IU1
IU1 is a potent and selective small-molecule inhibitor of Usp14, a proteasome-associated deubiquitinating enzyme in habiting the degradation of ubiquitin-protein conjugates, with the inhibition constant IC50 ranging from 4 to 5 μM. Structural analysis reveals IU1 is an active-site-directed thiol protease inhibitor that binds to the activated form of Usp14 preventing it docking on the proteasome in a rapid but reversible manner. According to the results of previous studies, it has shown that IU1 failed to inhibit Usp14 in the absence of proteasomes as well as eight deubiquitinating enzymes (DUBs) of human origin and Ub-AMC hrolysis by proteasomes lacking Usp14.
Reference
Byung-Hoon Lee, Min Jae Lee, Soyeon Park, Dong-Chan Oh, Suzanne Elsasser, Ping-Chung Chen, Carlos Gartner, Nevena Dimova, John Hanna, Steven P. Gygi, Scott M. Wilson, Randall W. King and Daniel Finley. Enhancement of proteasome activity by a small-molecule inhibitor of Usp14. Nature. 2010; 467(7312): 179-184
| Physical Appearance | A solid |
| Storage | Store at 4°C |
| M.Wt | 300.37 |
| Cas No. | 314245-33-5 |
| Formula | C18H21FN2O |
| Synonyms | Usp14 inhibitor |
| Solubility | insoluble in H2O; ≥10.9 mg/mL in DMSO; ≥81.8 mg/mL in EtOH |
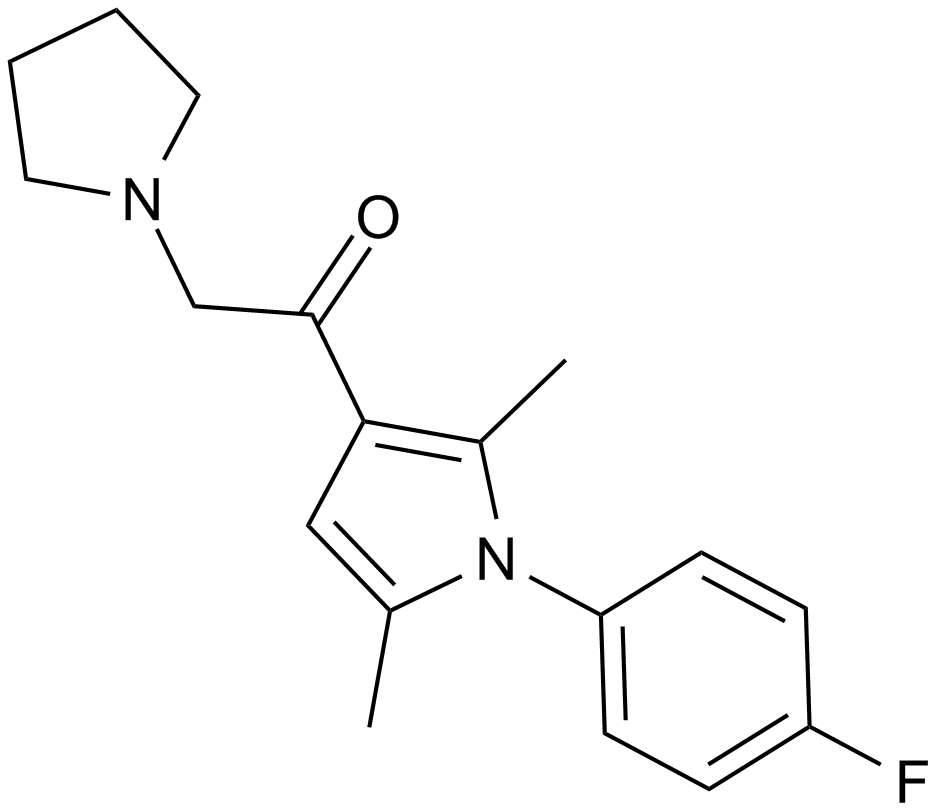
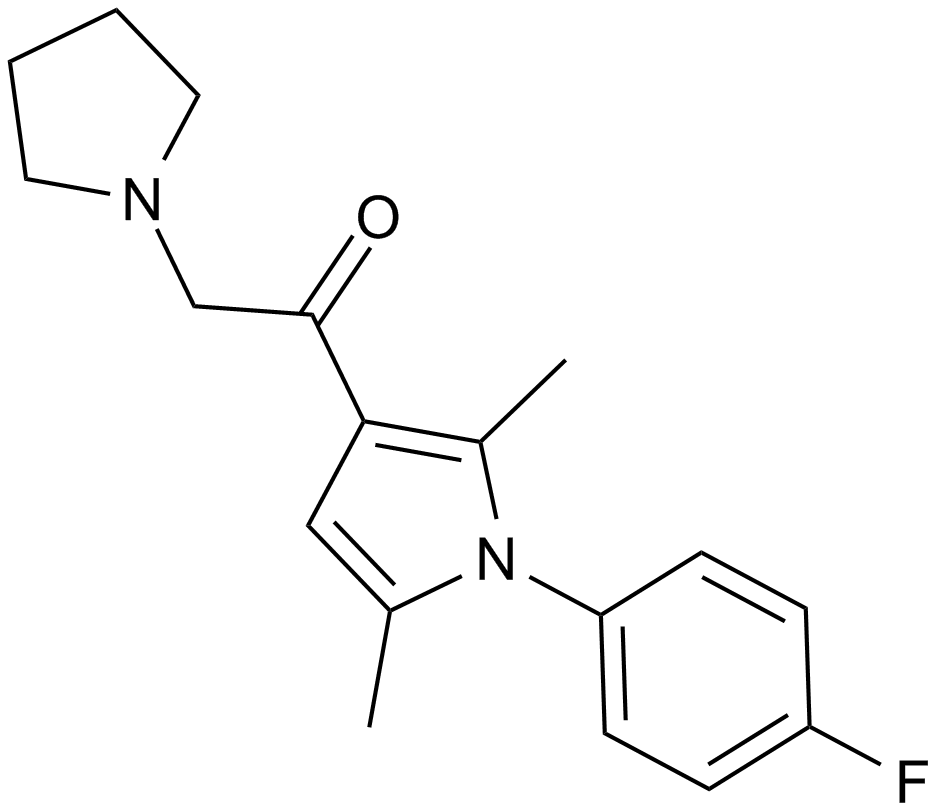
| Chemical Name | 1-[1-(4-fluorophenyl)-2,5-dimethylpyrrol-3-yl]-2-pyrrolidin-1-ylethanone |
| Canonical SMILES | Cc1cc(C(CN2CCCC2)=O)c(C)[n]1-c(cc1)ccc1F |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Description | IU1 is a cell-permeable, reversible and selective proteasome inhibitor of human USP14 with IC50 of 4.7 μM. | |||||
| Targets | USP14 | |||||
| IC50 | 4.7 μM | |||||
Quality Control & MSDS
- View current batch:
Chemical structure