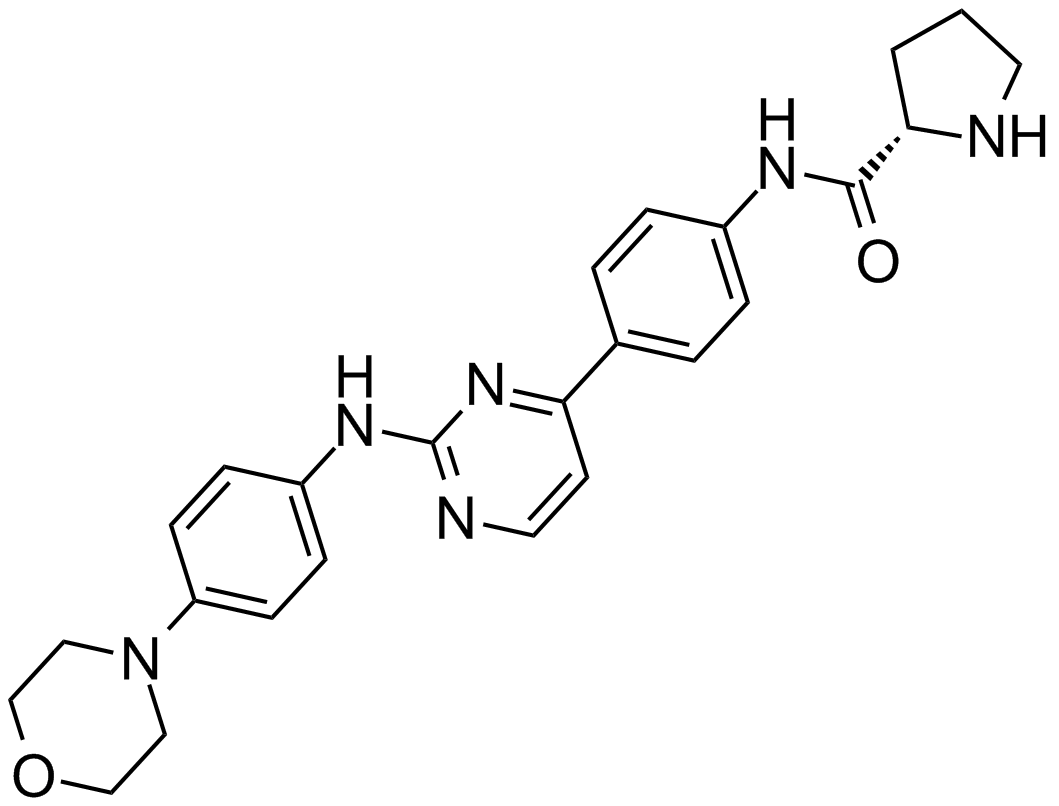
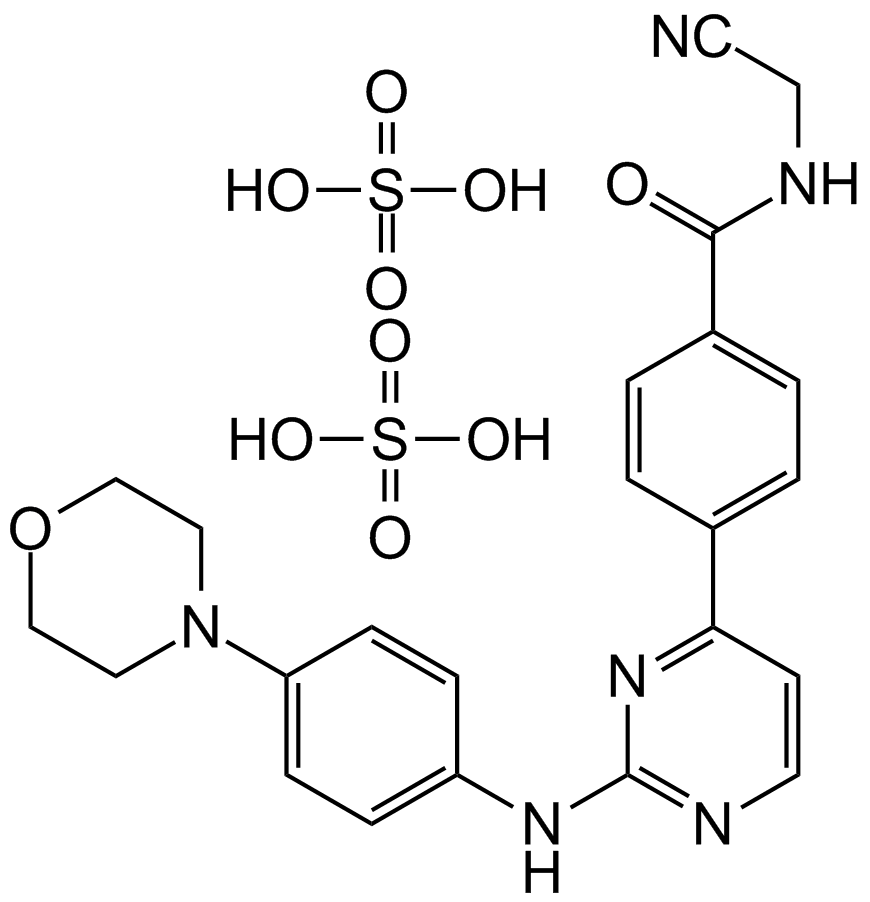
XL019
XL019 is a potent and selective inhibitor of JAK2 with IC50 value of 2.2 nM.
JAK kinases were first identified in 1989 and characterized by the presence of two tyrosine kinase domains. There are four members in the JAK family, which are JAK1, JAK2, JAK3 and TYK2. JAK2 played an important role in the pathways that control erythroid, myeloid and megakaryocytic development. JAK2 mediates signaling through erythropoietin receptor, thrombopoietin receptor and cytokine receptor that harbor the common β-chain (e.g. IL3 receptor) and IFN-γ receptors [38]. JAK2 inhibitors are a novel class of agents with promising results for treating patients with polycythemia vera (PV), essential thrombocythemia (ET) and myelofibrosis (MF) [1]. XL019 was selected as a clinical candidate and advanced into human clinical trials where it was evaluated in patients with PMF, post-PV, or post-ET MF [2].
XL019 shows good biochemical and cellular potency against JAK2 with good selectivity, against a panel of over 100 serine/threonine and tyrosine kinases, including other members of the JAK family [2]. Analogue XL019 was evaluated against a select panel of 118 kinases. Targets for which XL019 exhibited IC50 <1000 nM are displayed. XL019 is a highly selective JAK2 inhibitor displaying >50-fold selectivity against all kinases tested including JAK1 and TYK2. XL019 was a desirable CYP, hERG (16 μM), and P-glycoprotein inhibition (>20 μM).
XL019 had a superior pharmacodynamic profile and thus was evaluated in an efficacy experiment measuring growth inhibition of HEL.92.1.7 xenograft tumors in mice. Derivative XL019 demonstrated 60% and 70% inhibition when dosed orally at 200 mg/kg and 300 mg/kg respectively twice a day for 14 days. Thirty patients received XL019 for a median of 91 days. All of them have discontinued XL019 therapy. Toxicity: N/A Clinical trial: A Safety Study of XL-019 in Adults With Myelofibrosis [2].
References:
[1]. Fabio P.S. Santos, Srdan Verstovsek. JAK2 inhibitors: What's the true therapeutic potential?. Blood Reviews, 2011, 25: 53-63.
[2]. Srdan Verstovsek, Constantine S. Tam, Martha Wadleigh, et al. Phase I evaluation of XL019, an oral, potent, and selective JAK2 inhibitor. Leukemia Research, 2014, 38: 316-322.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 444.53 |
| Cas No. | 945755-56-6 |
| Formula | C25H28N6O2 |
| Synonyms | XL-019;XL 019 |
| Solubility | insoluble in EtOH; insoluble in H2O; ≥11.125 mg/mL in DMSO |
| Chemical Name | (2S)-N-[4-[2-(4-morpholin-4-ylanilino)pyrimidin-4-yl]phenyl]pyrrolidine-2-carboxamide |
| SDF | Download SDF |
| Canonical SMILES | C1CC(NC1)C(=O)NC2=CC=C(C=C2)C3=NC(=NC=C3)NC4=CC=C(C=C4)N5CCOCC5 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1]: | |
|
Cell lines |
HEL92.1.7 and KG-1 tumor cell lines, primary human erythroid cells |
|
Preparation method |
The solubility of this compound in DMSO is > 11.1 mg/mL. General tips for obtaining a higher concentration: Please warm the tube at 37 ℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reacting condition |
24h |
|
Applications |
XL019 downregulated STAT signaling in cell lines expressing both wild type and activated forms of JAK2. IC50s for inhibition of STAT5 phosphorylation by XL019 ranged from 623 nM (HEL92.1.7) to 3398 nM (KG-1) in tumor cell lines. XL019 showed increased potency in primary human erythroid cells, where the IC50 for inhibition of EPO-stimulated phospho-STAT5 was 64 nM. XL019 inhibited proliferation in cell lines harboring activated or overexpressed JAK2, including certain lines derived from patients with Hodgkin’s Lymphoma (L-1236, 928 nM IC50), AML (MV4-11, 992 nM IC50), essential thrombocythemia (SET-2, 386 nM IC50), and erythroleukemia (HEL92.1.7, 6777 nM IC50). |
| Animal experiment [1,2]: | |
|
Animal models |
Mice bearing HEL92.1.7, CFPAC-1 and DU 145 xenograft tumors |
|
Dosage form |
Oral administration, 30, 100, and 300 mg/kg. |
|
Application |
Oral administration of XL019 (30, 100, and 300 mg/kg) significantly inhibited downstream markers pSTAT1 and pSTAT3 with an ED50 of 42 mg/kg (pSTAT1) and 210 mg/kg (pSTAT3). XL019 demonstrated 60% and 70% inhibition when dosed orally at 200 mg/kg and 300 mg/kg respectively twice a day for 14 days. XL019 showed a superior pharmacodynamic profile and exhibited good oral absorption, and modest clearance and halflife across species. XL019 inhibited HEL.92.1.7 xenograft tumors growth in mice. XL019 showed potent effect on JAK-STAT signaling in HEL92.1.7, CFPAC-1 and DU 145 xenograft tumors. Twice daily dosing (bid) led to substantial tumor growth inhibition in the DU145 and HEL models (maximum tumor growth inhibition of 86% and 60%, respectively), accompanied by increases in tumor cell apoptosis (4 – 4.4 fold increase) and decreases in tumor microvasculature (44% reduction in DU 145 xenograft tumors). |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Verstovsek S, Pardanani A D, Shah N P, et al. A Phase I Study of XL019, a Selective JAK2 Inhibitor, in Patients with Primary Myelofibrosis and Post-Polycythemia Vera/Essential Thrombocythemia Myelofibrosis[J]. 2007. [2]. Forsyth T, Kearney P C, Kim B G, et al. SAR and in vivo evaluation of 4-aryl-2-aminoalkylpyrimidines as potent and selective Janus kinase 2 (JAK2) inhibitors[J]. Bioorganic & medicinal chemistry letters, 2012, 22(24): 7653-7658. |
|
| Description | XL019 is a potent and selective inhibitor of JAK2 with IC50 value of 2.2 nM. | |||||
| Targets | JAK2 | |||||
| IC50 | 2.2 nM | |||||
Quality Control & MSDS
- View current batch:
Chemical structure