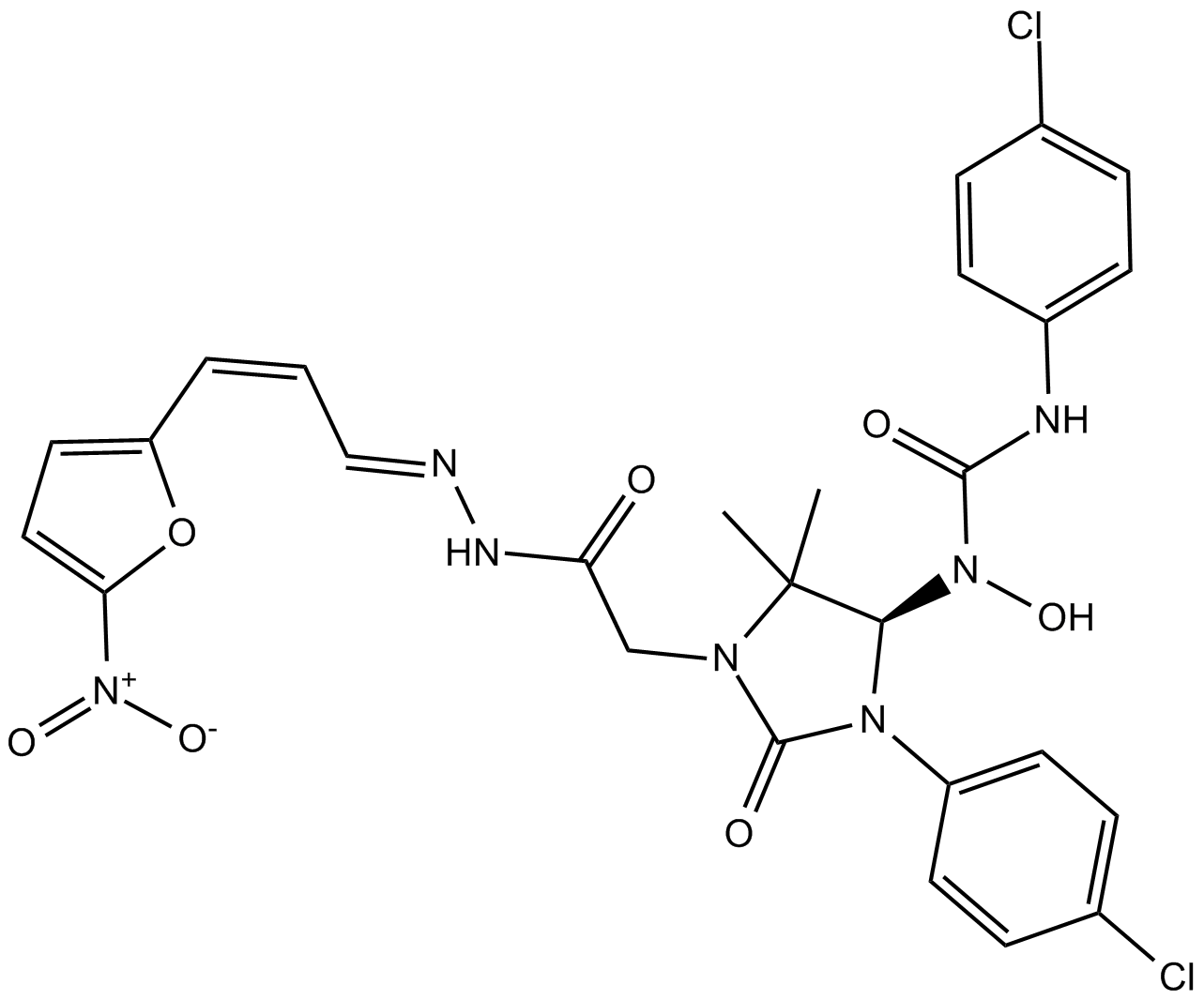
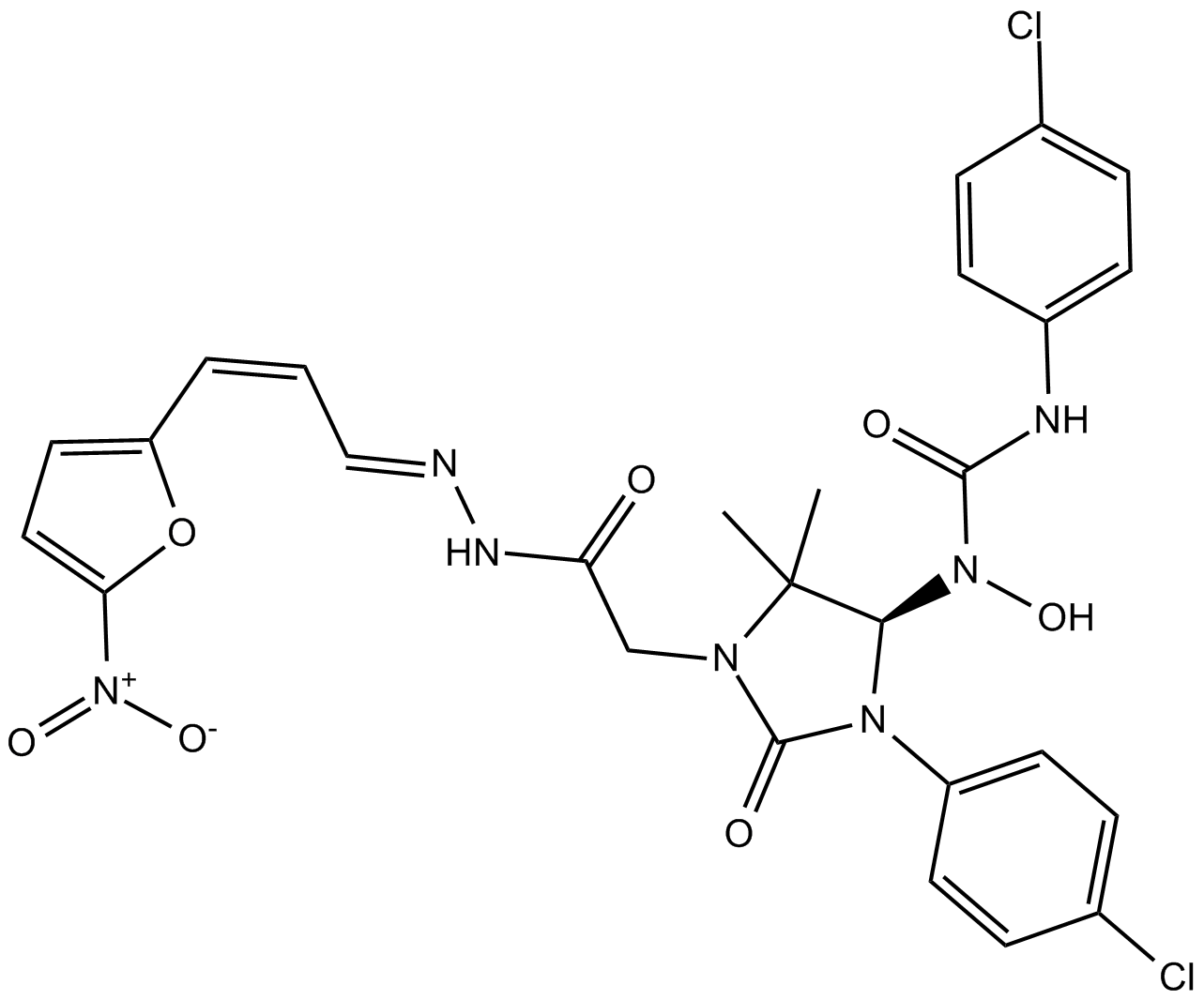
Eeyarestatin I
Eeyarestatin I (EerI [1]) is an inhibitor of endoplasmic reticulum-associated degradation (ERAD). It disturbs endoplasmic reticulum (ER) homeostasis and has anticancer activities resembling that of Bortezomib. ESI induced cell death in JEKO-1 cells with an IC50 of 4±1.2 µM [2]. ESI is also a potent inhibitor of protein translocation. The IC50 of ESI to ER translocation and N-glycosylation in vitro is ~70 µM [1].
ERAD pathway can eliminate misfolded ER proteins. Timely removing misfolded proteins from ER is involved in maintaining ER homeostasis [2]. N-Glycosylation is the most common and versatile protein modification, it occurs at the β-amide of the aspargine of the Asn-Xaa-Ser/Thr sequon [3]. ER mannosidase I could trigger the targeting of improperly folded glycoproteins to degradation [4].
In A9 cells, compared with Me2SO, treatment with EerI made polyubiquitinated MHC class I heavy chain (HC) accumulate in the cytosol. Compared with MG132, treatment with EerI resulted in very few deubiquitinated deglycosylated HC molecules, although a similar amount of accumulated polyubiquitinated HC [5]. In H1299 cells, treatment with EerI for 24 hrs significantly (p< 0.01) reduced cell proliferation (24.0%) as compared to the vehicle-treated control [6].
On the 2nd day and the 6th day after the subcutaneous injection of H1299 cells (8×106) complexed with Matrigel, athymic nude mice were treated with EerI (10 µM). EerI treatment significantly reduced tumor growth as compared to the DMSO vehicle control [6].
References:
[1]. Benedict C. S. Cross, Craig McKibbin, Anna C. Callan, et al. Eeyarestatin I inhibits Sec61-mediated protein translocation at the endoplasmic reticulum. Journal of Cell Science, 2009, 122:4393-4400.
[2]. Qiuyan Wang, Bidhan A. Shinkre, Jin-gu Lee, et al. The ERAD Inhibitor Eeyarestatin I Is a Bifunctional Compound with a Membrane-Binding Domain and a
p97/VCP Inhibitory Group. PLoS ONE, 2010, 5(11):e15479.
[3]. Shifra Ben-Dor, Nir Esterman, Eitan Rubin, et al. Biases and complex patterns in the residues flanking protein N-glycosylation sites. Glycobiology, 2004, 14(2):95-101.
[4]. Myriam Ermonval, Claudia Kitzmüller, Anne Marie Mir, et al. N-glycan structure of a short-lived variant of ribophorin I expressed in the MadIA214 glycosylation-defective cell line reveals the role of a mannosidase that is not ER mannosidase I in the process of glycoprotein degradation. Glycobiology, 2001, 11(7):565-576.
[5]. Qiuyan Wang, Lianyun Li and Yihong Ye. Inhibition of p97-dependent Protein Degradation by Eeyarestatin I. Journal of Biological Chemistry, 2008, 283(12):7445-7454.
[6]. Christopher W. Valle, Taehong Min, Manish Bodas, et al. Critical Role of VCP/p97 in the Pathogenesis and Progression of Non-Small Cell Lung Carcinoma. PLoS ONE, 2011, 6(12): e29073.
- 1. Xiaoxuan Li. "Hepatitis E virus ORF3 protein hijacking thioredoxin domain-containing protein 5 (TXNDC5) for its stability to promote viral particle release." J Virol. 2024 Apr 16;98(4):e0164923. PMID: 38548704
- 2. Xiaozhen Zhang, Mengyi Lao, et al. "Targeting cancer-associated fibroblast autophagy renders pancreatic cancer eradicable with immunochemotherapy by inhibiting adaptive immune resistance." Autophagy. 2024 Jun;20(6):1314-1334. PMID: 38174993
- 3. Yingying Shi, Chunqi Zhu, et al. "A Vaccination with Boosted Cross Presentation by ER‐Targeted Antigen Delivery for Anti‐Tumor Immunotherapy." Adv Healthc Mater. 2021 Jan 27;e2001934. PMID: 33502831
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 630.44 |
| Cas No. | 412960-54-4 |
| Formula | C27H25Cl2N7O7 |
| Solubility | Soluble in DMSO |
| Chemical Name | 3-(4-chlorophenyl)-1-((R)-3-(4-chlorophenyl)-5,5-dimethyl-1-(2-((E)-2-((Z)-3-(5-nitrofuran-2-yl)allylidene)hydrazinyl)-2-oxoethyl)-2-oxoimidazolidin-4-yl)-1-hydroxyurea |
| SDF | Download SDF |
| Canonical SMILES | ClC1=CC=C(C=C1)N2[C@@H](C(C)(C)N(CC(N/N=C/C=C\C3=CC=C([N+]([O-])=O)O3)=O)C2=O)N(C(NC(C=C4)=CC=C4Cl)=O)O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
-
Purity = 98.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
- Datasheet
Chemical structure