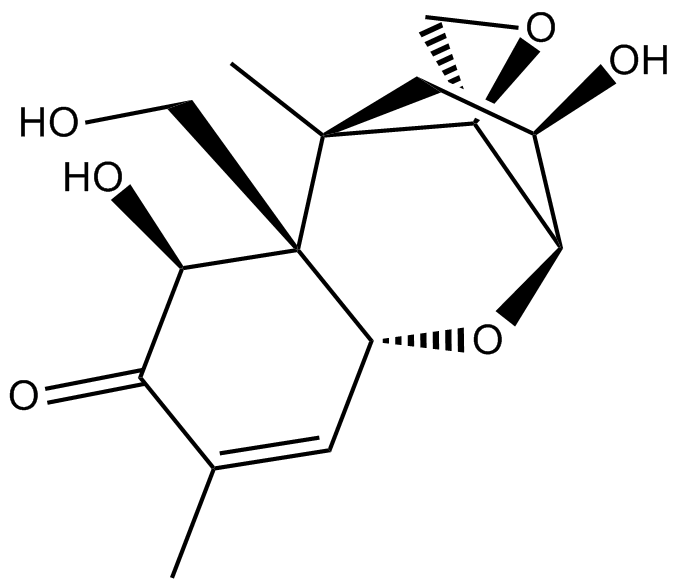
Deoxynivalenol
Deoxynivalenol (CAS 51481-10-8), commonly termed vomitoxin, is a mycotoxin produced by fungi and frequently found as a food contaminant. It primarily inhibits protein synthesis at the ribosomal level, inducing ribotoxic stress and subsequent cellular apoptosis. In vitro assays indicated cytotoxic response in human kidney epithelial cells at an EC50 of approximately 1.1 μM, and apoptosis induction in immune cells at concentrations ranging from 100 to 1000 ng/ml. In animal studies, exposure results in vomiting and impaired growth, immune regulation disturbances, and compromised physiologic barriers. Deoxynivalenol serves as a model compound for studying toxin-induced cellular stress and inflammation.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 296.32 |
| Cas No. | 51481-10-8 |
| Formula | C15H20O6 |
| Solubility | Soluble in DMSO |
| Chemical Name | (2R,2'S,3R,5R,5aR,6S,9aR)-3,6-dihydroxy-5a-(hydroxymethyl)-5,8-dimethyl-2,3,4,5,5a,6-hexahydrospiro[2,5-methanobenzo[b]oxepine-10,2'-oxiran]-7(9aH)-one |
| Canonical SMILES | C[C@](C[C@H]([C@H]1O[C@@H]2C=C(C)C3=O)O)([C@@]11OC1)[C@@]2(CO)[C@@H]3O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure