Valinomycin
LD50 = 2.5 mg/kg for mouse and 5 mg/kg for rabbit [1]
Valinomycin is a dodecadepsipeptide antibiotic, which is obtained from several Streptomyces strains. Valinomycin functions as a potassium-specific transporter and promotes the movement of K+ through lipid membranes. Excessive K+ efflux is an ionic mechanism underlying apoptosis.
In vitro: Valinomycin caused substantial CHO cells death within 12 h of treatment. Several apoptotic events were identified in valinomycin-treated CHO cells, including caspase-3 activation, phosphatidylserine (PS) membrane translocation, and mitochondrial membrane depolarization during the first few hours of treatment. K+ efflux was reduced by elevating extracellular K+ concentrations [2].
In vivo: Valinomycin is irritant in the case of eye and skin contact. Inhalation of valinomycin can cause breathing disturbances and even loss of conscious. Lethal doses (LD50) for mouse and rabbit is 2.5 mg/kg and 5 mg/kg respectively. Valinomycin also shows to provoke lots of chronic effects, including damage of the central and peripheral nervous system, eyes, lens and cornea [1].
Clinical trial: Because valinomycin’s toxicity to eukaryotic cells, it can not be used in human therapy [1].
References:
[1] Kroteń MA, Bartoszewicz M, Swiecicka I. Cereulide and valinomycin, two important natural dodecadepsipeptides with ionophoretic activities. Pol J Microbiol. 2010;59(1):3-10.
[2] Abdalah R1, Wei L, Francis K, Yu SP. Valinomycin-induced apoptosis in Chinese hamster ovary cells. Neurosci Lett. 2006 Sep 11;405(1-2):68-73. Epub 2006 Jul 20.
| Physical Appearance | A crystalline solid |
| Storage | Desiccate at -20°C |
| M.Wt | 1111.32 |
| Cas No. | 2001-95-8 |
| Formula | C54H90N6O18 |
| Solubility | ≥51.1 mg/mL in DMSO; ≥51.6 mg/mL in EtOH; insoluble in H2O |
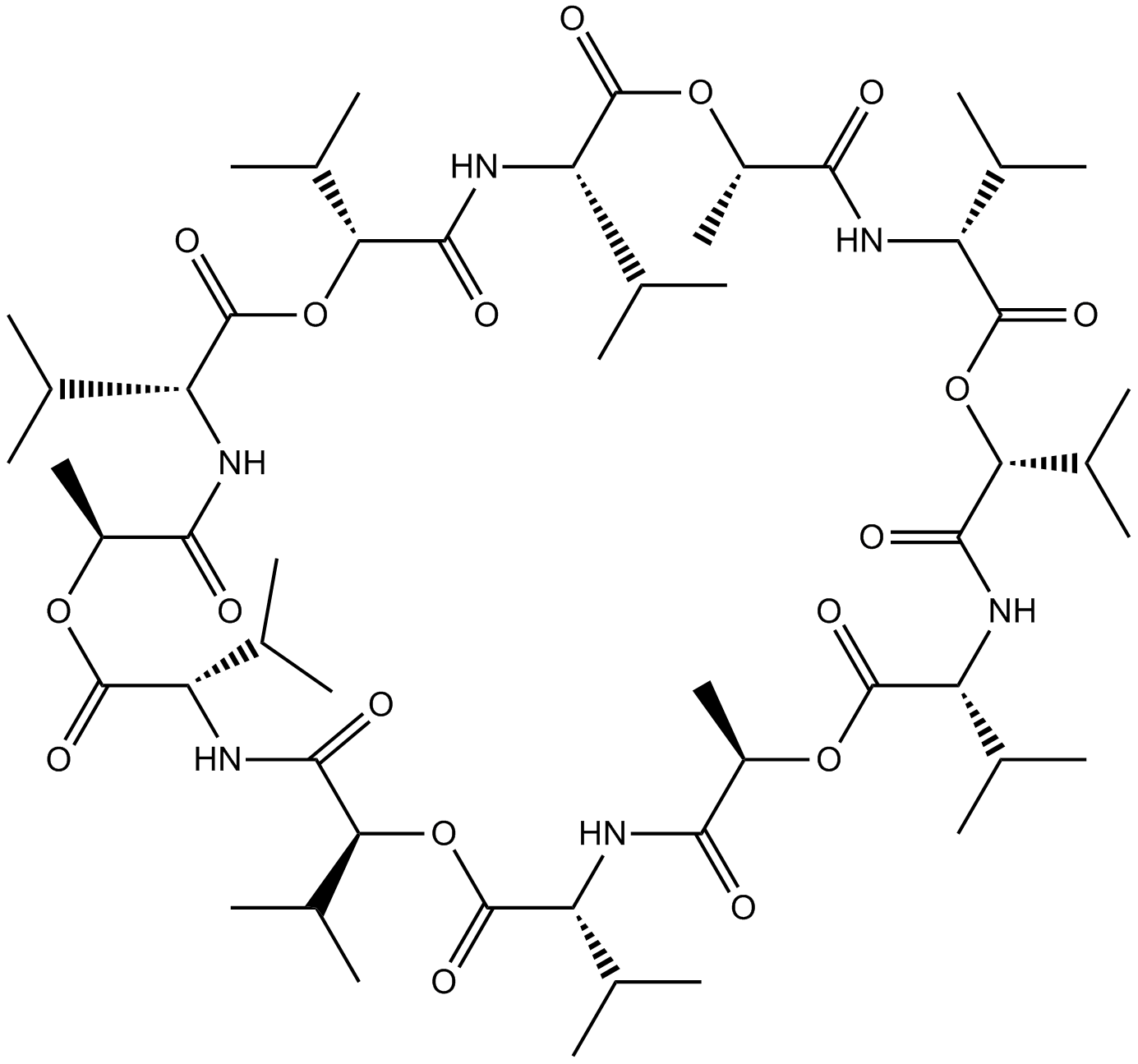
| Chemical Name | (3S,6R,9R,12S,15S,18S,21R,24R,27R,30R,33R,36S)-3,6,9,15,18,21,27,30,33-nonaisopropyl-12,24,36-trimethyl-1,7,13,19,25,31-hexaoxa-4,10,16,22,28,34-hexaazacyclohexatriacontane-2,5,8,11,14,17,20,23,26,29,32,35-dodecaone |
| Canonical SMILES | O=C([C@@H](C(C)C)OC([C@@H](C(C)C)NC1=O)=O)N[C@@H](C(O[C@H](C)C(N[C@@H](C(O[C@H](C(N[C@H](C(O[C@@H](C)C(N[C@@H](C(O[C@@H](C(N[C@H](C(O[C@H]1C)=O)C(C)C)=O)C(C)C)=O)C(C)C)=O)=O)C(C)C)=O)C(C)C)=O)C(C)C)=O)=O)C(C)C |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure