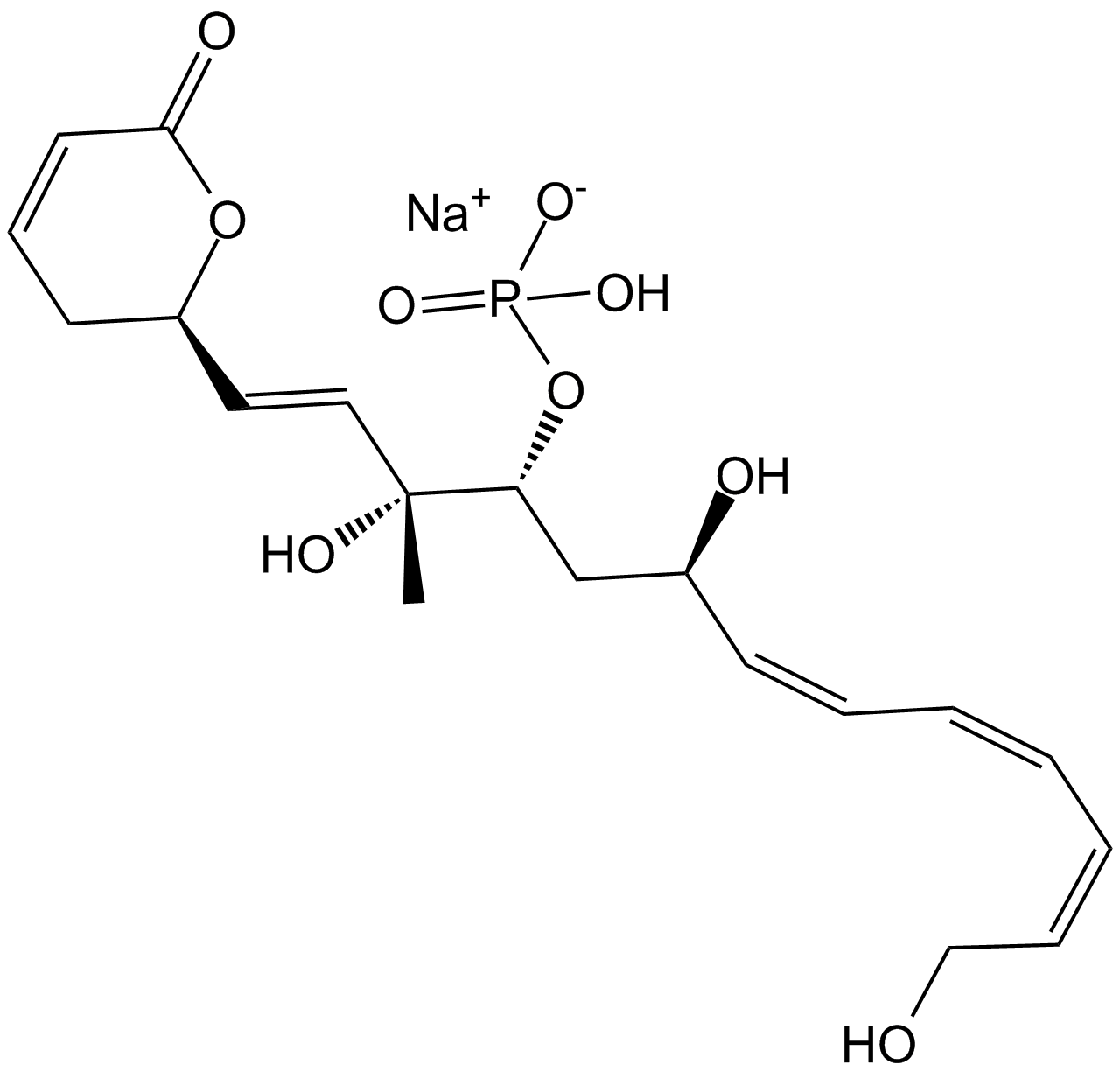
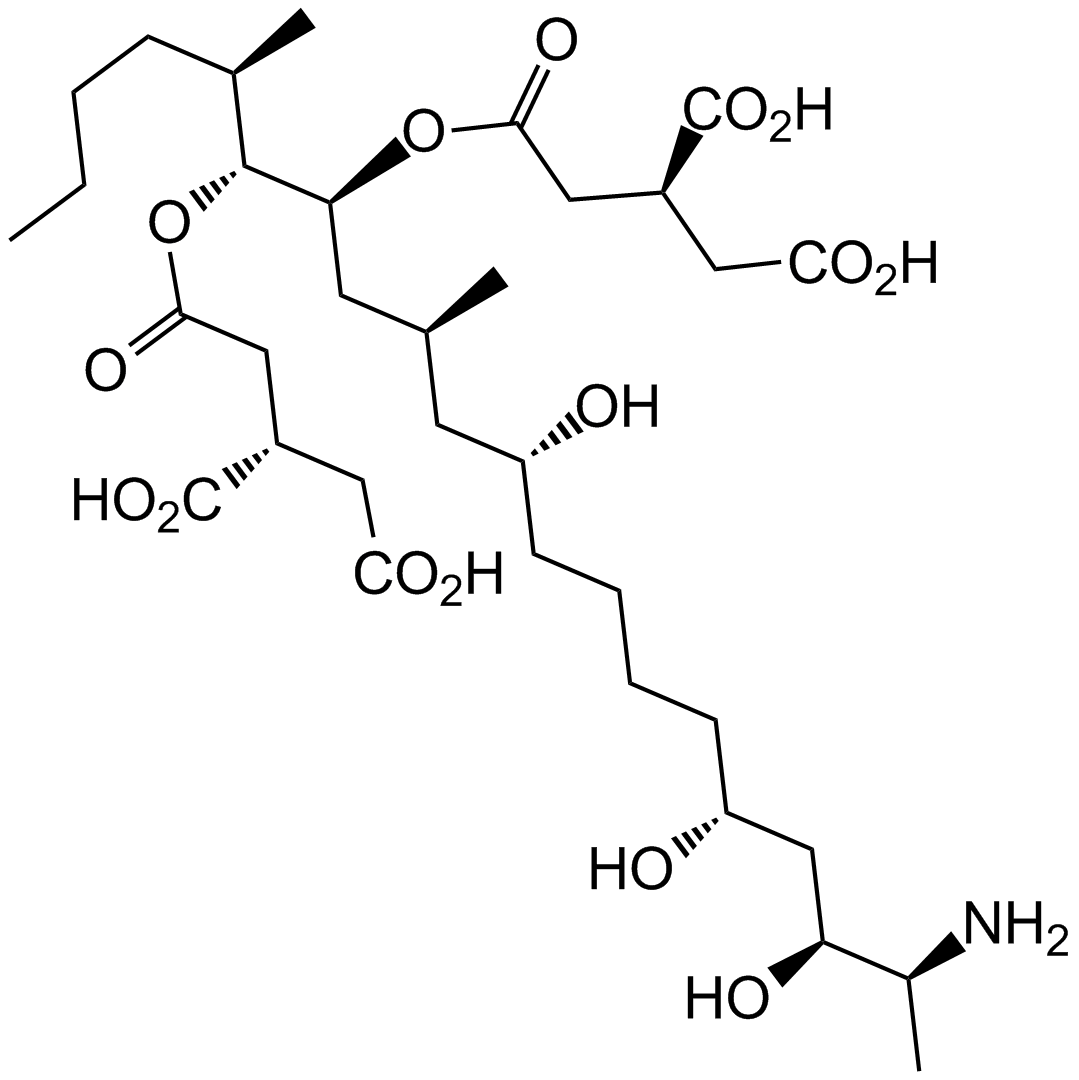
Fostriecin sodium salt
IC50: Inhibit protein phosphatase types 2A (PP2A) and 4 (PP4) intensively with an IC50 of 1.5 nM and 3 nM respectively. Inhibit topoisomerase II (Topo II) and protein phosphatase type 1 (PP1) slightly with an IC50 of 40 μM and 131 μM respectively.
Fostriecin, an anti-tumor antibiotic originates from Streptomyces pulveraceus, is considered to exert its activity by interfering the reversible phosphorylation of several important proteins in cell cycle. Fostriecin sodium salt also exhibits anti-tumor activity via suppressing enzymes including PP2A, PP4, PP1 and Topo II. [1]
In vitro: A human tumor cloning assay was adopted to investigate the antineoplastic activity of fostriecin. Variety of histologic tumor cells exposure to 10 mg/ml fostriecin resulted in 27% in vitro anticancer response. In another study using HeLa cells, by suppressing the Topo II catalytic activity, 0.22 mM fostriecin could lead to more than 90% inhibition of cancer cell DNA synthesis. [2,3]
In vivo: Fostriecin showed intensively cytotoxicity against a number of cancer cell lines and had a strong antitumor effect in animals. 10 mg/ml fostriecin could substantially suppress P388 and L1210 leukemias in mice. [2,3]
Clinical trial: Fostriecin reached clinical phase I study sponsored by the National Cancer Institute to investigate its antitumor activity in human. With dosages escalated from 2 to 20 mg/m2/day, fostriecin was administered as a 60-min intravenous (IV) infusion to 20 patients in cancer for 5 days. Although the trials were suspended due to the concerns about the storage stability of this naturally originated compound, this discovery ignited a broad interest in derivatives of fostriecin. [1,4]
References:
[1] Zhang X, Wu J, Fang L, Willis WD. The effects of protein phosphatase inhibitors on the duration of central sensitization of rat dorsal horn neurons following injection of capsaicin. Mol Pain. 2006 Jul; 2(23). DOI:10.1186/1744-8069-2-23.
[2] Scheithauer W, Von Hoff DD, Clark G, Shillis JL, Elslager EF. In vitro activity of the novel antitumor antibiotic fostriecin (CI-920) in a human tumor cloning assay. Eur J Cancer Res & Clin Oncol. 1986 Aug; 22(8): 921-6.
[3] Gedik CM, Collins AR. Comparison of effects of fostriecin, novobiocin, and camptothecin, inhibitors of DNA topoisomerases, on DNA replication and repair in human cells. Nnucleic Acids Res. 1990 Aug; 18(4):1008-13.
[4] Le LH, Erlichman C, Pillon L, Thiessen JJ, Day A, Wainman N, Eisenhauer EA, Moore MJ. Phase I and pharmacokinetic study of fostriecin given as an intravenous bolus daily for five consecutive days.
Invest New Drugs. 2004 Apr; 22(2):159-67.
| Physical Appearance | Colourless lyophilised solid |
| Storage | Desiccate at -20°C |
| M.Wt | 452.37 |
| Cas No. | 87860-39-7 |
| Formula | C19H26O9PNa |
| Solubility | Soluble in H2O |
| Chemical Name | sodium;[(3R,4R,6R,7E,9E,11E)-3,6,13-trihydroxy-3-methyl-1-[(2R)-6-oxo-2,3-dihydropyran-2-yl]trideca-1,7,9,11-tetraen-4-yl] hydrogen phosphate |
| SDF | Download SDF |
| Canonical SMILES | C[C@]([C@@H](C[C@H](/C=C/C=C/C=C/CO)O)OP([O-])(O)=O)(C=C[C@@H](CC=C1)OC1=O)O.[Na+] |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment[1]: | |
|
Cell lines |
HeLa cells |
|
Preparation method |
The solubility of this compound in sterile water is 100 mM. General tips for obtaining a higher concentration: Please warm the tube at 37 ℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reacting condition |
30 min, 0.22 mM |
|
Applications |
Fostriecin is an inhibitor of topoisomerase II. It blocks an early step in the reaction and does not accumulate broken DNA intermediates. Fostriecin causes a strong but delayed inhibition of DNA synthesis in human Hela cells. |
| Animal experiment [2]: | |
|
Animal models |
B6D2F1 mice with subcutaneous Colon 38 tumours. |
|
Dosage form |
Intraperitoneal injection, 65 mg/kg |
|
Application |
When administered as a single dose, fostriecin caused extensive necrosis of tumours after 24 h and induced significant delays in the growth of advanced subcutaneous tumours by at least 10 days. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Gedik C M, Collins A R. Comparison of effects of fostriecin, novobiocin, and camptothecin, inhibitors of DNA topoisomerases, on DNA replication and repair in human cells[J]. Nucleic acids research, 1990, 18(4): 1007-1013. [2]. Baguley B C, Calveley S B, Crowe K K, et al. Comparison of the effects of flavone acetic acid, fostriecin, homoharringtonine and tumour necrosis factor α on colon 38 tumours in mice[J]. European Journal of Cancer and Clinical Oncology, 1989, 25(2): 263-269. | |
Quality Control & MSDS
- View current batch:
Chemical structure