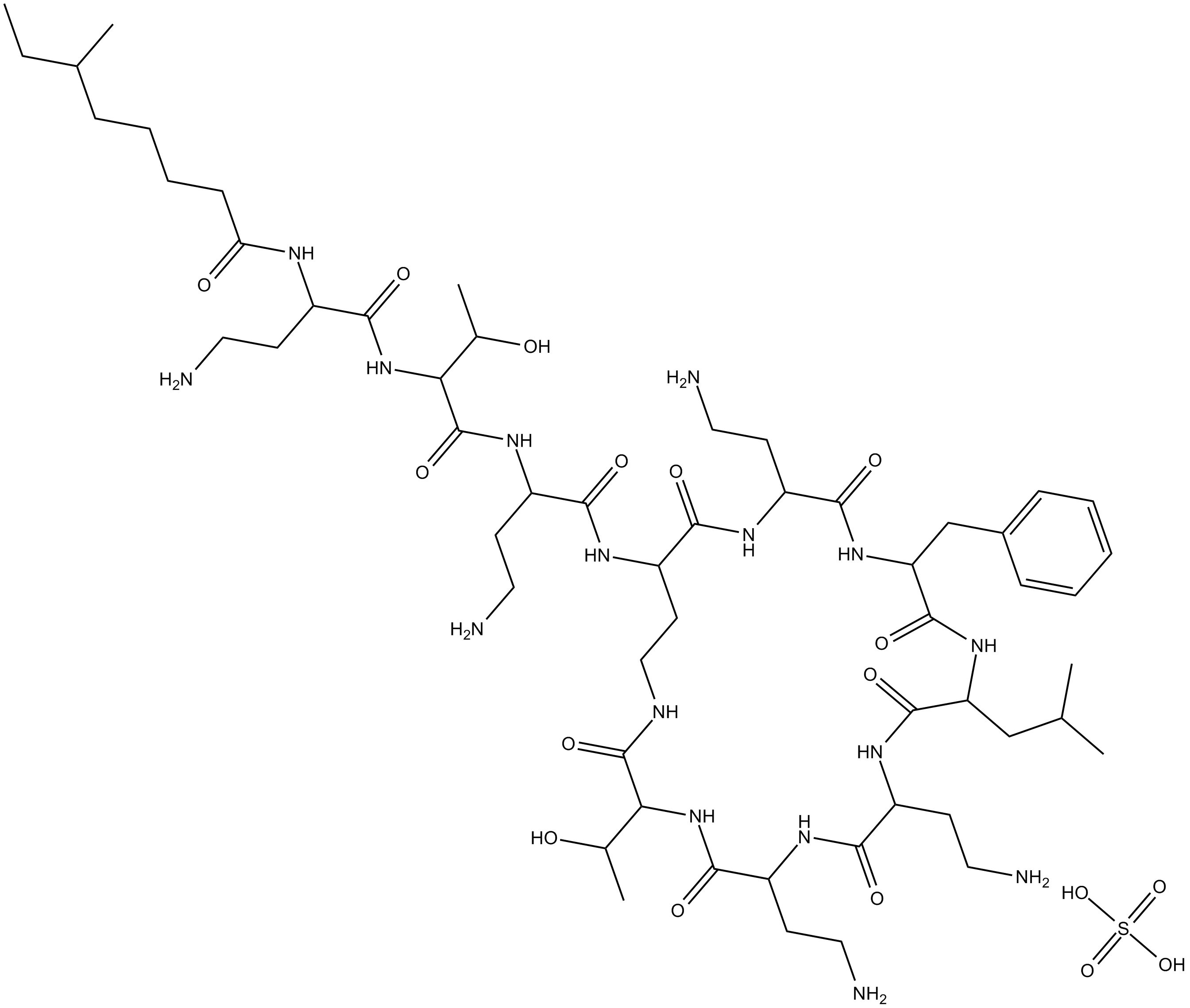
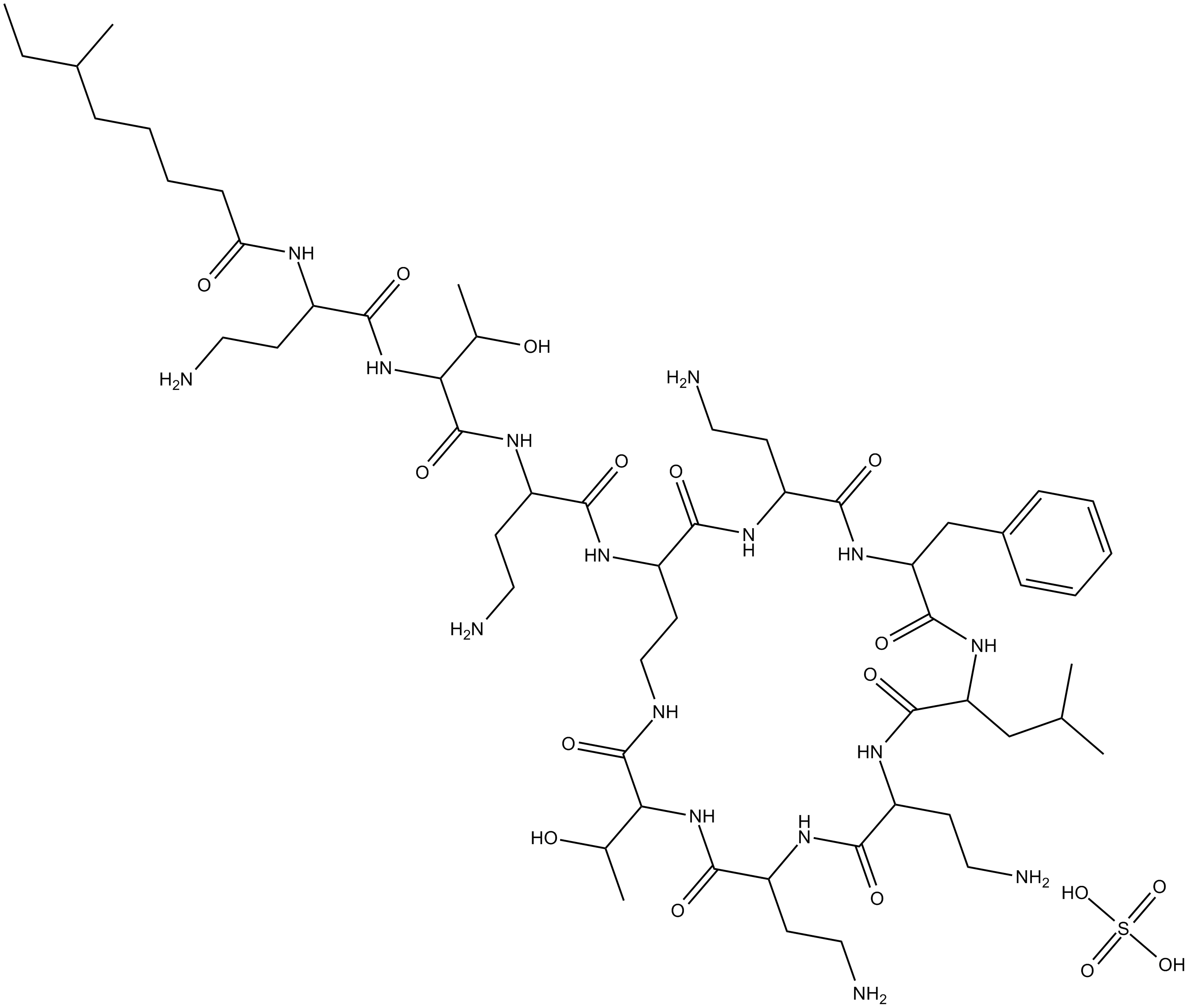
Polymyxin B (sulfate)
Polymyxin B, a mixture of polymyxins B1 and B2, is obtained from Bacillus polymyxa strains. Polymyxin B exhibits bactericidal activity against major multidrug-resistant gram-negative bacteria, most fungi and gram-positive bacteria. As basic polypeptides of about eight amino acids, polymyxins B1 and B2 have cationic detergent action on cell membranes. Moreover, polymyxin B is used for infections with gram-negative organisms, which may be nephrotoxic and neurotoxic. Polymyxin B is appropriate to treat the infections of the meninges, urinary tract, and blood stream, triggered by susceptible Pseudomonas aeruginosa strains.
In vitro: Polymyxin B elicited up-regulation of dendritic cells (DCs) maturation markers, including the increase in the in the expression of co-stimulatory molecule CD86 and HLA-class I and II molecules. Polymyxin B induced a progressive increase in the adhesion property of human DCs. In addition, polymyxin B triggered the activation of the ERK1/2 pathway and IκB-α/NF-κB pathways [1].
In vivo: Male bacteraemia ddY mice were subcutaneously treated with polymyxin B at a dose of 5, 10, 15 or 20 mg/kg for 7 days. Polymyxin B, in a dose-dependent fashion, improved the survival both of OU-01062- and OU-98039-infected mice. In polymyxin B-treated mice, except for 5 mg/kg polymyxin B, the viable cell counts had a tendency to reduce steadily in each concentration group. It was showed a rapid and marked decline of bacterial cell count between 3 to 6 h after infection [2].
References:
[1]. Valentinis, B., Bianchi, A., Zhou, D., Cipponi, A., Catalanotti, F., Russo, V., & Traversari, C. Direct Effects of Polymyxin B on Human Dendritic Cells Maturation: THE ROLE OF I B- /NF- B AND ERK1/2 PATHWAYS AND ADHESION. Journal of Biological Chemistry. 2005; 280(14): 14264-14271.
[2]. Miyajima, Y., Hiramatsu, K., Mizukami, E., Morinaga, R., Ishii, H., & Shirai, R. et al. In vitro and in vivo potency of polymyxin B against IMP-type metallo-β-lactamase-producing Pseudomonas aeruginosa. International Journal of Antimicrobial Agents. 2008; 32(5): 437-440.
- 1. Guanqing Chen, Xinliang Yan, et al. "Characterization and transmission dynamics of carbapenemase-encoding genes in carbapenem-resistant Enterobacter cloacae isolated from eight teaching hospitals in Guangdong province, China (2022–2024)." BMC Microbiol. 2025 Oct 17;25(1):667. PMID: 41107722
- 2. Puspendu Sardar, Benjamin S Beresford-Jones, et al. "Gut microbiota-derived hexa-acylated lipopolysaccharides enhance cancer immunotherapy responses." Nat Microbiol.2025 Mar;10(3):795-807. PMID: 39929976
- 3. Shuiping Yan, Jian Zheng, et al. "Effect of Shufeng Xingbi Therapy on Th1/Th2 immune balance and intestinal flora in rats with allergic rhinitis." bioRxiv. March 29, 2025
- 4. Benjamin S. Beresford-Jones, Puspendu Sardar, et al. "Hexa-acylated lipopolysaccharides from the gut microbiota enhance cancer immunotherapy responses." bioRxiv, August 04, 2024
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 1301.6 |
| Cas No. | 1405-20-5 |
| Formula | C56H98N16O13·H2SO4 |
| Synonyms | Aerosporin,Mastimyxin |
| Solubility | ≤2mg/ml in PBS(pH7.2) |
| Chemical Name | N-(4-amino-1-((1-((4-amino-1-oxo-1-((6,9,18-tris(2-aminoethyl)-15-benzyl-3-(1-hydroxyethyl)-12-isobutyl-2,5,8,11,14,17,20-heptaoxo-1,4,7,10,13,16,19-heptaazacyclotricosan-21-yl)amino)butan-2-yl)amino)-3-hydroxy-1-oxobutan-2-yl)amino)-1-oxobutan-2 |
| SDF | Download SDF |
| Canonical SMILES | O=C(C(NC(C(CCN)NC(CCCCC(C)CC)=O)=O)C(C)O)NC(CCN)C(NC(CCNC(C(NC(C(NC(C(CCN)N1)=O)CCN)=O)C(O)C)=O)C(NC(CCN)C(NC(CC2=CC=CC=C2)C(NC(CC(C)C)C1=O)=O)=O)=O)=O.O=S(O)(O)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure