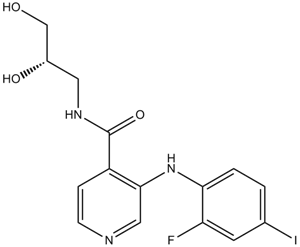
Pimasertib (AS-703026)
IC50: ranging from 0.005 to 2 μM for the growth of MM cell lines
The MEK/ERK pathway transmits mitogenic signals to the nucleus and thus regulates the expression of genes that are critical for survival, proliferation, and differentiation. This pathway is often upregulated in cancer as result of oncogenic mutations. MEK is a crucial kinase in this pathway and has been targeted for the therapeutic agents development that block the MEK/ERK signaling cascade. AS 703026 is a novel small molecule MEK inhibitor.
In vitro: AS 703026 binds to the distinctive MEK allosteric site and therefore exhibits exquisite kinase selectivity. The AS 703026 binding to MEK is independent of ATP, and results in enzyme inhibition by prevention of activation. This inhibitor shows strong antiproliferative effect on tumor cell lines bearing oncogenic mutations or amplifications along the MEK/ERK pathway [1].
In vivo: In relevant mouse xenograft models, AS 703026 inhibits tumor growth after oral administration. The effect on tumor growth is dose-dependant and correlates with target modulation in both tumors and blood [1].
Clinical trial: 16 patients were enrolled in the trial. 10 and 6 patients were treated daily with 45 and 60 mg of AS 703026 plus FOLFIRI, respectively. The MTD was considered to be 45 mg/day. Of the 15 patients in the efficacy analysis group, 2 patients had partial response, 9 patients had stable disease, 3 patients had progressive disease as their best overall response and 1 patient could not be evaluated.
Reference:
[1] Goutopoulos A, Askew BC, Bankston D, Clark A, Dhanabal M, Dong R, Fischer D, Healey B, Jiang X, Josephson K, Lin J, Ma J, Noonan T, Qiu D, Rocha C, Romanelli A, Shutes A, Spooner E, Tian H, H Y. AS703026: a novel allosteric MEK inhibitor. AACR Annual meeting. 2009 Abstract#4776.
[2] Macarulla T, Cervantes A, Tabernero J et al. Phase I study of FOLFIRI plus pimasertib as second-line treatment for KRAS-mutated metastatic colorectal cancer. Br J Cancer. 2015 Jun 9;112(12):1874-81.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 431.2 |
| Cas No. | 1236699-92-5 |
| Formula | C15H15FIN3O3 |
| Solubility | ≥21.55 mg/mL in DMSO; insoluble in H2O; ≥10.36 mg/mL in EtOH with ultrasonic |
| Chemical Name | N-[(2S)-2,3-dihydroxypropyl]-3-(2-fluoro-4-iodoanilino)pyridine-4-carboxamide |
| SDF | Download SDF |
| Canonical SMILES | C1=CC(=C(C=C1I)F)NC2=C(C=CN=C2)C(=O)NCC(CO)O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1, 2]: | |
|
Cell lines |
human multiple myeloma (MM) |
|
Preparation method |
The solubility of this compound in DMSO is >21.6 mg/mL. General tips for obtaining a higher concentration: Please warm the tube at 37 ℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reacting condition |
0.1-10 μM |
|
Applications |
In human multiple myeloma (MM), AS703026 (5, 0.5, and 0.1 μM) inhibited growth and survival of MM cells and cytokine-induced osteoclast differentiation. In MM cells, AS703026 inhibited the growth of MM cell lines in a dose-dependent manner, with IC50s ranging from 0.005 to 2 μM. The IC50s of AS703026 against INA-6, U266, H929 cells are 10 nM, 5 nM, and 200 nM, respectively. AS703026 induced apoptosis and modulated the cell cycle profile. AS703026 (10 μmol/L) inhibited ERK pathway, proliferation, and transformation in cetuximab-resistant D-MUT cells. |
| Animal experiment [1, 2]: | |
|
Animal models |
CB17 SCID mice bearing human H929 MM xenografts |
|
Dosage form |
Oral administration, 15, 30 mg/kg, twice daily |
|
Application |
In the human H929 MM xenograft model in CB17 SCID mice, AS703026 (15, 30 mg/kg, twice daily, p.o.) significantly inhibited tumor growth in a time-dependent manner, with no significant body weight loss. AS703026-treated H929 tumors from mice exhibited increased cleaved caspase 3 and TUNEL. AS703026 treatment significantly reduced the percentage of CD34+ cells and microvascular density in H929 tumors. AS703026 (10 mg/kg, p.o.) inhibited tumor growth of cetuximab-resistant tumor attributed by K-ras mutation. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Kim K, et al. Blockade of the MEK/ERK signalling cascade by AS703026, a novel selective MEK1/2 inhibitor, induces pleiotropic anti-myeloma activity in vitro and in vivo. Br J Haematol, 2010, 149(4), 537-549. [2]. Yoon J, et al. MEK1/2 inhibitors AS703026 and AZD6244 may be potential therapies for KRAS mutated colorectal cancer that is resistant to EGFR monoclonal antibody therapy. Cancer Res, 2011, 71(2), 445-453. [3]. Park SJ, et al. The MEK1/2 inhibitor AS703026 circumvents resistance to the BRAF inhibitor PLX4032 in human malignant melanoma cells. Am J Med Sci. 2013 Dec;346(6):494-8. |
|
| Description | Pimasertib (AS-703026) is a highly selective, potent, ATP non-competitive allosteric inhibitor of MEK1/2 with IC50 values of 5 nM-2 μM in human multiple myeloma (MM) cell lines. | |||||
| Targets | MEK1/2 | |||||
| IC50 | 5 nM-2 μM | |||||
Quality Control & MSDS
- View current batch:
Chemical structure