GSK621
GSK621 is a specific and potent agonist of AMP-activated protein kinase (AMPK) with IC50 values ranging from 13 to 30 µM for cell lines [1].
AMPK is a heterotrimeric serine/threonine kinase. It is a sensor of cellular energy. It regulates multiple cellular metabolic pathways. Activation of AMPK inactivates the phosphorylation of acetyl-Coa carboxylase (ACC), and hence inhibits the biosynthesis of fatty acid. Activation of AMPK also inhibits protein synthesis that is dependent on mammalian target of rapamycin complex 1. AMPK also promotes autophagy, fatty acid oxidation, glucose uptake and glycolysis [1].
In cells, at the opposite, GSK621 showed more potency to activate AMPK than A-769662, based on the ACC phosphorylation level. In MOLM-14 cells, 200 µM A-769662 is just as potent as 30 µM GSK621 to induce the phosphorylation of ULK1 (S555) and ACC (S79), two direct AMPK substrates. In AML cell lines (OCI-AML3, HL-60 and MOLM-14) and primary AML samples, the phosphorylation at AMPKα T172, a marker of AMPK activation, was markedly increased, and the phosphorylation of ULK1 (S555) and ACC (S79) was stimulated by GSK621 [1].
In animals with xenograft MOLM-14 cells, intraperitoneal injection of GSK621 at 30 mg/kg twice daily reduced leukemia growth and markedly extended survival compared to treatment with 10 mg/kg GSK621 twice daily or vehicle. These results correlated to increased AMPK activity indicated by the induction of apoptosis and increased ACC S79 phosphorylation [1].
Reference:
[1]. Sujobert P, Poulain L, Paubelle E, et al. Co-activation of AMPK and mTORC1 induces cytotoxicity in acute myeloid leukemia. Cell reports, 2015, 11(9): 1446-1457.
- 1. Jun Xiao, Shuang Wang, et al. "25-Hydroxycholesterol regulates lysosome AMP kinase activation and metabolic reprogramming to educate immunosuppressive macrophages." Immunity. 2024 May 14;57(5):1087-1104.e7. PMID: 38640930
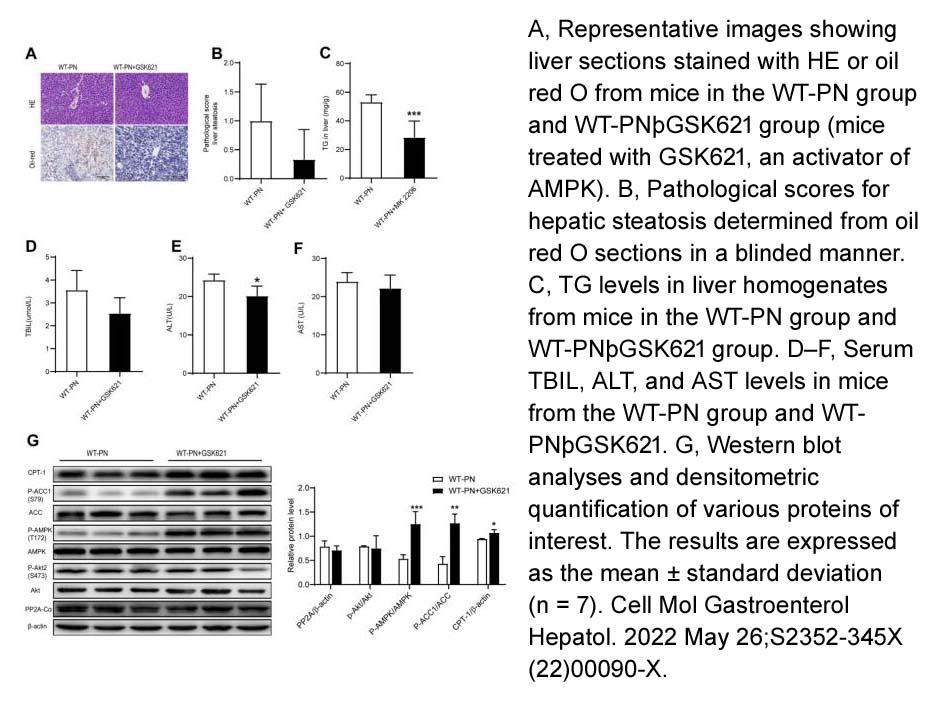
- 2. Gulisudumu Maitiabula, Feng Tian, et al. " Liver PP2A-Cα protects from parenteral nutrition-associated hepatic steatosis." Cell Mol Gastroenterol Hepatol. 2022 May 26;S2352-345X(22)00090-X. PMID: 35643235
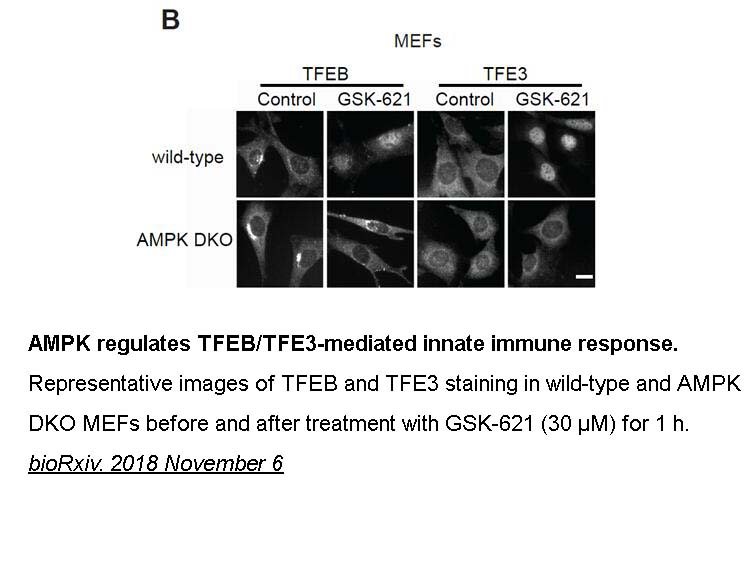
- 3. Leeanna El-Houjeiri, Elite Possik, et al. "The transcription factors TFEB and TFE3 link the FLCN-AMPK signaling axis to innate immune response and pathogen resistance."bioRxiv. 2018 November 6.
| Physical Appearance | A crystalline solid |
| Storage | Store at 2-8°C |
| M.Wt | 489.91 |
| Cas No. | 1346607-05-3 |
| Formula | C26H20ClN3O5 |
| Solubility | insoluble in H2O; insoluble in EtOH; ≥28.5 mg/mL in DMSO |
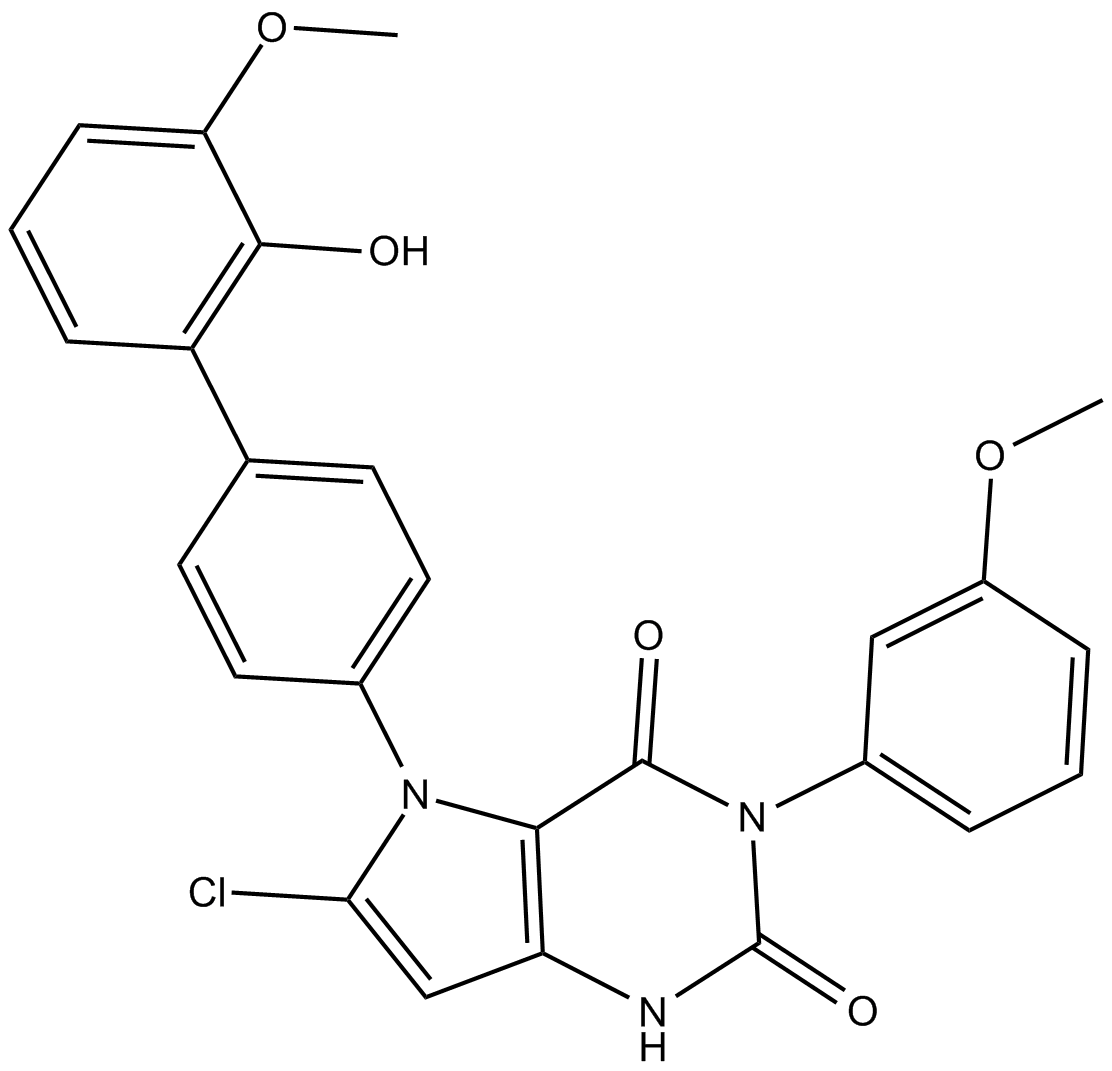
| Chemical Name | 6-chloro-5-(2'-hydroxy-3'-methoxy-[1,1'-biphenyl]-4-yl)-3-(3-methoxyphenyl)-1H-pyrrolo[3,2-d]pyrimidine-2,4(3H,5H)-dione |
| SDF | Download SDF |
| Canonical SMILES | COC1=CC=CC(C2=CC=C(N3C(Cl)=CC(NC(N4C5=CC=CC(OC)=C5)=O)=C3C4=O)C=C2)=C1O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Cell experiment [1]: | |
|
Cell lines |
MV4-11, OCI-AML3, OCI-AML2, HL-60, Kasumi, HEL, UT7, NB4, TF-1, KG1A, Nomo p28, SKM-1, U937, YHP1, MOLM-14, Mo7e, K562, MOLM-13, EOL-1 and SET-2 cell lines |
|
Preparation method |
This compound is soluble in DMSO. General tips for obtaining a higher concentration: Please warm the tube at 37 °C for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below - 20 °C for several months. |
|
Reacting condition |
30 μM; 4 days |
|
Applications |
In a set of 20 cell lines, GSK621 inhibited the proliferation of all 20 lines, with the IC50 values ranged from 13 to 30 μM, and promoted apoptosis in 17 cell lines. |
| Animal experiment [1]: | |
|
Animal models |
Mice bearing MOLM-14 cell xenografts |
|
Dosage form |
10 or 30 mg/kg; i.p.; b.i.d. |
|
Applications |
In mice bearing MOLM-14 cell xenografts, GSK621 (30 mg/kg; i.p.; b.i.d.) reduced leukemia growth, and substantially extended survival. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Sujobert P, Poulain L, Paubelle E, et al. Co-activation of AMPK and mTORC1 induces cytotoxicity in acute myeloid leukemia. Cell reports, 2015, 11(9): 1446-1457. |
|
Quality Control & MSDS
- View current batch:
Chemical structure
Related Biological Data
Related Biological Data