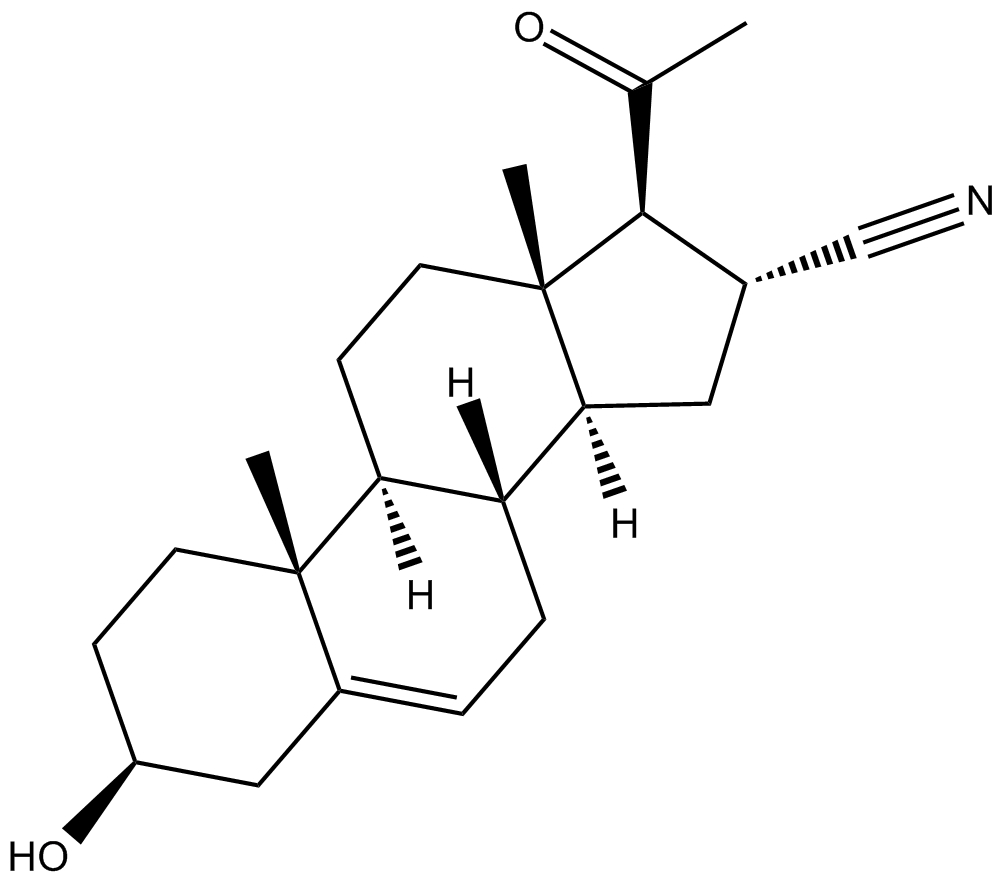
Pregnenolone Carbonitrile
Pregnenolone Carbonitrile (CAS 1434-54-4), known as Pregnenolone-16α-carbonitrile (PCN), is a pregnane X receptor (PXR) agonist primarily utilized in biomedical research investigating xenobiotic metabolism pathways. By activating rodent nuclear PXR, PCN stimulates expression of cytochrome P450 enzymes, notably of the CYP3A subfamily, enhancing hepatic detoxification and clearance of diverse foreign substances. Independently of PXR, PCN exhibits antifibrotic activity in liver fibrosis models, shown by inhibition of hepatic stellate cell trans-differentiation in vitro and decreased hepatic fibrosis in vivo. PCN thus serves as a useful tool for studying both PXR-dependent gene regulatory mechanisms and PXR-independent anti-fibrogenic effects.
- 1. Peipei Hao, Ruifeng Ding, et al. "Sea buckthorn regulates PXR/CAR/NF - κB signaling and restores CYP2C metabolic function in BCG - induced hepatitis." J Ethnopharmacol. 2025 Jul 24:351:120142 PMID: 40513919
- 2. Cheng-Hua Wu, Shuang Hu, et al. "Pregnane X receptor alleviates sepsis-induced liver injury through activation of yes-associated protein in mice." Acta Pharmacol Sin. 2025 Apr 15 PMID: 40234620
- 3. Yifei Zhang, Jie Yang, et al. "The reversal of PXR or PPARα activation-induced hepatomegaly." Toxicol Lett. 2024 Jun:397:79-88. PMID: 38734220
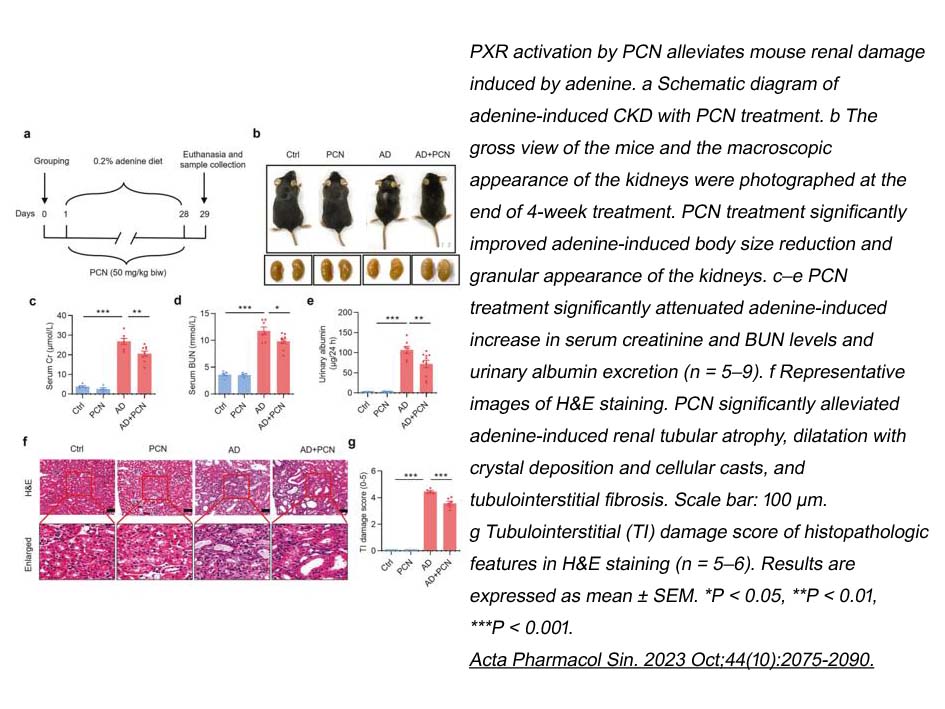
- 4. Wen-Hua Ming, Zhi-Lin Luan, et al. "Pregnane X receptor activation alleviates renal fibrosis in mice via interacting with p53 and inhibiting the Wnt7a/β-catenin signaling." Acta Pharmacol Sin. 2023 Oct;44(10):2075-2090. PMID: 37344564
- 5. Yingjian Chen, Tianqi Cui, et al. "Hepatic ZBTB22-mediated detoxification ameliorates acetaminophen-induced liver injury by inhibiting pregnane X receptor signaling." iScience. 2023 Mar 2;26(4):106318. PMID: 36950116
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 341.5 |
| Cas No. | 1434-54-4 |
| Formula | C22H31NO2 |
| Synonyms | PCN,Pregnenolone 16α-carbonitrile,SC-4674 |
| Solubility | insoluble in H2O; insoluble in EtOH; ≥14.17 mg/mL in DMSO |
| Chemical Name | 3β-hydroxy-20-oxo-pregn-5-ene-16α-carbonitrile |
| SDF | Download SDF |
| Canonical SMILES | C[C@](CC1)([C@@H](C[C@H]2C#N)[C@H]3[C@H]1[C@@](C)(CC[C@@H](C1)O)C1=CC3)[C@H]2C(C)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data