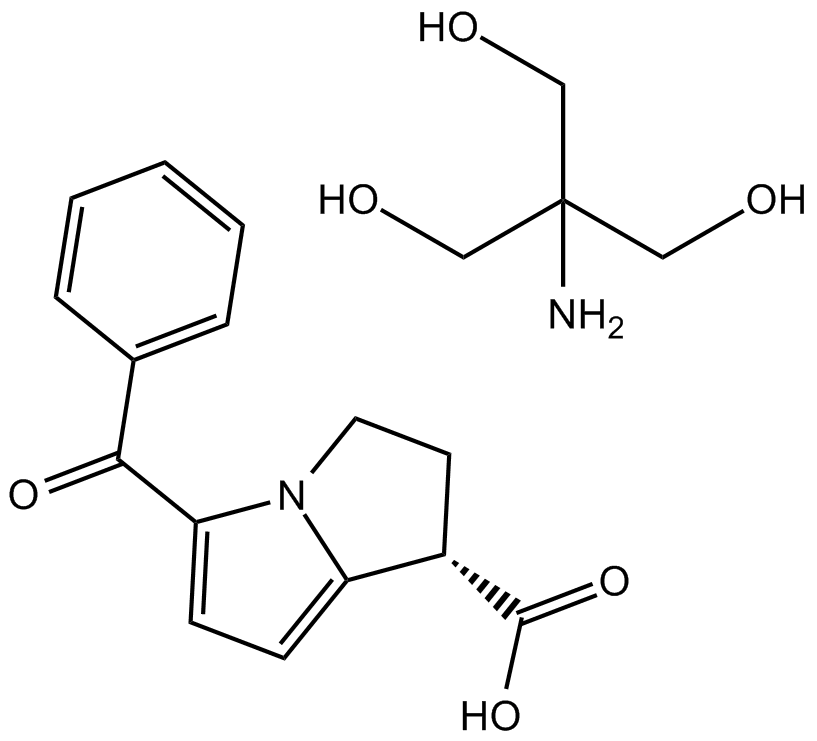
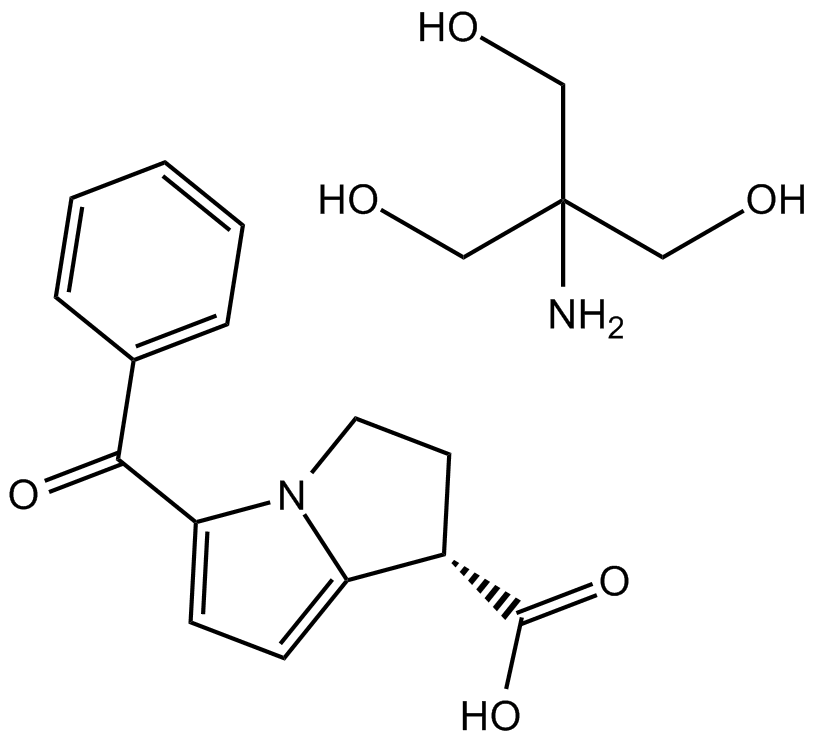
Ketorolac tromethamine salt
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 376.4 |
| Cas No. | 74103-07-4 |
| Formula | C19H24N2O6 |
| Solubility | ≥13.1 mg/mL in DMSO; ≥9.9 mg/mL in EtOH with ultrasonic; ≥92.8 mg/mL in H2O |
| Chemical Name | 2-amino-2-(hydroxymethyl)propane-1,3-diol;5-benzoyl-2,3-dihydro-1H-pyrrolizine-1-carboxylic acid |
| SDF | Download SDF |
| Canonical SMILES | C1CN2C(=CC=C2C(=O)C3=CC=CC=C3)C1C(=O)O.C(C(CO)(CO)N)O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1]: | |
|
Cell lines |
HEL, Mono Mac 6 and RAW 264.7 cells |
|
Preparation method |
The solubility of this compound in DMSO is > 10 mM. General tips for obtaining a higher concentration: Please warm the tube at 37 °C for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below - 20 °C for several months. |
|
Reacting condition |
0.025 ~ 300 μM |
|
Applications |
In HEL cells (COX-1) and LPS-induced Mono Mac 6 cells (COX-2), Ketorolac tromethamine salt inhibited eicosanoid formation with the IC50 value of 0.025 μM and 0.039 μM, respectively. However, in supernatants of LPS-induced RAW 264.7 cells, it did not significantly inhibit NO accumulation at the dose up to 300 μM. |
| Animal experiment [2]: | |
|
Animal models |
Male Wistar rats |
|
Dosage form |
0.3 ~ 30 mg/kg; p.o. |
|
Applications |
At all doses, Ketorolac tromethamine salt significantly inhibited COX-1 activity and gastric PG synthesis. At the doses ≥ 1 mg/kg, Ketorolac tromethamine salt inhibited COX-1 activity by 95% and gastric PG synthesis by > 88%, without causing obvious gastric damage. At the dose ≤ 3 mg/kg, Ketorolac tromethamine salt did not significantly affect COX-2 activity, but at the doses of 10 and 30 mg/kg, it inhibited COX-2 activity by 75% and 91%, respectively. Meanwhile, it caused significant gastric damage as well. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Berg J, Fellier H, Christoph T, Grarup J, Stimmeder D. The analgesic NSAID lornoxicam inhibits cyclooxygenase (COX)-1/-2, inducible nitric oxide synthase (iNOS), and the formation of interleukin (IL)-6 in vitro. Inflamm Res. 1999 Jul;48(7):369-79. [2]. Wallace JL, McKnight W, Reuter BK, Vergnolle N. NSAID-induced gastric damage in rats: requirement for inhibition of both cyclooxygenase 1 and 2. Gastroenterology. 2000 Sep;119(3):706-14. | |
Quality Control & MSDS
- View current batch:
Chemical structure