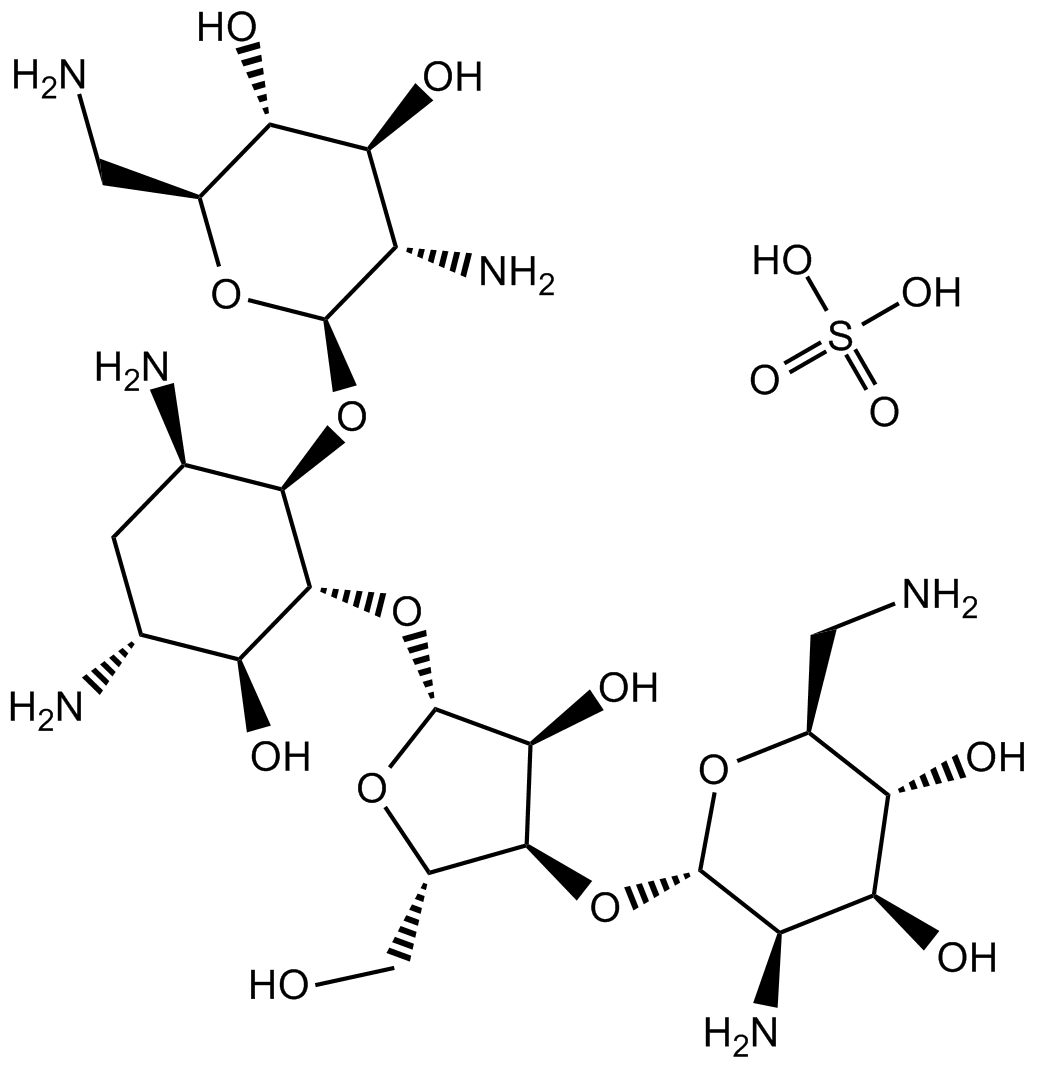
Neomycin sulfate
Neomycin sulfate (CAS 1405-10-3) is an aminoglycoside antibiotic that interacts with nucleic acid structures and ion channels, influencing several biological processes. It inhibits hammerhead ribozyme cleavage reactions by preferentially stabilizing the ribozyme-substrate ground-state complex, thereby impeding catalytic turnover. In HIV-1 research, neomycin disrupts the interaction between Tat protein and the viral RNA TAR element via an allosteric, noncompetitive mechanism. Additionally, neomycin binds specifically to DNA triplex structures, particularly stabilizing TAT triplets. It also demonstrates voltage- and concentration-dependent blockage of ryanodine receptor channels, mainly from the luminal side. These properties make neomycin sulfate valuable for mechanistic studies involving RNA/DNA structure interactions and channel functions.
- 1. Ning Wang, Chengyang Sun, et al. "Gut microbiota-derived indoleacetic acid attenuates neuroinflammation and neurodegeneration in glaucoma through ahr/rage pathway." J Neuroinflammation. 2025 Jul 10;22(1):179 PMID: 40640940
- 2. Shuiping Yan, Jian Zheng, et al. "Effect of Shufeng xingbi Therapy on Th1/Th2 immune balance and intestinal flora in rats with allergic rhinitis." bioRxiv. March 29, 2025.
- 3. Ivana Mladin. "Metode preoperativne intranazalne dekolonizacije u svrhu redukcije infekcija operacijskog polja." University Department of Health Studies. 2024-12-06
- 4. Fuqiang Ma, Weiming Zhang, et al. "Epimedii Folium decoction ameliorates osteoporosis in mice through NLRP3/caspase-1/IL-1β signalling pathway and gut-bone axis." Int Immunopharmacol. 2024 Aug 20:137:112472. PMID: 38897131
- 5. Su D, Guo X, et al. "Tumor-neuroglia interaction promotes pancreatic cancer metastasis." Theranostics. 2020;10(11):5029-5047. PMID:32308766
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 712.72 |
| Cas No. | 1405-10-3 |
| Formula | C23H46N6O13·H2SO4 |
| Solubility | ≥33.75 mg/mL in H2O; insoluble in DMSO; insoluble in EtOH |
| Chemical Name | (2R,3R,4R,5R,6R)-5-amino-2-(aminomethyl)-6-[(1R,2S,3S,4R,6S)-4,6-diamino-2-[(2S,3R,4R,5R)-4-[(3R,4R,5R,6S)-3-amino-6-(aminomethyl)-4,5-dihydroxyoxan-2-yl]oxy-3-hydroxy-5-(hydroxymethyl)oxolan-2-yl]oxy-3-hydroxycyclohexyl]oxyoxane-3,4-diol;sulfuric acid |
| Canonical SMILES | C1C(C(C(C(C1N)OC2C(C(C(C(O2)CN)O)O)N)OC3C(C(C(O3)CO)OC4C(C(C(C(O4)CN)O)O)N)O)O)N.OS(=O)(=O)O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure