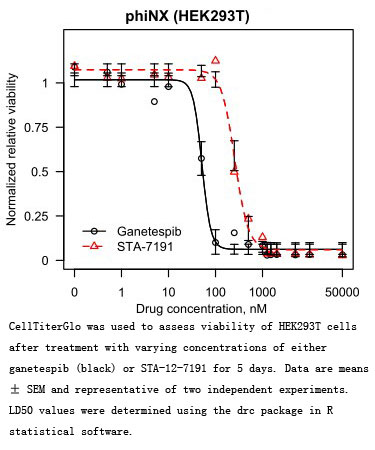
Related Biological Data
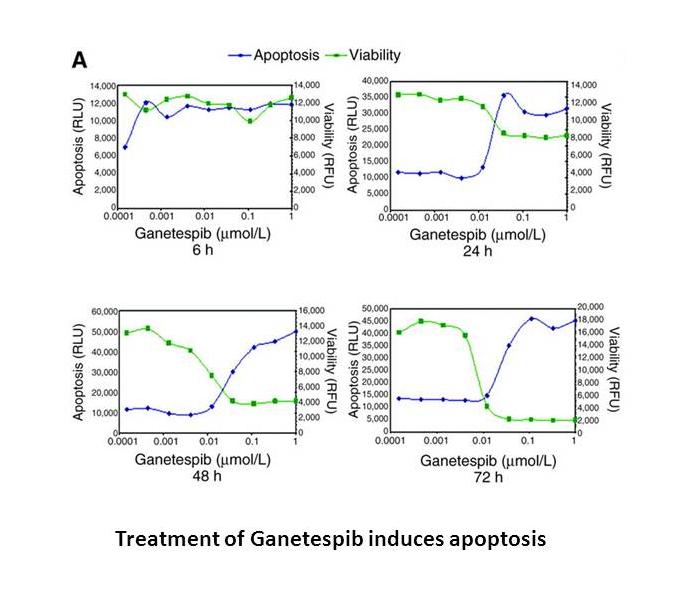
Related Biological Data
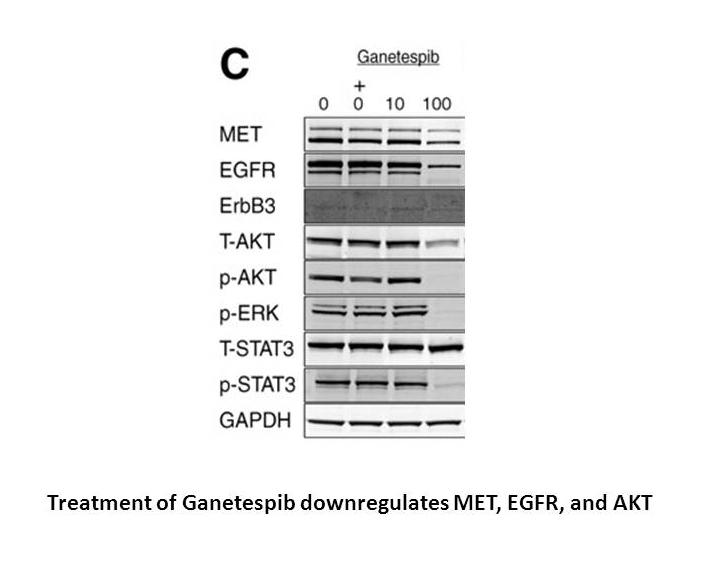
Related Biological Data
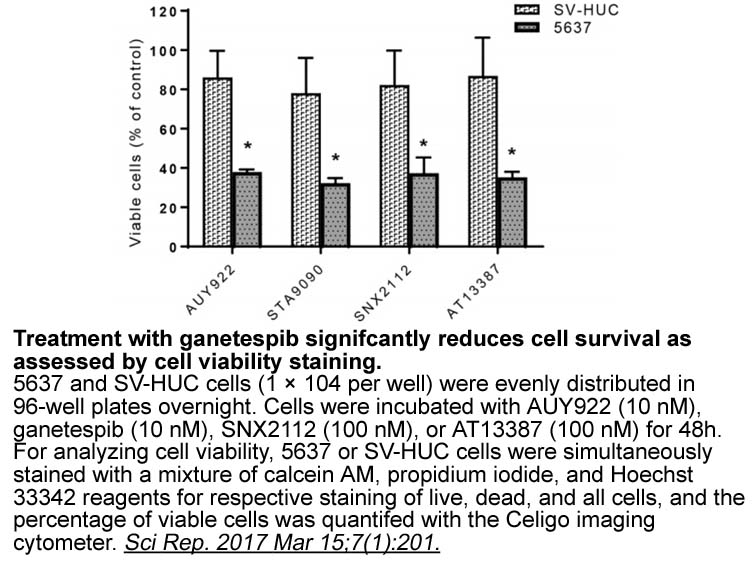
Related Biological Data
Related Biological Data
Related Biological Data