CHIR-090
CHIR-090 is a very potent, low, tight-binding inhibitor of LpxC with Ki value of 4.0 nM [1].
LpxC is a zinc-dependent amidase and present in almost all Gram-negative bacteria. LpxC is a promising target for the development of novel antibiotic substances against multigrug-resistant Gram-negative bacteria [2].
CHIR-090 is a potent LpxC inhibitor and has a different selectivity with the reported LpxC inhibitor L-161. When tested with Escherichia coli LpxC, administration of CHIR-090 showed tight inhibition with Ki value of 4.0 nM, Ki*=0.5 nM, K5=1.9/min and K6=0.18/min [1]. In bacterial P.aeruginosa efflux pupm mutants, CHIR-090 treatment showed inhibition function on MexAB-Oprm, MexCD-OprJ and MexEF-OprN [2]. CHIR-090 showed remarkable antibiotic activity against both E.coli and P.aeruginosa by inhibiting LpxC orthologs at low nM concentrations [3].
In E.coli W3110RL with R.legumunosarum lpxC replacement of E.coli lpxC, CHIR-090 (1 to 10 μg/ml) treatment had no effect on strain growth on LB agar plates while wild-type cells stopped growing after about 2 h in the presence of 1 μg/ml CHIR-090 [1].
References:
[1].Barb, A.W., et al., Inhibition of lipid A biosynthesis as the primary mechanism of CHIR-090 antibiotic activity in Escherichia coli. Biochemistry, 2007. 46(12): p. 3793-802.
[2].Barb, A.W. and P. Zhou, Mechanism and inhibition of LpxC: an essential zinc-dependent deacetylase of bacterial lipid A synthesis. Curr Pharm Biotechnol, 2008. 9(1): p. 9-15.
[3].McClerren, A.L., et al., A slow, tight-binding inhibitor of the zinc-dependent deacetylase LpxC of lipid A biosynthesis with antibiotic activity comparable to ciprofloxacin. Biochemistry, 2005. 44(50): p. 16574-83.
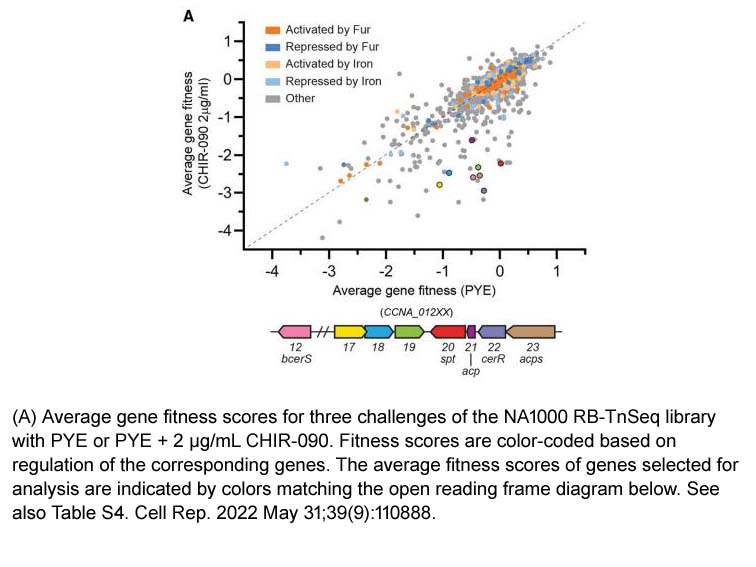
- 1. Justin J. Zik, Sung Hwan Yoon, et al. "Caulobacter lipid A is conditionally dispensable in the absence of fur and in the presence of anionic sphingolipids." Cell Rep. 2022 May 31;39(9):110888. PMID: 35649364
- 2. Basta DW, Angeles-Albores D, et al. "Heat-shock proteases promote survival of Pseudomonas aeruginosa during growth arrest." Proc Natl Acad Sci U S A. 2020;117(8):4358–4367. PMID: 32029587
- 3. David W. Basta. "Genetic Determinants of Growth Arrest Survival in the Bacterial Pathogen Pseudomonas aeruginosa and the Role of Proteases." CALIFORNIA INSTITUTE OF TECHNOLOGY.2019.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 437.49 |
| Cas No. | 728865-23-4 |
| Formula | C24H27N3O5 |
| Synonyms | CHIR 090;CHIR090 |
| Solubility | ≥21.85 mg/mL in DMSO; insoluble in EtOH; insoluble in H2O |
| Chemical Name | N-[(2S,3R)-3-hydroxy-1-(hydroxyamino)-1-oxobutan-2-yl]-4-[2-[4-(morpholin-4-ylmethyl)phenyl]ethynyl]benzamide |
| SDF | Download SDF |
| Canonical SMILES | CC(C(C(=O)NO)NC(=O)C1=CC=C(C=C1)C#CC2=CC=C(C=C2)CN3CCOCC3)O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Kinase experiment [1]: | |
|
Assay of LpxC Activity |
UDP-3-O-(R-3-hydroxymyristoyl)-N-acetylglucosamine and [α-32P] UDP-3-O-(R-3-hydroxymyristoyl)-N-acetylglucosamine were prepared enzymatically as previously described. Assays of LpxC activity were performed with 5 μM substrate, except where noted; additionally, 10% DMSO was added to the assay mixtures and held constant at that level when inhibitor (dissolved in DMSO) was added. Except where noted, the concentration of the enzyme was at least 10-fold less than the concentration of either the inhibitor or the substrate. When pre-incubated with or without inhibitor prior to being assayed, the enzyme was diluted in 25 mM sodium phosphate, pH 7.4, containing 1 mg/mL BSA and 10% DMSO. The pre-incubation mixture was held on ice for 15 min before the reaction was initiated by means of a 1:4 dilution of the enzyme into the assay cocktail. Initial velocities were calculated from the linear portion of reaction progress curves ( |
| Cell experiment [1]: | |
|
Cell lines |
wild-type E. coli W3110 and E. coli W3110RL |
|
Preparation method |
The solubility of this compound in DMSO is >21.9mg/mL. General tips for obtaining a higher concentration: Please warm the tube at 37℃ for 10 minutes and/or shake it in the ultrasonic bath for a while. Stock solution can be stored below -20℃ for several months. |
|
Reacting condition |
1 μg/mL |
|
Applications |
In the presence of 1 μg/mL CHIR-090, wild-type E. coli W3110 stopped growing after about 2 h. The E. coli W3110RL, in which R. leguminosarum lpxC replaces the chromosomal copy of E. coli lpxC, was resistant to CHIR-090. The MIC of CHIR-090 against W3110RL in liquid medium was 100 μg/mL, compared to 0.25 μg/ mL for W3110. |
|
References: [1].Barb, A.W., et al., Inhibition of lipid A biosynthesis as the primary mechanism of CHIR-090 antibiotic activity in Escherichia coli. Biochemistry, 2007. 46(12): p. 3793-802. |
|
| Description | CHIR-090 is a potent inhibitor of LpxC | |||||
| Targets | LpxC | bacterial | ||||
| IC50 | ||||||
Quality Control & MSDS
- View current batch:
Chemical structure

Related Biological Data

Related Biological Data