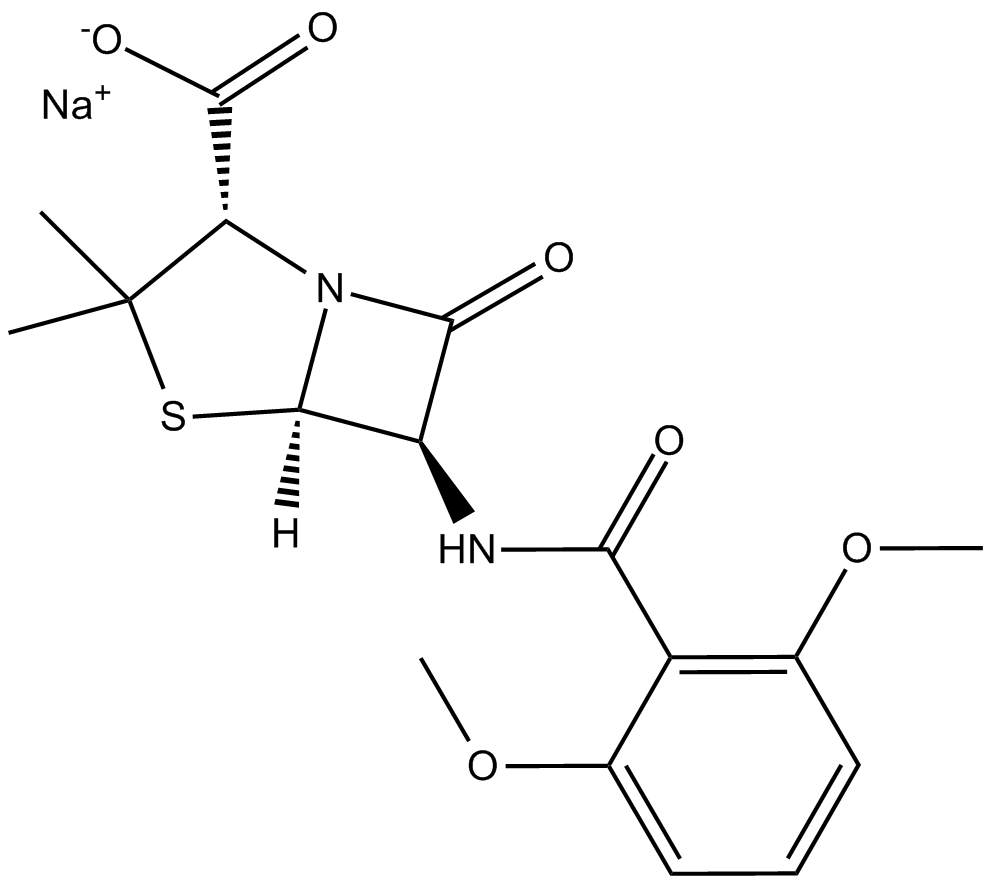
Methicillin sodium salt
Methicillin sodium salt (CAS No. 132-92-3) is the sodium salt form of Methicillin (CAS No. 61-32-5), a semi-synthetic anti-staphylococcal penicillin antibiotic. Its core target is bacterial penicillin-binding proteins (PBPs); it exerts a bactericidal effect by inhibiting peptidoglycan cross-linking to block bacterial cell wall synthesis. It is effective against methicillin-sensitive Staphylococcus aureus (MSSA) but ineffective against methicillin-resistant Staphylococcus aureus (MRSA) carrying the mecA gene (which encodes low-affinity PBP2a). There are no clear IC₅₀/EC₅₀ data, and the minimum inhibitory concentration (MIC) is the main activity indicator: the MIC range against MSSA is 0.125~2 μg/mL, while the MIC against MRSA is >8 μg/mL (defined as resistant). The commonly used concentration in laboratory settings is 0.06~16 μg/mL (agar/broth dilution method for drug susceptibility testing). The clinically effective therapeutic concentration corresponds to intravenous injection doses: for adults, 4~12 g daily (administered in 4 divided doses); for children, 50~100 mg/kg daily (administered in 4 divided doses), with a peak blood concentration of 10~40 μg/mL. It is indicated for skin and soft tissue infections, sepsis, pneumonia, and other conditions caused by MSSA. Currently, due to the prevalence of MRSA, its clinical application has been significantly reduced. Common adverse reactions include allergic reactions and gastrointestinal discomfort, and cross-allergy risks need to be vigilant.
References:
[1] Turner NA, Sharma-Kuinkel BK, Maskarinec SA, Eichenberger EM, Shah PP, Carugati M, Holland TL, Fowler VG Jr. Methicillin-resistant Staphylococcus aureus: an overview of basic and clinical research. Nat Rev Microbiol. 2019 Apr;17(4):203-218. doi: 10.1038/s41579-018-0147-4. PMID: 30737488; PMCID: PMC6939889.
| Storage | Store at -20°C |
| M.Wt | 402.4 |
| Cas No. | 132-92-3 |
| Formula | C17H19N2O6S·Na |
| Synonyms | BRL 1241,SQ 16,123,X 1497 |
| Solubility | ≥14.4 mg/mL in DMSO |
| Chemical Name | (2S,5R,6R)-6-[(2,6-dimethoxybenzoyl)amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid, monosodium salt |
| Canonical SMILES | CC(C)([C@@H]1C([O-])=O)S[C@H]([C@@H]2NC(c(c(OC)ccc3)c3OC)=O)N1C2=O.[Na+] |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure