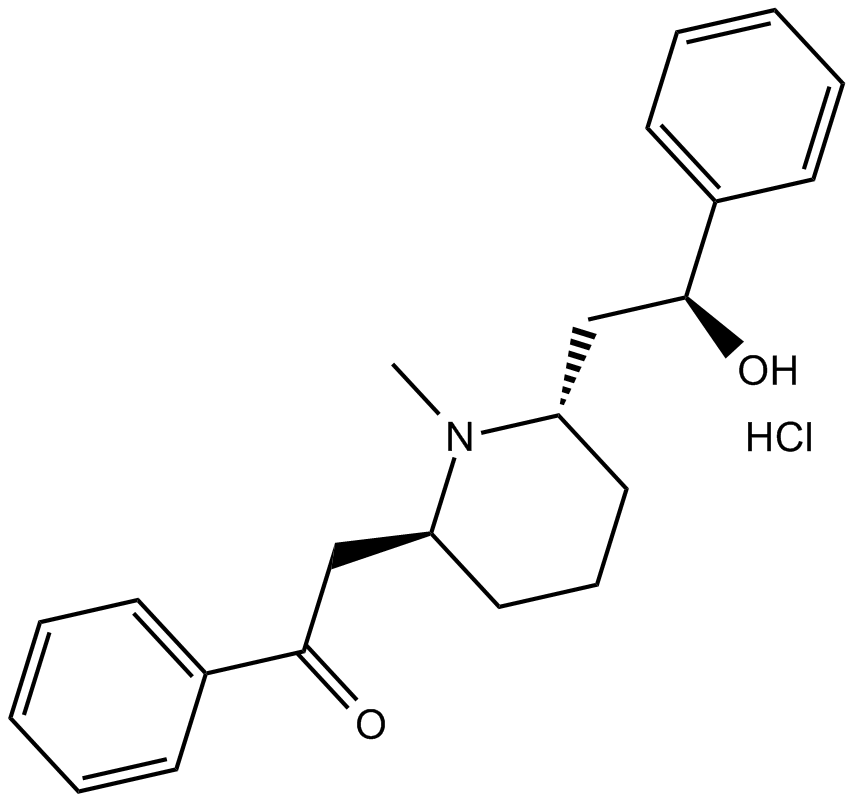
(-)-Lobeline hydrochloride
(-)-Lobeline hydrochloride is a modulator of nicotinic acetylcholine receptors (nAChRs) with various binding affinity. It displays very low affinity (Ki > 10000nM) for α7 receptors, but high affinity for rodent and human α4β2 receptors (Ki=1.4-2 nM). [1] It inhibits α7 nAChRs with an IC50 value of 8.5 µM. [2]
nAChRs are composed of different subunits, and multiple populations of nAChRs exist; brain nAChRs are primarily of the α4β2 and α7 type. [1]
In vitro, clastogenicity of lobeline and possible interactions between lobeline and ethyl alcohol were investigated in a mutagen-sensitivity assay on cultures of human lymphoblastoid cell lines. Lobeline alone was not clastogenic, but there was a marked increase in genetic damage resulting from a coclastogenic interaction between lobeline and ethyl alcohol. [5]
In vivo, male C57BL/6J mice were individually housed and acclimatized to 10% alcohol. Lobeline is a partial nicotinic agonist that attenuates alcohol consumption and preference in male C57BL/6J mice. [3] CF-1 male mice received an intraperitoneal injection of lobeline (5 or 10mg/kg). Lobeline did not show genotoxic or mutagenic effects and did not increase the ethanol-induced genotoxic effects in blood. Lobeline also protected blood cells against oxidative damage induced by hydrogen peroxide. [4]
Reference:
1. Flammia D1, Dukat M, Damaj MI, et al. Lobeline: structure-affinity investigation of nicotinic acetylcholinergic receptor binding. J Med Chem. 1999 Sep 9; 42 (18):3726-31.
2. Briggs CA, McKenna DG. Activation and inhibition of the human alpha7 nicotinic acetylcholine receptor by agonists. Neuropharmacology. 1998 Sep; 37 (9):1095-102.
3. Farook JM, Lewis B, Gaddis JG, et al. Lobeline, a nicotinic partial agonist attenuates alcohol consumption and preference in male C57BL/6J mice. Physiol Behav. 2009 Jun 22; 97 (3-4):503-6.
4. da Costa E Silva LD, Rodrigues LC1, Dos Santos VR1.et al. Evaluation of mutagenic and genotoxic activities of lobeline and its modulation on genomic instability induced by ethanol. Life Sci. 2014 May 17; 103 (2):73-8.
5. Brown NM, Trizna Z, Pathak S. Clastogenic interactions between lobeline sulfate and ethyl alcohol: a cytogenetic study. Anticancer Res. 1992 Sep-Oct; 12 (5):1467-9.
| Physical Appearance | A solid |
| Storage | Store at RT |
| M.Wt | 373.92 |
| Cas No. | 134-63-4 |
| Formula | C22H27NO2·HCl |
| Solubility | ≥37.5 mg/mL in EtOH with ultrasonic; ≥37.8 mg/mL in H2O with gentle warming and ultrasonic; ≥74.8 mg/mL in DMSO |
| Chemical Name | 2-((2S,6S)-6-((S)-2-hydroxy-2-phenylethyl)-1-methylpiperidin-2-yl)-1-phenylethanone hydrochloride |
| SDF | Download SDF |
| Canonical SMILES | O[C@H](C1=CC=CC=C1)C[C@H]2N(C)[C@H](CC(C3=CC=CC=C3)=O)CCC2.Cl |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure