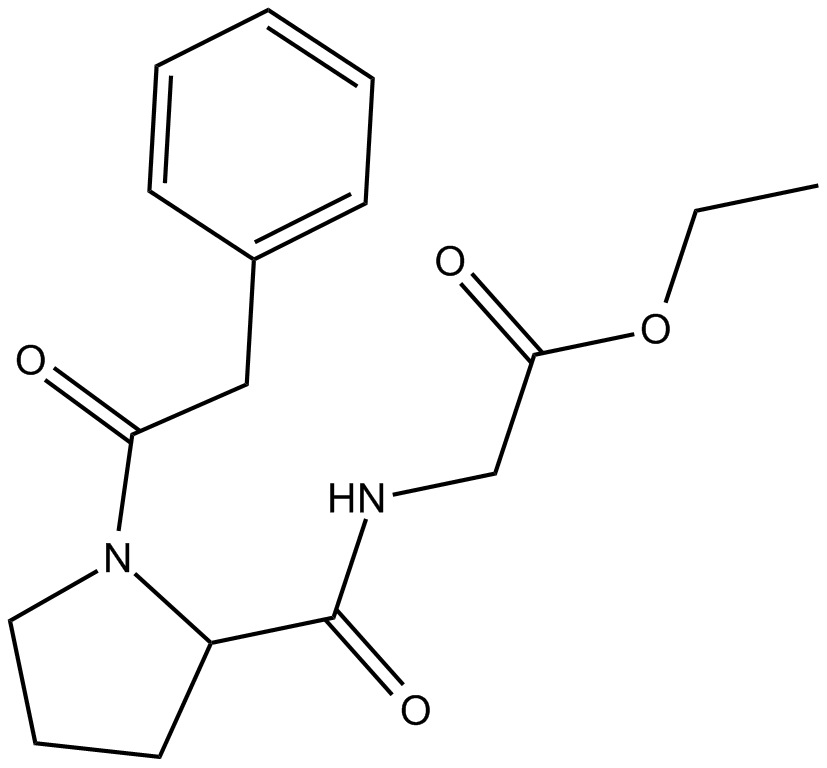
Noopept
Catalog No.
A3663
Nootropic and neuroprotective agent
Featured Products
Description:
IC50 Value: N/A
Noopept (GVS-111) is a medication promoted and prescribed in Russia and neighbouring countries as a nootropic.
in vitro: Nooglutil exhibits pharmacologically significant competition with a selective agonist of AMPA receptors ([G-3H]Ro 48-8587) for the receptor binding sites (with IC50 = 6.4 +/- 0.2 microM), while the competition of noopept for these receptor binding sites was lower by an order of magnitude (IC50 = 80 +/- 5.6 microM) [1]. GVS-111 significantly increased neuronal survival after H(2)O(2)-treatment displaying a dose-dependent neuroprotective activity from 10 nM to 100 microM, and an IC(50) value of 1.21+/-0.07 microM. GVS-111 inhibited the accumulation of intracellular free radicals and lipid peroxidation damage in neurons treated with H(2)O(2) or FeSO(4), suggesting an antioxidant mechanism of action [2].
in vivo: N-Phenylacetyl-L-prolylglycine ethyl ester (GVS-111) administered intravenously at a dose of 0.5 mg/kg/day, for the first time 1 h after ischaemic lesion and then for 9 post-operative days, with the last administration 15 min before testing, attenuated the deficit [3]. GVS-111 itself was not found in rat brain 1 h after 5 mg/kg i.p. administration up to limit of detection (LOD) under high performance liquid chromatography (HPLC) conditions [4]. The most pronounced antiinflammatory effect of dipeptide was observed on the model of adjuvant arthritis in rats, where the drug administered over 25 days in a daily dose of 0.5 mg/kg (i.m.) or 5 mg/kg (p.o.) significantly reduced the chronic immune inflammation (on the 12th day, by 94.0 and 74.1%, respectively) [5].
Toxicity: Noopept administered in this dose range induced no irreversible pathologic changes in the organs and systems studied and exhibited no allergenic, immunotoxic, and mutagen activity [6].
Clinical trial: Discontinued
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 318.37 |
| Cas No. | 157115-85-0 |
| Formula | C17H22N2O4 |
| Synonyms | GVS-111;GVS111;GVS 111;SGS-111;SGS 111;SGS111 |
| Solubility | Soluble in DMSO |
| Chemical Name | ethyl 2-(1-(2-phenylacetyl)pyrrolidine-2-carboxamido)acetate |
| SDF | Download SDF |
| Canonical SMILES | CCOC(CNC(C1CCCN1C(CC2=CC=CC=C2)=O)=O)=O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
-
Purity = 98.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
Chemical structure