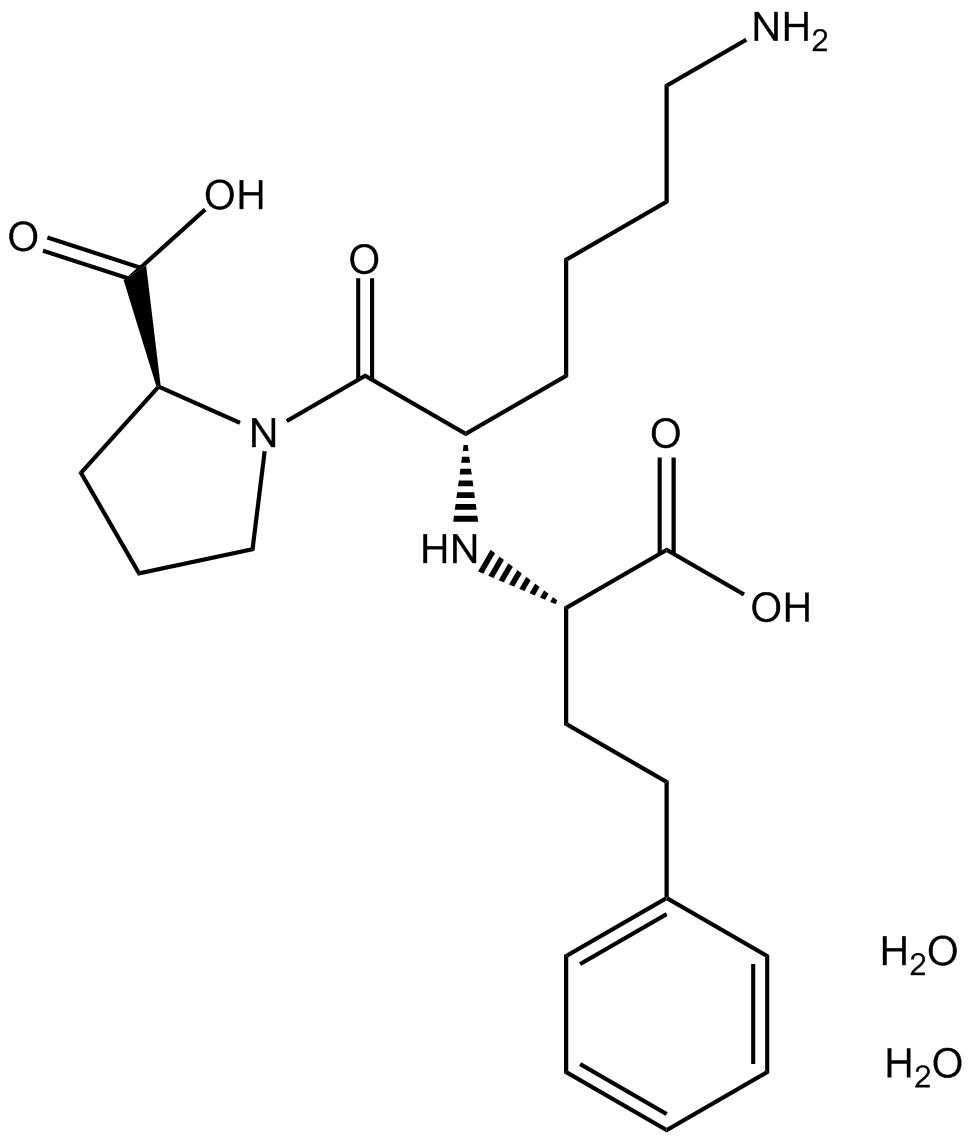
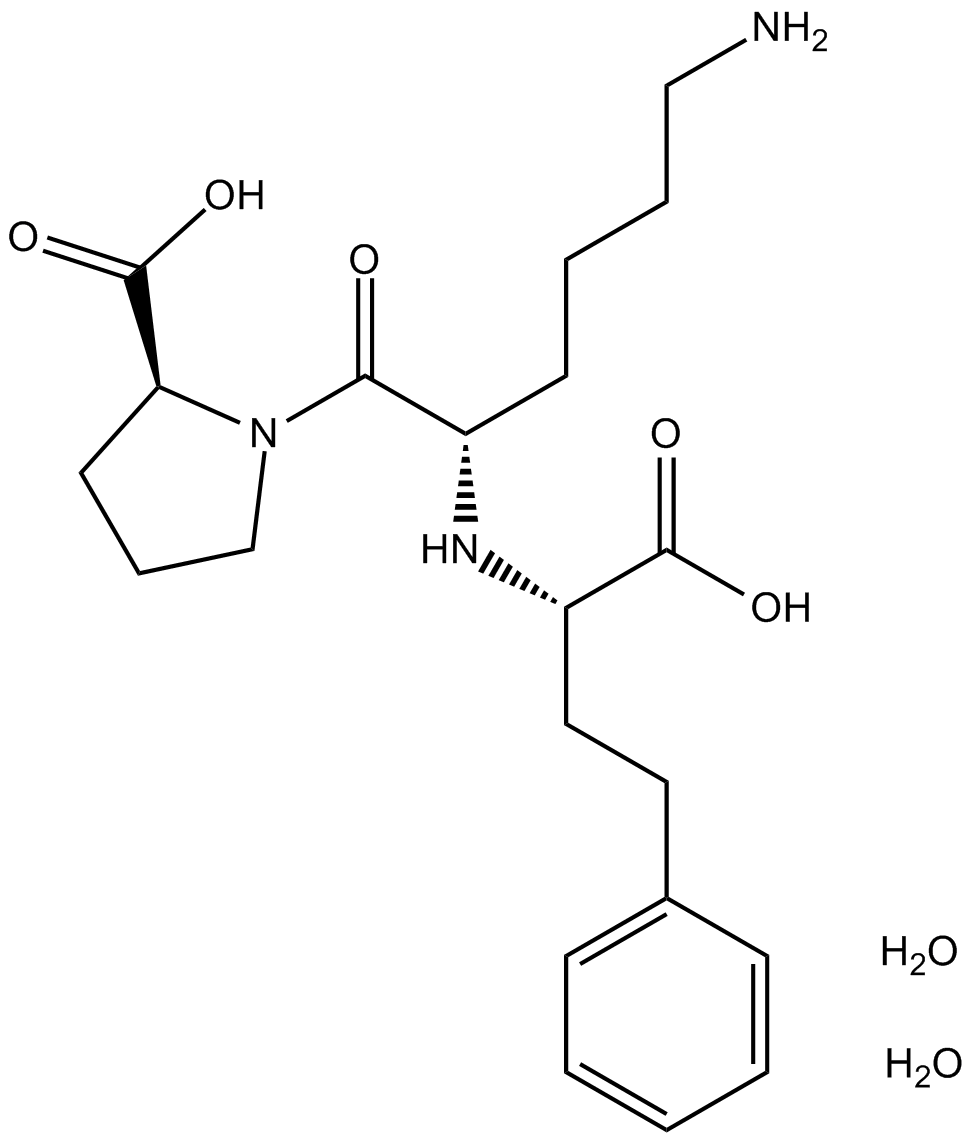
Lisinopril dihydrate
Lisinopril is a long-acting inhibitor of angiotensin converting enzyme (ACE) with IC50 value of 4.7nM [1].
Lisinopril is a lysine analogue of MK 421. Both of them are the inhibitors of ACE and found to significantly inhibit the pressor response to exogenous angiotensin I. The administration of lisinopril can lower the diastolic and systolic blood pressure and higher the heart rate. Lisinopril is found to reduce the activity of plasma angiotensin converting enzyme markedly. It increases the level of plasma rennin while decreases the levels of plasma Angiotensin II and aldosterone. Lisinopril is a drug now used for many disorders including hypertension, heart failure, acute myocardial infarction and diabetic nephropathy. lisinopril dehydrate is the commercial form of this drug [2, 3].
References:
[1] Arora P K, Chauhan A. ACE INHIBITORS: A COMPREHENSIVE REVIEW. International Journal of Pharmaceutical Sciences & Research, 2013, 4(2).
[2] Brunner DB, Desponds G, Biollaz J, Keller I, Ferber F, Gavras H, Brunner HR, Schelling JL. Effect of a new angiotensin converting enzyme inhibitor MK 421 and its lysine analogue on the components of the renin system in healthy subjects. Br J Clin Pharmacol. 1981 May;11(5):461-7.
[3] Sorrenti M, Catenacci L, Cruickshank DL, Caira MR. Lisinopril dihydrate: single-crystal x-ray structure and physicochemical characterization of derived solid forms. J Pharm Sci. 2013 Oct;102(10):3596-603.
| Physical Appearance | A solid |
| Storage | Desiccate at RT |
| M.Wt | 441.52 |
| Cas No. | 83915-83-7 |
| Formula | C21H35N3O7 |
| Solubility | insoluble in EtOH; ≥2.46 mg/mL in H2O with gentle warming and ultrasonic |
| Chemical Name | (2S)-1-[(2S)-6-amino-2-[[(1S)-1-carboxy-3-phenylpropyl]amino]hexanoyl]pyrrolidine-2-carboxylic acid;dihydrate |
| SDF | Download SDF |
| Canonical SMILES | C1CC(N(C1)C(=O)C(CCCCN)NC(CCC2=CC=CC=C2)C(=O)O)C(=O)O.O.O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure