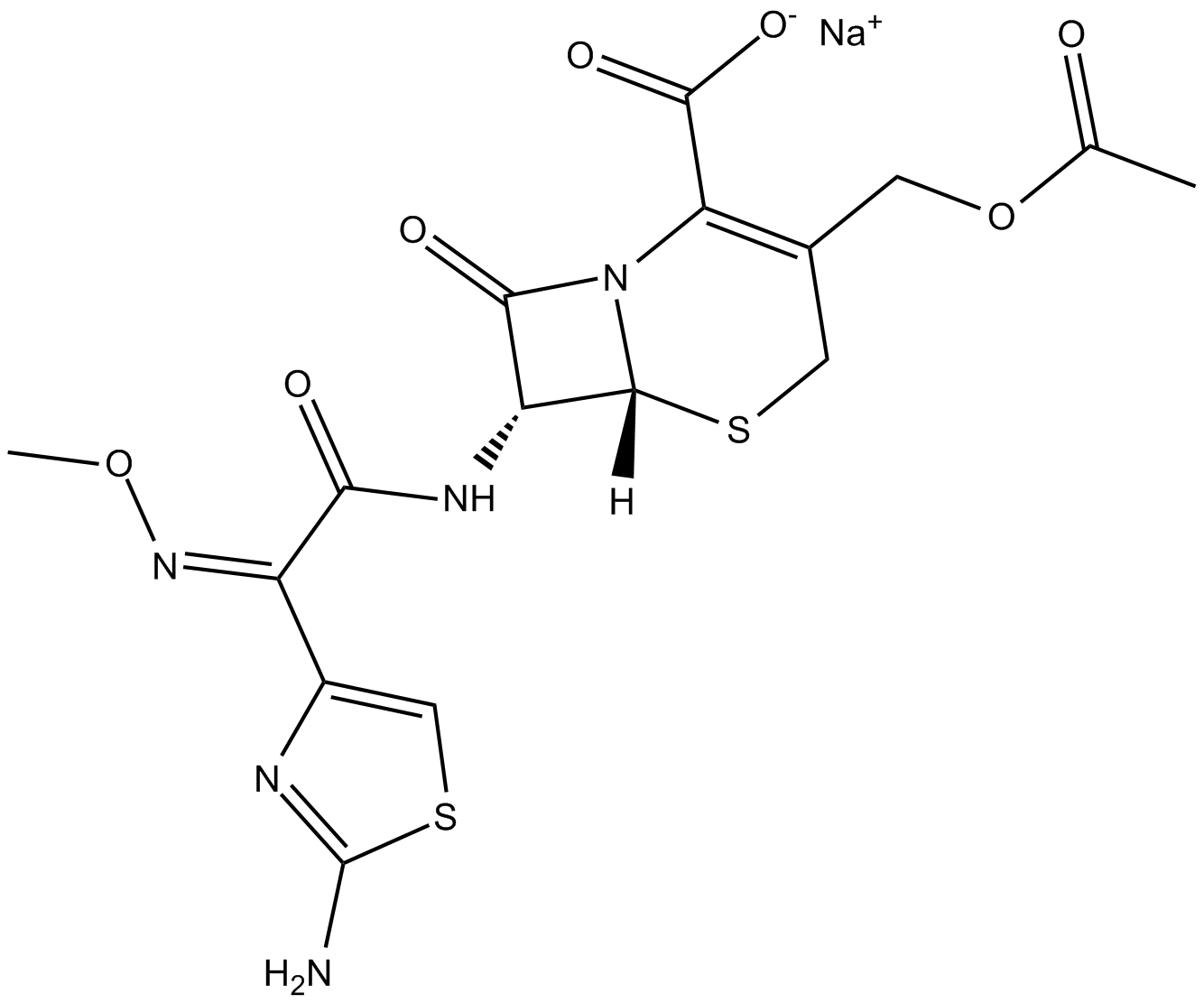
Cefotaxime (sodium salt)
Cefotaxime sodium (CAS 64485-93-4) is a third-generation cephalosporin antibiotic belonging to the β-lactam group, characterized by inhibition of bacterial cell-wall synthesis through binding to penicillin-binding proteins. In vitro analyses show strong inhibitory efficacy against various bacterial species; notably, cefotaxime exhibits an MIC below 0.1 μg/ml against Streptococcus pneumoniae. It also demonstrates high activity against Haemophilus influenzae, including β-lactamase-producing strains. In vivo mouse model studies revealed that combination therapy of cefotaxime with minocycline enhanced survival following Vibrio vulnificus infection compared to single-agent treatments. Clinically, cefotaxime has been evaluated in pneumonia therapy, demonstrating good tolerance and therapeutic utility in targeting susceptible bacterial pathogens.
| Storage | Store at -20°C |
| M.Wt | 477.5 |
| Cas No. | 64485-93-4 |
| Formula | C16H16N5O7S2·Na |
| Solubility | ≥18.8 mg/mL in DMSO |
| Chemical Name | (6R)-3-[(acetyloxy)methyl]-7R-[[(2Z)-2-(2-amino-4-thiazolyl)-2-(methoxyimino)acetyl]amino]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid, monosodium salt |
| Canonical SMILES | CC(OCC(CS[C@@H]1[C@@H]2NC(/C(\c3c[s]c(N)n3)=N\OC)=O)=C(C([O-])=O)N1C2=O)=O.[Na+] |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
Quality Control & MSDS
- View current batch:
Chemical structure