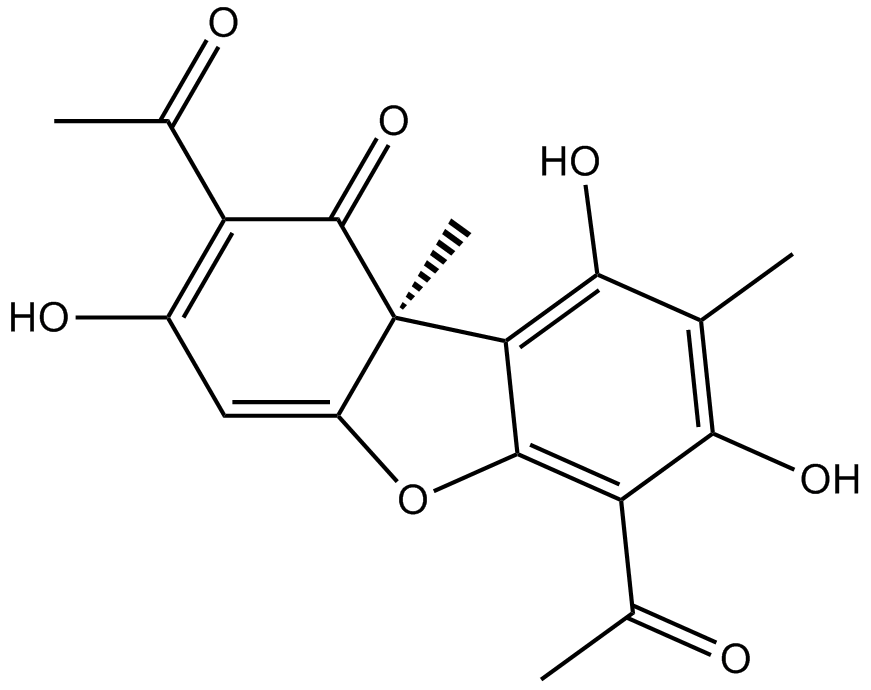
(+)-Usniacin
MIC: 0.05 μg/62.5 μl to 3.1 μg/62.5 μl
Microorganisms can colonize a wide variety of medical devices, putting patients at risk for systemic and local infectious complications, including local-site infections, endocarditis, and catheter-related bloodstream infections. (+)-Usniacin is a secondary lichen metabolite that possesses antimicrobial activity against various planktonic gram-positive bacteria.
In vitro: (+)-Usniacin showed antimicrobial activity against the same microorganisms as that of acetone extract. Among the three analogues it was the most active one having quite low MIC values. Furthermore, (+)-Usniacin did not show any activity against A. hydrophila and B. cereus whereas (D)-usnic acid did. On the other hand, (+)-Usniacin was active against Y. enterocolitica whereas (D)-usnic acid was not active [1].
In vivo: No animal in-vivo study has been reproted so far.
Clinical trials: Trials carried out in volunteers showed that mouth-rinse with (+)-Usniacin preparations exerted a selective and long lasting action against S. mutans. The adherence of S. mutans to smooth surfaces was not increased by the presence of subinhibiting concentrations of (+)-Usniacin. These characteristics make (+)-Usniacin a suitable candidate for topical use in oral medicine [2].
References:
[1] Tay T, Türk AO, Yilmaz M, Türk H, Kivanç M. Evaluation of the antimicrobial activity of the acetone extract of the lichen Ramalina farinacea and its (+)-usnic acid, norstictic acid, and protocetraric acid constituents. Z Naturforsch C. 2004 May-Jun;59(5-6):384-8.
[2] Ghione M, Parrello D, Grasso L. Usnic acid revisited, its activity on oral flora. Chemioterapia. 1988 Oct;7(5):302-5.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 344.32 |
| Cas No. | 7562-61-0 |
| Formula | C18H16O7 |
| Solubility | insoluble in H2O; insoluble in EtOH; ≥8.03 mg/mL in DMSO with gentle warming |
| Canonical SMILES | C[C@@](C(Oc1c(C(C)=O)c(O)c2C)=CC(O)=C3C(C)=O)(c1c2O)C3=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Description | (+)-Usniacin is a naturally occurring dibenzofuran derivative | |||||
| Targets | ||||||
| IC50 | ||||||
Quality Control & MSDS
- View current batch:
Chemical structure