LY335979 (Zosuquidar 3HCL)
LY335979 is a selective inhibitor of P-Gp with IC50 value of 1.2 nM [1, 2].
P-gp (P-glycoprotein) is a member of ATP-binding cassette (ABC) transporters and plays a pivotal role in pumping many foreign substances out of cells. It has been reported that abnormal expression of P-Gp is correlated with the multidrug resistance of tumor cells [3].
LY335979 is a potent P-Gp inhibitor and has a different selectivity with the reported P-Gp inhibitor cyclosporin A or verapamil. In drug-resistant cell line HL60/VCR with highly expression of P-Gp, LY335979 exhibited highly restore ability of P-Gp than cyclosporin A or verapamil and the IC 50 value of 1.2 nM [1]. When tested with a panel of cell lines over-expressed P-Gp (CEM/VLB100, MCF-7/ADR, 2780AD, P388/ADR, and UCLA-P3.003VLB), administration of LY335979 reversed the cells resistance to Vinblastine, Doxorubicin, Btoposide and Taxol by inhibiting P-Gp activity [2].
In female nude mice model with UCLA-P3.003VLB MDR tumor cells subcutaneous xenograft, pre-treated with LY335979 (30mg/kg) restored tumor cells sensitivity to Taxol (20 mg/kg) which combination markedly suppressed solid tumor growth compared with control group [2].
References:
1. Green, L.J., P. Marder, and C.A. Slapak, Modulation by LY335979 of P-glycoprotein function in multidrug-resistant cell lines and human natural killer cells. Biochem Pharmacol, 2001. 61(11): p. 1393-9.
2. Dantzig, A.H., et al., Reversal of P-glycoprotein-mediated multidrug resistance by a potent cyclopropyldibenzosuberane modulator, LY335979. Cancer Res, 1996. 56(18): p. 4171-9.
3. Hu, T., et al., Reversal of P-glycoprotein (P-gp) mediated multidrug resistance in colon cancer cells by cryptotanshinone and dihydrotanshinone of Salvia miltiorrhiza. Phytomedicine, 2014. 21(11): p. 1264-72.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 637.0 |
| Cas No. | 167465-36-3 |
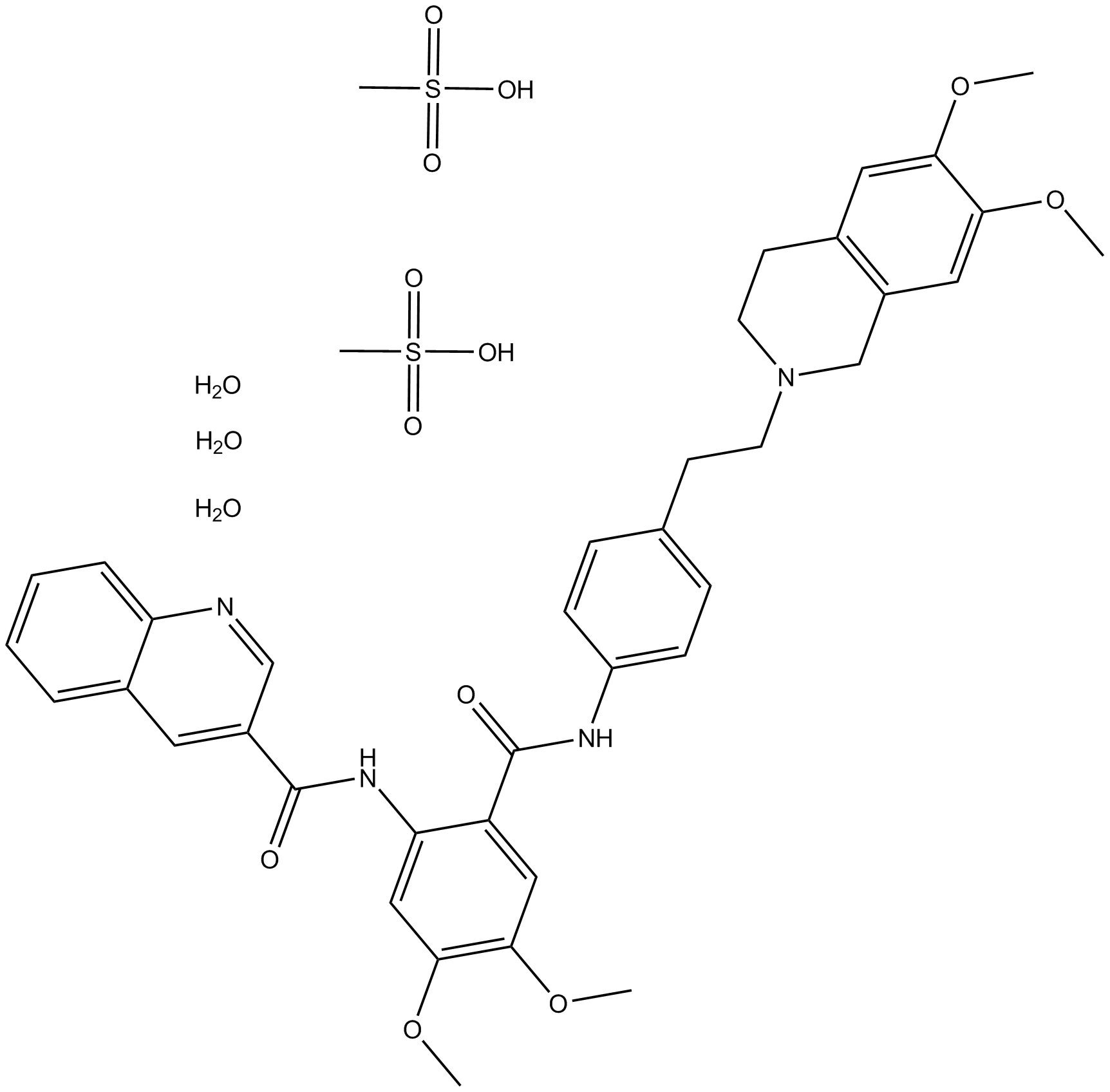
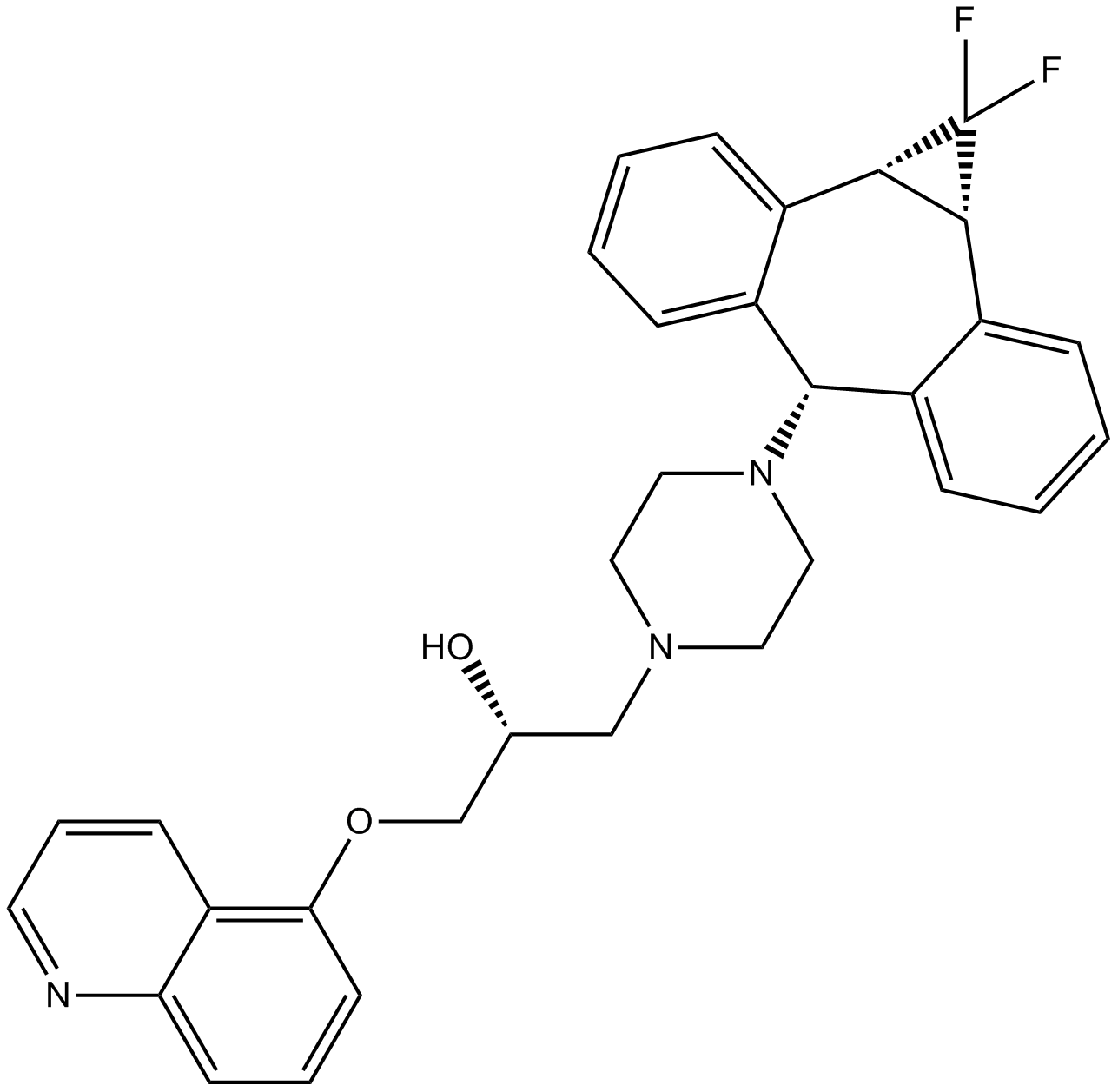
| Formula | C32H34Cl3F2N3O2 |
| Synonyms | RS 33295-198; Zosuquidar trihydrochloride; LY335979; LY-335979 |
| Solubility | insoluble in EtOH; ≥17.1 mg/mL in DMSO; ≥31.9 mg/mL in H2O |
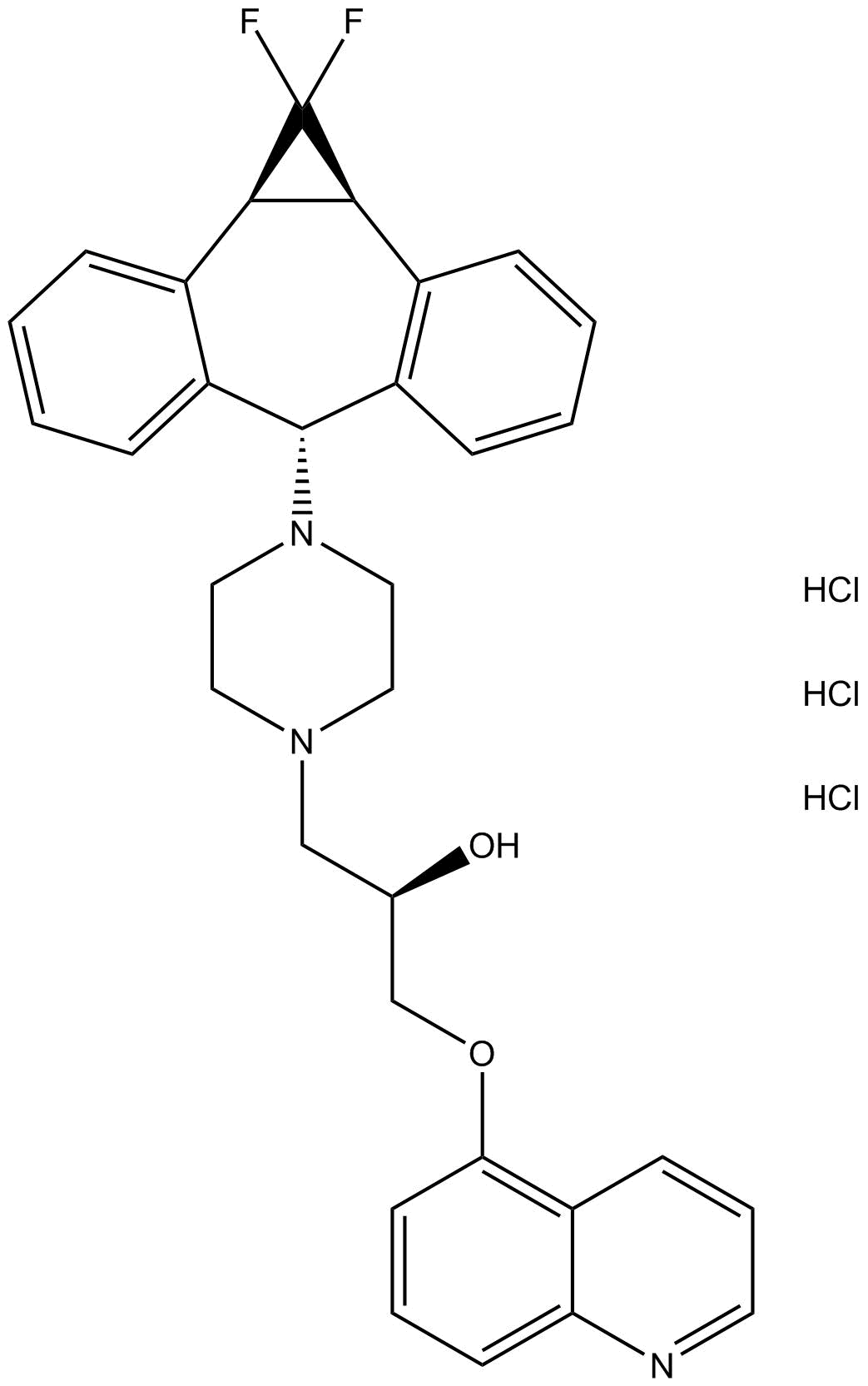
| Chemical Name | (R)-1-(4-((1aR,6s,10bS)-1,1-difluoro-1,1a,6,10b-tetrahydrodibenzo[a,e]cyclopropa[c][7]annulen-6-yl)piperazin-1-yl)-3-(quinolin-5-yloxy)propan-2-ol trihydrochloride |
| SDF | Download SDF |
| Canonical SMILES | FC1([C@H]2[C@@H]1C3=C([C@@H](C4=CC=CC=C42)N5CCN(CC5)C[C@H](COC6=C7C=CC=NC7=CC=C6)O)C=CC=C3)F.Cl.Cl.Cl |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Kinase experiment [1]: | |
|
ATPase assay |
P-Glycoprotein ATPase activity was measured by the liberation of inorganic phosphate from ATP. The assay was measured in a 96-well plate for 90 mins at 37 °C. Membranes (8 ~ 10 μg protein) were incubated in a total volume of 100 μL of buffer A containing 5 mM sodium azide, 1 mM ouabain, 1 mM EGTA, 3 mM ATP, an ATP regenerating system composed of 5 mM phosphoenolpyruvate, and 3.6 units/mL pyruvate kinase in the presence and absence of 1 mM sodium vanadate. Pgp-ATPase activity was defined as the vanadate-sensitive portion of the total ATPase activity. Plates were read 3 mins after the addition of the detection solution. The absorbance was measured at 690 nm by a microtiter dish reader. A phosphate standard curve was used to calculate the μmol of phosphate formed. Samples were measured in triplicate. |
| Cell experiment [1]: | |
|
Cell lines |
CEM/VLB100, MCF-7/ADR, 2780AD, P388/ADR and UCLA-P3.003VLB cells |
|
Preparation method |
The solubility of this compound in DMSO is > 10 mM. General tips for obtaining a higher concentration: Please warm the tube at 37 °C for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below - 20 °C for several months. |
|
Reacting condition |
0.1 ~ 2 μM |
|
Applications |
LY335979 was an effective modulator, with maximal activity in reversing the sensitivity of resistant cells to the various anticancer drugs (Vinblastine, Doxorubicin, Btoposide and Taxol) at the concentrations of 0.1 ~ 2 μM. At the concentration of 0.05 μM, the modulator activity of LY335979 was diminished by about 50%. |
| Animal experiment [1]: | |
|
Animal models |
Nude mice bearing UCLA-P3.003VLB tumor cells |
|
Dosage form |
30 mg/kg; i.p. |
|
Applications |
In a Pgp-expressing human non-small cell lung carcinoma xenograft model, the combination therapy of 20 mg/kg Taxol and 30 mg/kg LY335979 significantly suppressed solid tumor growth at days 12 and 19. In addition, no increased weight loss was observed. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Dantzig, A.H., et al., Reversal of P-glycoprotein-mediated multidrug resistance by a potent cyclopropyldibenzosuberane modulator, LY335979. Cancer Res, 1996. 56(18): p. 4171-9. |
|
| Description | Zosuquidar (LY335979) is a potent modulator of P-glycoprotein-mediated multi-drug resistance with a Ki value of 60 nM. | |||||
| Targets | P-gp | |||||
| IC50 | 60 nM(Ki) | |||||
Quality Control & MSDS
- View current batch:
Chemical structure
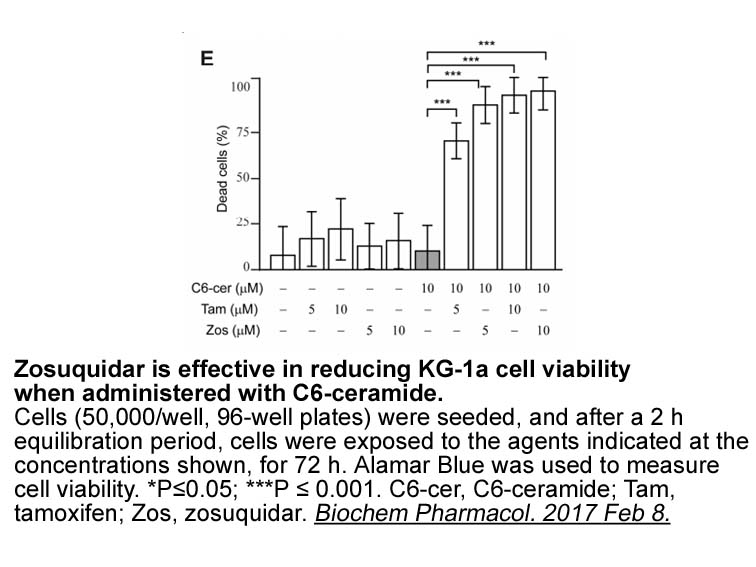
Related Biological Data