CJ-42794
CJ-42794 (CAS 847728-01-2) is a potent and selective antagonist targeting prostaglandin E receptor subtype 4 (EP4). It exhibits high affinity for EP4 receptors (Ki: 3.16 nM) with significantly lower potency at EP2 receptors (Ki: 631 nM), and negligible affinity for EP1 or EP3. By blocking EP4-mediated signaling, CJ-42794 prevents prostaglandin E2-induced intracellular cAMP elevation, thereby reversing PGE2-dependent inhibition of inflammatory responses, such as lipopolysaccharide-induced TNF-α secretion. In animal models, administration of CJ-42794 delays gastric ulcer healing through suppression of VEGF expression and angiogenesis. This compound serves as a valuable tool for investigating EP4-associated physiological and pathological processes.
| Storage | Store at -20°C |
| M.Wt | 413.8 |
| Cas No. | 847728-01-2 |
| Formula | C22H17ClFNO4 |
| Synonyms | RQ-00015986 |
| Solubility | insoluble in H2O; ≥11.7 mg/mL in DMSO; ≥54.6 mg/mL in EtOH |
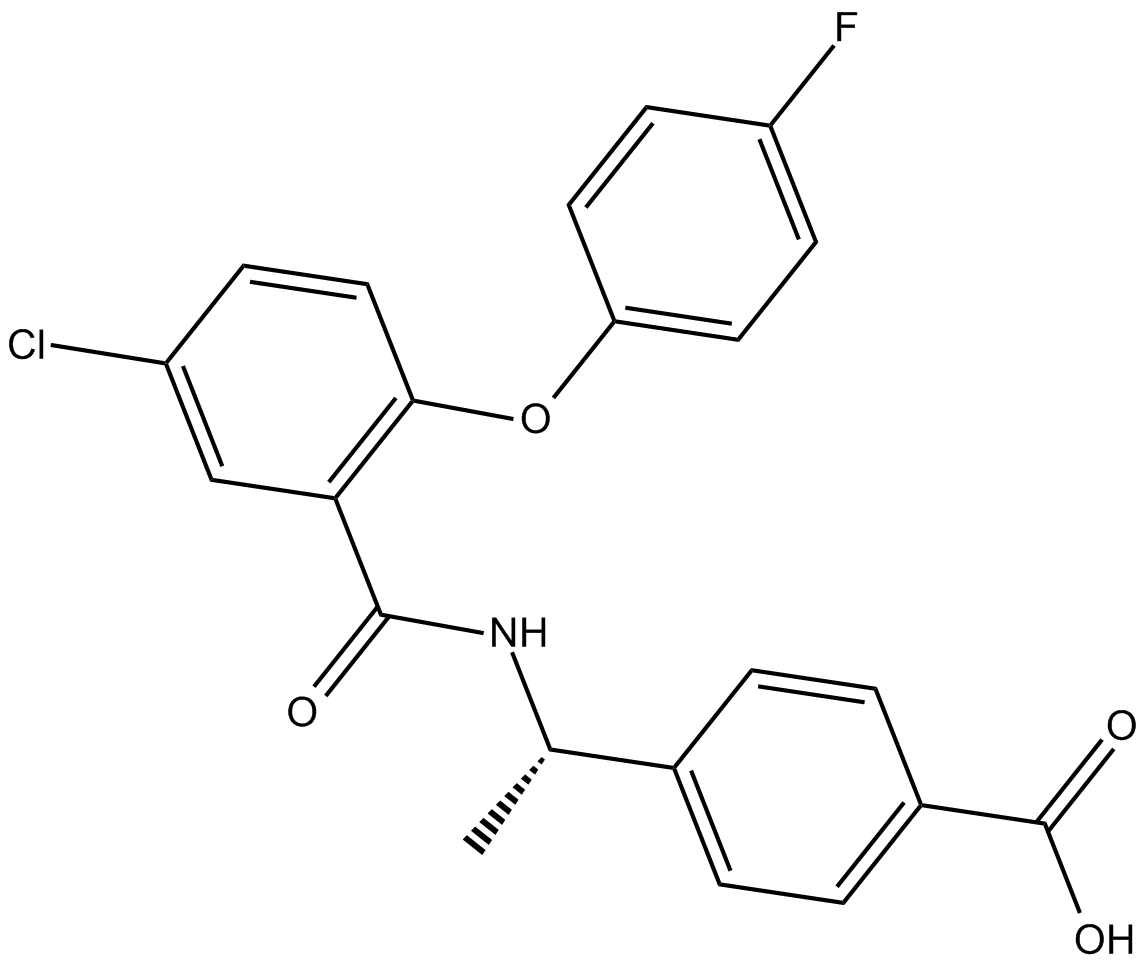
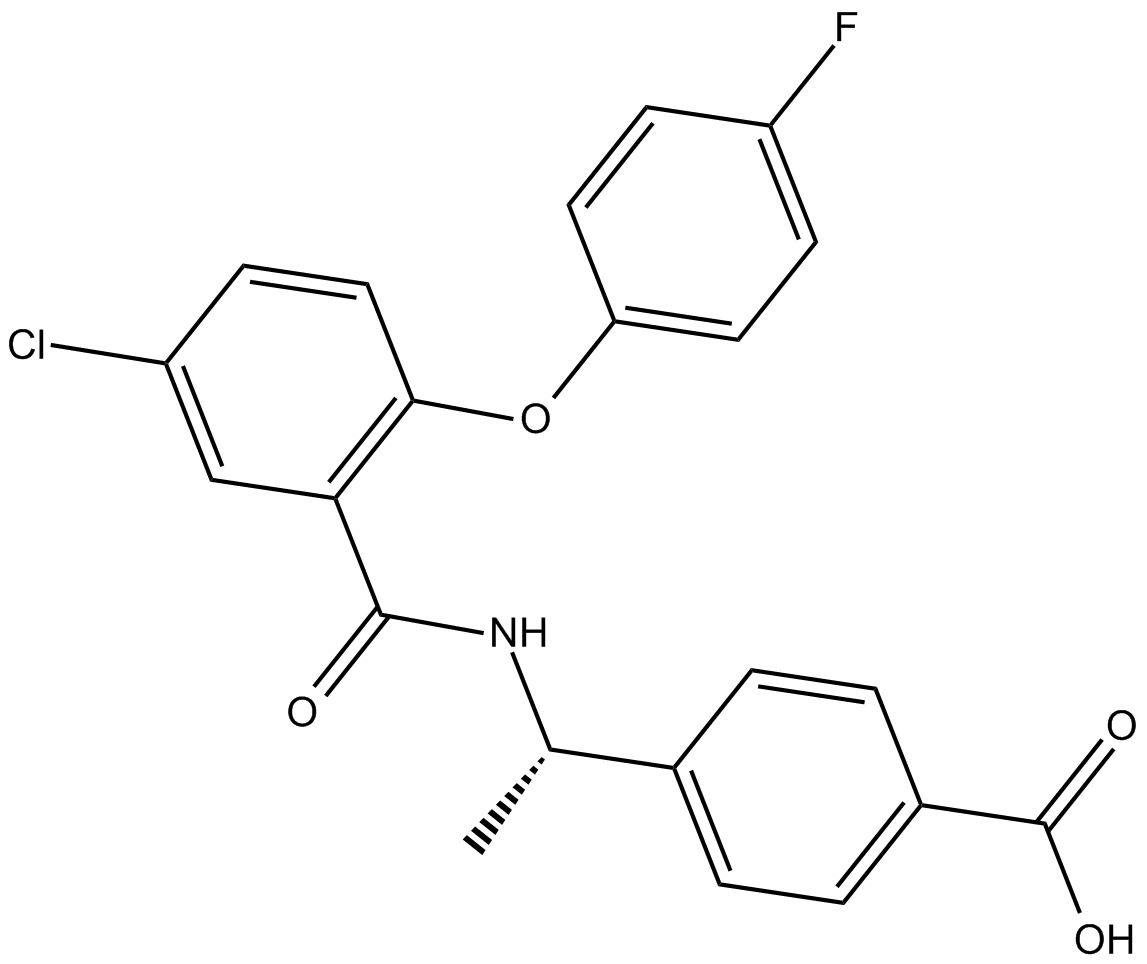
| Chemical Name | 4-[(1S)-1-[[5-chloro-2-(4-fluorophenoxy)benzoyl]amino]ethyl]-benzoic acid |
| SDF | Download SDF |
| Canonical SMILES | FC1=CC=C(OC2=C(C(N[C@@H](C)C3=CC=C(C(O)=O)C=C3)=O)C=C(Cl)C=C2)C=C1 |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure