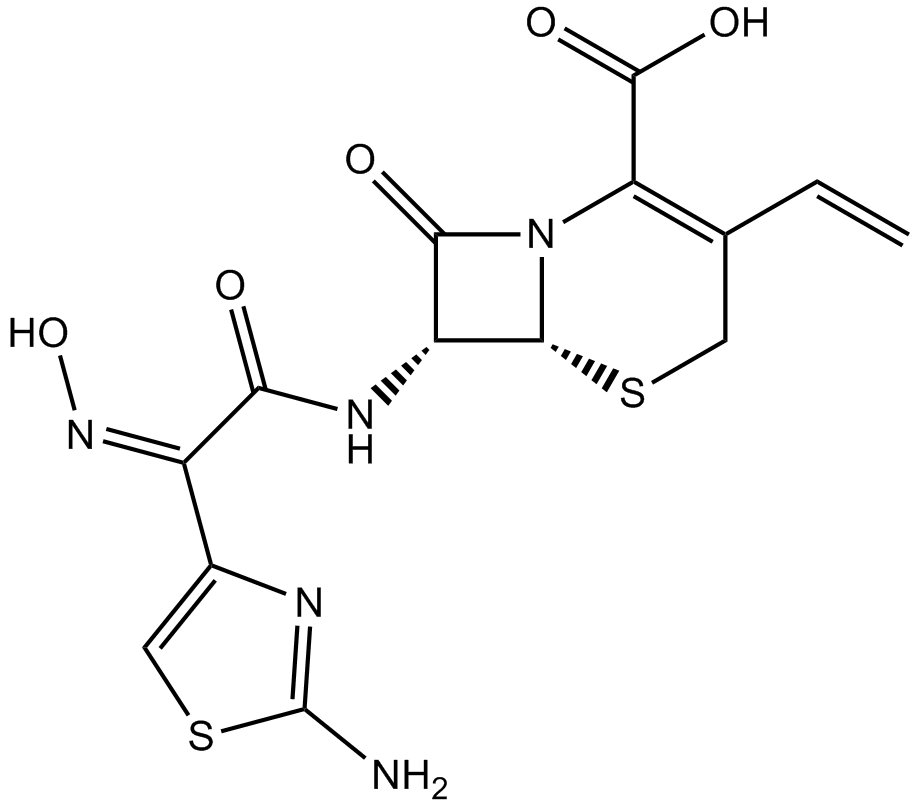
Cefdinir
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 395.41 |
| Cas No. | 91832-40-5 |
| Formula | C14H13N5O5S2 |
| Solubility | insoluble in EtOH; insoluble in H2O; ≥19.55 mg/mL in DMSO |
| Chemical Name | (6R,7R)-7-[[(2Z)-2-(2-amino-1,3-thiazol-4-yl)-2-hydroxyiminoacetyl]amino]-3-ethenyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid |
| SDF | Download SDF |
| Canonical SMILES | C=CC1=C(N2C(C(C2=O)NC(=O)C(=NO)C3=CSC(=N3)N)SC1)C(=O)O |
| Shipping Condition | Small Molecules with Blue Ice, Modified Nucleotides with Dry Ice. |
| General tips | We do not recommend long-term storage for the solution, please use it up soon. |
| Animal experiment [1]: | |
|
Animal models |
Acute kidney injury in male Wistar rat models |
|
Dosage form |
10 mg/kg(p.o.) |
|
Application |
After cisplatin administration, rats were orally administered cefdinir on day 6. It was observed that cefdinir significantly increased plasma exposure compared to the control group. At the same time, cumulative urinary excretion was significantly reduced in the cisplatin-induced AKI group. It could be estimated that morbid condition could significantly affect cefdinir's release in vivo. These results indicated that under the conditions of cisplatin-induced AKI, the pharmacokinetics of cefdinir changed significantly. |
|
Other notes |
Please test the solubility of all compounds indoor, and the actual solubility may slightly differ with the theoretical value. This is caused by an experimental system error and it is normal. |
|
References: [1]. Wang H, Sun P, Wang C, Meng Q, Liu Z, Huo X, Sun H, Ma X, Peng J, Liu K. Pharmacokinetic changes of cefdinir and cefditoren and its molecular mechanisms in acute kidney injury in rats. J Pharm Pharmacol. 2018 Nov;70(11):1503-1512. doi: 10.1111/jphp.12994. Epub 2018 Jul 25. PMID: 30047127. |
|
Quality Control & MSDS
- View current batch:
Chemical structure