GW3965
GW3965 (CAS 405911-09-3) is a selective agonist of liver X receptors (LXRs), nuclear receptor transcription factors comprising subtypes LXRα and LXRβ, crucial in lipid, cholesterol, and glucose metabolism. GW3965 activates human LXRα and LXRβ with EC50 values of approximately 190 nM and 30 nM, respectively. In cellular models, GW3965 enhances expression of target genes such as ABCA1 and IDOL, leading to suppressed LDLR expression, decreased lipid accumulation, and induced tumor cell death. Animal studies demonstrate GW3965-mediated inhibition of tumor growth and amyloid pathology, illustrating its utility in oncology and neurodegenerative disease research.
- 1. Zijie Jin, Ruotong Yin, et al. "Dapagliflozin ameliorates hepatic steatosis via suppressing LXRα-mediated synthesis of lipids and bile acids." Biochem Pharmacol. 2024 May:223:116167. PMID: 38527558
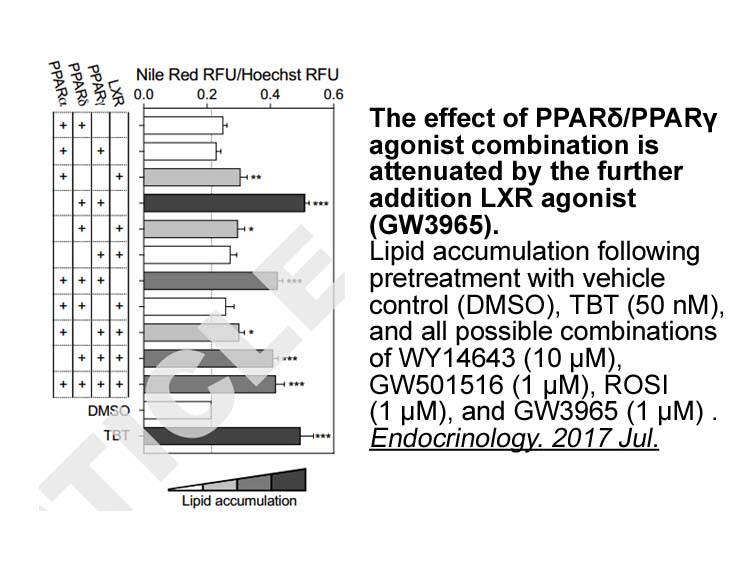
- 2. Bassem M. Shoucri, Eric S. Martinez, et al. "Retinoid X receptor activation alters the chromatin landscape to commit mesenchymal stem cells to the adipose lineage." Endocrinology. 2017 Jul.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 582.05 |
| Cas No. | 405911-09-3 |
| Formula | C33H31ClF3NO3 |
| Synonyms | GW 3965;GW-3965 |
| Solubility | Soluble in DMSO |
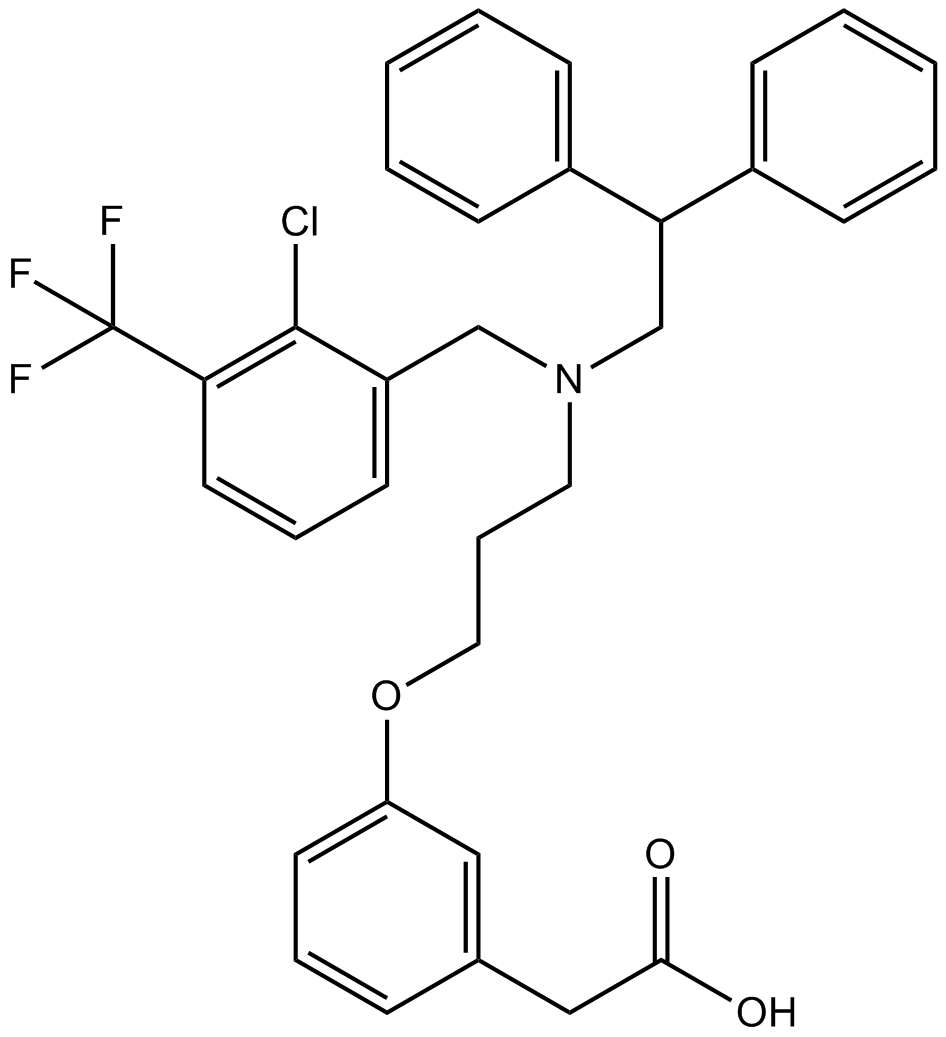
| Chemical Name | 2-[3-[3-[[2-chloro-3-(trifluoromethyl)phenyl]methyl-(2,2-diphenylethyl)amino]propoxy]phenyl]acetic acid |
| Canonical SMILES | C1=CC=C(C=C1)C(CN(CCCOC2=CC=CC(=C2)CC(=O)O)CC3=C(C(=CC=C3)C(F)(F)F)Cl)C4=CC=CC=C4 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1,2]: | |
|
Cell lines |
U87 and U87-EGFRvIII GBM cells, |
|
Preparation method |
Soluble in DMSO. General tips for obtaining a higher concentration: Please warm the tube at 37 ℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reacting condition |
2-5 μM, 4 days |
|
Applications |
In U87 and U87-EGFRvIII GBM cells, treatment with GW3965 (2-5 μM) for 4 days dose-dependently inhibited growth and promoted tumor cell death. Low-dose GW3965 (1 or 2 μM) induced ABCA1. In U87 and U87-EGFRvIII GBM cells, GW3965 (5 μM, 24h) upregulated expression of the cholesterol transporter gene ABCA1 and the E3 ubiquitin ligase IDOL and reduced LDLR levels. GW3965 (1 or 5 μM) displayed a minor inhibitory effect on fibrinogen binding and P-selectin exposure. GW3965 (10 μM) reduced the levels of fibrinogen and P-selectin on the platelet surface. |
| Animal experiment [1,2]: | |
|
Animal models |
SCID/Beige mice xenografted with isogenic human malignant glioma cells (U87, U87-EGFRvIII); C57BL/6 mice |
|
Dosage form |
Oral gavage, 40 mg/kg, daily for 12 days |
|
Application |
In mice bearing U87/EGFRvIII cells, GW3965 (40 mg/kg daily by oral gavage) for 12 days potently inhibited GBM growth, promoted tumor cell death and inhibited tumor growth. In C57BL/6 mice, GW3965 (2 mg/kg, i.v.) increased bleeding time and modulated platelet thrombus formation. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Guo D, Reinitz F, Youssef M, et al. An LXR agonist promotes glioblastoma cell death through inhibition of an EGFR/AKT/SREBP-1/LDLR–dependent pathway[J]. Cancer discovery, 2011. [2]. Spyridon M, Moraes L A, Jones C I, et al. LXR as a novel antithrombotic target[J]. Blood, 2011, 117(21): 5751-5761. |
|
| Description | GW3965 is an orally-active agonist of LXRα and LXRβ with EC50 values of 190 and 30 nM, respectively. | |||||
| Targets | LXRα | LXRβ | ||||
| IC50 | 190 nM | 30 nM | ||||
Quality Control & MSDS
- View current batch:
-
Purity = 98.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
- Datasheet
Chemical structure
Related Biological Data