WH-4-023
WH-4-023 is a potent and selective Lck and Src inhibitor with IC50 values of 2 and 6 nM, respectively [1]. It is also an inhibitor of IC50 values of 10, 22 and 60 nM for SIK 1, 2 and 3, respectively [2].
Lck and Src are cytoplasmic tyrosine kinases of the Src family expressed in T cells and NK cells [1]. SIKs restrict the formation of regulatory macrophages and that their inhibition greatly stimulates the production of IL-10 and other anti-inflammatory molecules [2].
WH-4-023 inhibited a number of protein tyrosine kinases that possess a Thr residue at the gatekeeper site, such as FGF and Ephrin receptors, Src family members (Src, Lck, and Yes) and BTK. WH-4-023 also inhibited the SIKs and did not inhibit any other member of the AMPK-related kinase subfamily, which all possess a large hydrophobic residue (Met or Leu) at the gatekeeper site. WH-4-023 inhibited SIK 1, 2 and 3 with IC50 values of 10, 22 and 60 nM, respectively. Also, WH-4-023 increased LPS-stimulated IL-10 production and greatly suppressed proinflammatory cytokine secretion [2].
References:
[1]. Martin MW, Newcomb J, Nunes JJ, et al. Novel 2-aminopyrimidine carbamates as potent and orally active inhibitors of Lck: synthesis, SAR, and in vivo antiinflammatory activity. J Med Chem, 2006, 49(16): 4981-4991.
[2]. Clark K, MacKenzie KF, Petkevicius K, et al. Phosphorylation of CRTC3 by the salt-inducible kinases controls the interconversion of classically activated and regulatory macrophages. Proc Natl Acad Sci U S A, 2012, 109(42): 16986-16991.
| Physical Appearance | A solid |
| Storage | Desiccate at -20°C |
| M.Wt | 568.67 |
| Cas No. | 837422-57-8 |
| Formula | C32H36N6O4 |
| Solubility | ≥56.9 mg/mL in DMSO; insoluble in H2O; ≥2.42 mg/mL in EtOH with gentle warming and ultrasonic |
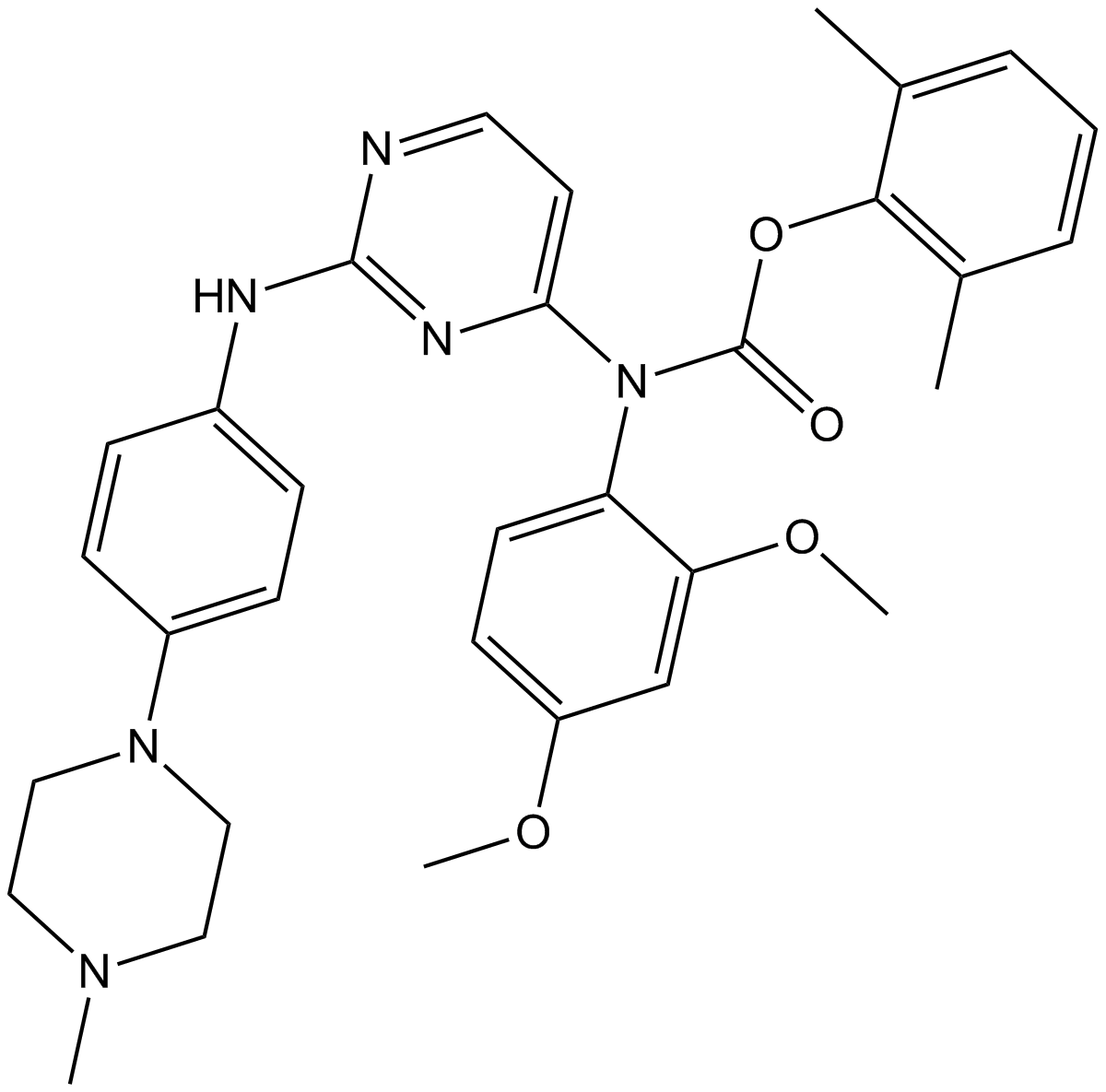
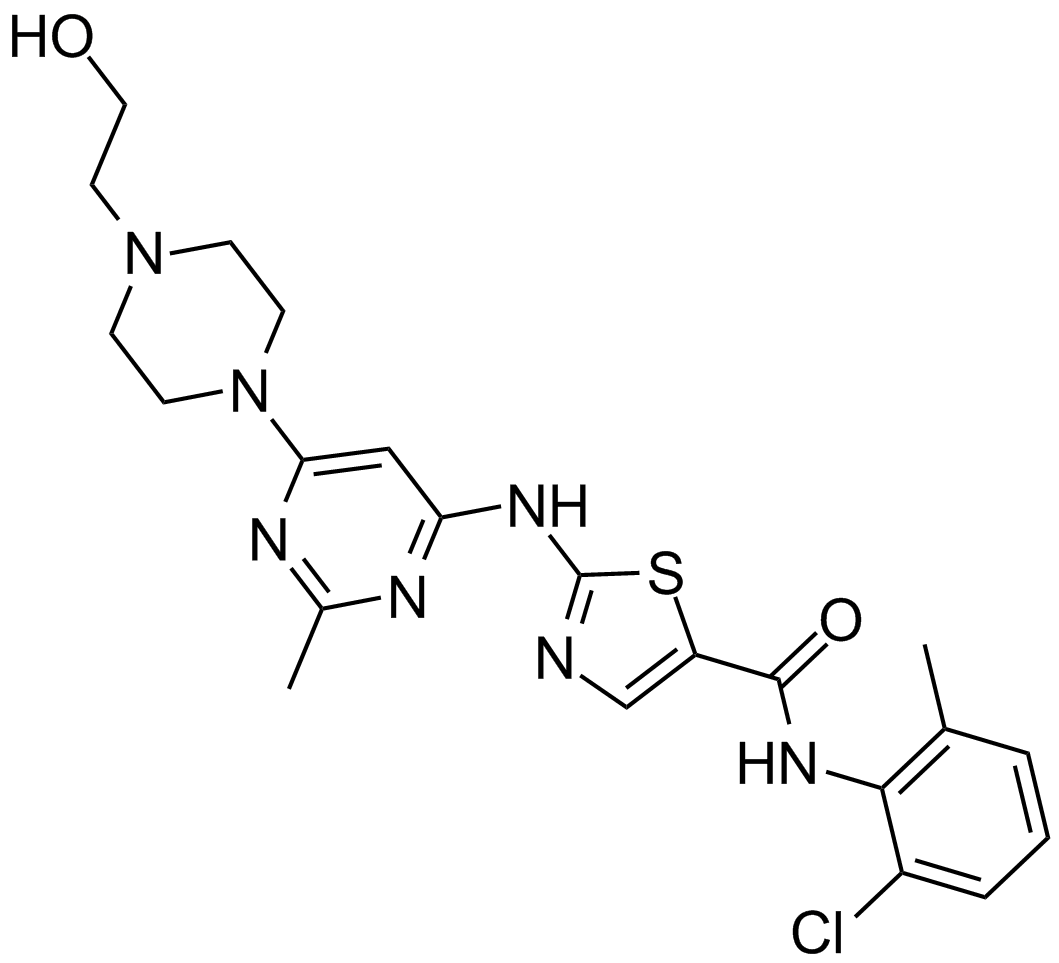
| Chemical Name | 2,6-dimethylphenyl (2,4-dimethoxyphenyl)(2-((4-(4-methylpiperazin-1-yl)phenyl)amino)pyrimidin-4-yl)carbamate |
| Canonical SMILES | CN1CCN(C2=CC=C(NC3=NC(N(C4=CC=C(OC)C=C4OC)C(OC5=C(C)C=CC=C5C)=O)=CC=N3)C=C2)CC1 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Kinase experiment[1]: | |
|
Lck kinase assays |
To determine the inhibitory activity of WH-4-023 against Lck, kinase assays were conducted. The Lck HTRF kinase assay involved ATP-dependent phosphorylation of a biotinylated substrate peptide of gastrin with or without WH-4-023. The final concentration of ATP, gastrin and Lck were 0.5 μM, 1.2 μM and 250 pM, respectively. Buffer were made up as follows: 50 mM HEPES pH=7.5, 2 mM DTT, 5 mM MnCl2, 20 mM MgCl2, 50 mM NaCl, 0.05% BSA. The reaction was quenched and ended by adding 160 μL detection reagent. Detection reagent was prepared as follows: 50 mM Tris, pH=7.5, 3 mM EDTA, 100 mM NaCl, 0.05% BSA, 0.1% Tween-20. Before reading, streptavidin allophycocyanin and anti-phosphotyrosine Ab were added at a final concentration of 0.4 μg/mL and 0.025 nM, respectively. Assay plate was read with excitation at 320 nm and emission at 615 and 655 nm. |
| Cell experiment[2]: | |
|
Cell lines |
Naive human pluripotent cells |
|
Preparation method |
The solubility of this compound in DMSO is > 10 mM. General tips for obtaining a higher concentration: Please warm the tube at 37 ℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reacting condition |
1 μM |
|
Applications |
WH-4-023 is a potent and selective Src and Lck inhibitor with IC50 values of 6 and 2 nM, respectively[1].Naive human pluripotent cells were maintained in media containing WH-4-023. WH-4-023 improves the fraction of viable GFP+ cells. Withdrawal of the WH-4-023 results in a change in morphology and slight decrease of OCT4-DPE-GFP reporter activity[2]. |
|
References: [1]. Martin M W, Newcomb J, Nunes J J, et al. Novel 2-aminopyrimidine carbamates as potent and orally active inhibitors of Lck: synthesis, SAR, and in vivo antiinflammatory activity[J]. Journal of medicinal chemistry, 2006, 49(16): 4981. [2]. Theunissen T W, Powell B E, Wang H, et al. Systematic Identification of Defined Conditions for Induction and Maintenance of Naive Human Pluripotency[J]. Cell stem cell, 2014, 15(4): 471. | |
Quality Control & MSDS
- View current batch:
Chemical structure